Abstract
Tropical areas are vital for conserving endangered tree species. However, barriers to the propagation and storage of plant material still limit preservation outcomes. Seed traits are essential in conservation but have rarely been applied in the tropics. In this work, we compile information about the germination and seed desiccation tolerance behaviour of 953 arboreal species from the Osa Peninsula, Costa Rica. We analysed how those traits are related to the growing strata, seed size, and dispersal mode and how they can be used to improve conservation initiatives based on the species distribution, abundance, extinction risk, and ex situ cultivation. Over a third of the species are probably recalcitrant but this number increase to almost half among the endangered and endemic ones. Most species have seeds dispersed by animals, but wind dispersal is usually orthodox and dominant in secondary forests. All species with tiny seeds and most understorey ones with small seeds are orthodox. Approximately half of the species have dormant seeds, while nearly one-third of nondormant species have cohorts with extended germination. Almost half of the species are conserved ex situ; however, several endangered, endemics could be expected to be considered exceptional. Seed traits could target orthodox species for seed banking in tropical forests, including the potential of secondary forests and a subset of oligarch species. For the recalcitrant ones, approaches based on the risk of extinction, distribution, abundance, and ex situ status allow for prioritizing collection and conservation strategies. Under this approach, we develop a list with priority species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plant diversity loss has become a significant concern. According to a recent estimate, 39% of vascular plants and a third of tree species are threatened with extinction (Lughadha et al. 2020; BGCI 2021a). In the short term, this trend appears to be continuing because extinction drivers, such as habitat loss, deforestation, invasive species, and global warming, are still increasing. Without urgent actions, many species could disappear during the next few centuries (Barnosky et al. 2011). Worldwide initiatives to protect endangered taxa and restore landscapes for conservation have become a priority. For example, Target 8 of the Global Strategy for Plant Conservation (GSPC) states that “75% of threatened plant species are held in ex situ collections, preferably in the country of origin and with at least 20% available for recovery programs” (Sharrock 2012). Moreover, target 12 from Aichi Biodiversity aims to prevent the extinction of threatened species (CBD 2020), and by 2030, the Bonn challenge aims to restore 350 million hectares of deforested and degraded landscapes (UICN 2020).
Due to the high level of biodiversity, tropical areas have become priority sites in which one main challenge is to protect the high levels of diversity. For example, tree species in the Neotropics are estimated to be ca. 24 thousand (BGCI 2021a). A fundamental but underused tool in the tropics that could guide conservation efforts is the knowledge of seed traits, especially regarding germination, and desiccation tolerance (Ribeiro et al. 2016; Kildisheva et al. 2016). Seed dispersal and germination are crucial for natural forest regeneration and restoration (Cole et al. 2011; Werden et al. 2020a). Similarly, seed desiccation tolerance is the most crucial trait in seed bank conservation. Orthodox seeds tolerate intense desiccation and survive freezing. By contrast, recalcitrant seeds do not tolerate desiccation levels below 75% relative humidity at 15 °C, are short-lived, and, therefore, cannot be stored in the long term in seed banks with conventional methods. Recently, Pence et al. (2022a, b) developed a framework for defining exceptional species for ex situ conservation based on four seed traits: 1. species in which seeds do not occur or are extremely difficult to obtain; 2. species with recalcitrant seeds; 3. species with seeds that cannot survive long-term storage; or 4. species with deeply dormant seeds. Following these criteria, many tropical species could be expected to be considered exceptional.
Most woody tropical species have very low abundances, making collecting seeds with adequate genetic variability difficult. Even if sources are available, collection at the wrong time, unviable or poor-quality seeds, lack of knowledge on ideal germination conditions or how to break dormancy, and poor dispersal in the field can result in low seedling establishment and poor conservation outcomes (Merritt and Dixon 2011; Abeli et al. 2020). Moreover, tropical forests are characterized by a high proportion of recalcitrant species, especially among woody, canopy, or large-seeded species (Tweddle et al. 2003; Wyse et al. 2018). Estimates have shown that approximately 8% of world plant species are recalcitrant (Dickie and Pritchard 2002; Wyse and Dickie 2017), increasing to almost a third in warm tropical regions (Lima et al. 2014; Wyse and Dickie 2018). Since no data are available for most species, decision-making keys (Sommerville et al. 2021) and models based on different traits have been provided as guidance on possible seed desiccation behaviour (Daws et al. 2006; Lima et al. 2014; Wyse and Dickie 2018). Seed longevity has been associated with dispersal mode, moisture content, seed size, growth form, and growing environment (Mondoni et al. 2011; Hoyle et al. 2015; Solberg et al. 2020; Wang et al. 2021). Often, the behaviour of relative taxa at the genus level is a good predictor of desiccation tolerance when no other information is available (Wyse and Dickie 2017, 2018).
Many tropical species also have dormancy or long, erratic germination patterns (Garwood 1983; Sautu et al. 2006). For example, in a study of 94 species from the moist tropical forest in Panama, almost half of the species were dormant (Sautu et al. 2007). Even for the nondormant ones, there is often a continuous germination response even in the same cohort with extended germination over time (Blakesley et al. 2002; Sautu et al. 2006). An additional characteristic of tropical areas is the high proportion of species dispersed by animals. Although this is expected due to the richness of fauna, the role of animal digestion in germination has shown variable results between species (Wehncke and Dalling 2005; Saldaña-Vázquez et al. 2019). After a review of the literature from 366 species, Soltani et al. (2018) found a strong relationship between the type of dormancy and the effect on germination after animal ingestion. As such, for nondormant species, ingestion by animals might be more of a requirement for dispersal than germination (Saldaña-Vázquez et al. 2019).
The recognized importance of seed traits has led to increased knowledge of manipulating seeds exclusively for restoration purposes (Blakesley et al. 2002; Núñez-Cruz et al. 2018; Kildisheva et al. 2020). As a result, guidelines have been developed to sample seeds (Luna-Nieves et al. 2019), evaluate their germination (De Santana et al. 2012), and measure their quality (Pedrini and Dixon 2020). This has been accompanied by an increase in the number of studies focused on finding the ideal germination requirements (Aud and Ferraz 2012; Sánchez et al. 2015; Valverde-Rodríguez et al. 2019) and desiccation tolerance (Lima et al. 2014; Chau et al. 2019) of wild species. Ex situ initiatives have been encouraged, with seed banks being one of the main strategies due to their low cost and high efficiency. For example, in Europe, 62% of threatened species are already conserved in seed banks (Rivière and Müller 2018), while The Millennium Seed Bank have conserved almost 40 000 taxa from which 10% are either endangered or extinct in the wild (Liu et al. 2018). Despite several limitations, there is also a high capacity and potential of seed banks for ex situ conservation in many tropical countries (León-Lobos et al. 2012). As they increase their role as centres of plant conservation, seed preservation is also becoming an essential tool in botanical gardens (O’Donnell and Sharrock 2017). It has been estimated that 41% of threatened species are already present in botanical gardens (Mounce et al. 2017), with underrepresentation in those of tropical origin (Mounce et al. 2017; Da Costa et al. 2018).
Since studying the seed characteristics of every tropical species is still unfeasible, analysing the seed traits of forest communities could be used as an approach for decision-making. For example, a review of extremely endangered species in China estimated that approximately 60% of them will require cryopreservation for successful ex situ conservation (Wade et al. 2016). A study on the dry forests of Ecuador and Peru found that most species are orthodox and dormant and could be stored with conventional methods but require pre germination treatments for their use (Romero-Saritama and Pérez Ruiz 2016). Similarly, Galindo-Rodriguez and Roa-Fuentes (2017) concluded that most of the tree species of the Colombian dry forest are dispersed by animals, which could become a problem for restoration and dispersal in fragmented or degraded sites. The latter authors also provided a list of recommended species for forest restoration based on seed traits. However, very few reviews about seed traits for conservation purposes are available for humid tropical forests (Chau et al. 2019). Moreover, none of them has taken into account the abundance and distribution of the species and their ex situ conservation status to develop a conservation strategy. Here, we analysed the current information about seed traits regarding desiccation tolerance and germination patterns for the arboreal flora from the Osa Peninsula (OP) and Golfo Dulce Region (GDR), a hotspot of biodiversity in southern Costa Rica. We analysed how these data are linked with the seed size, mode of dispersal, and forest strata in which the species grows. Then, we discuss how this information could be used to improve conservation efforts based on the risk of extinction, geographical distribution, local abundance, and its current ex situ conservation status. Our goal is to provide a case study based on seed traits to serve as an example that could improve any other plant conservation and restoration effort in humid tropical rainforests.
Materials and methods
Study area: The Golfo Dulce Region and the Osa Peninsula are in Southwestern Costa Rica, Central America, and comprise an area of approximately 150 000 ha, most of it under some category of protection (Fig. 1a,b). The average annual precipitation in different places of the peninsula ranges between 3000 and 7000 mm, with an average temperature of 24–26 °C. The area is the largest remaining tropical forest in the Pacific region of Central America. It is considered a hotspot of diversity with frequent records of new plant species, including canopy trees (e.g., Santamaría-Aguilar et al. 2021). The site has been relatively well explored in botanical terms, especially regarding trees (Hofhansl et al. 2019). There are current lists available for the flora in the region (Weber et al. 2001; Aguilar-Fernández et al. 2017). The arboreal flora has a strong affinity with the flora of Northwest South America, but more than half of the species are also found in Mesoamerica (Cornejo et al. 2012). There are at least two long-term restoration projects on the site that aim to recover degraded landscapes and promote the connection of biological corridors in the region by using native species (Weissenhofer et al. 2019). Detailed information about the area's weather, vegetation, topography, and soils can be found in (Weissenhofer et al. 2008).
Map of the study area and example species: the Osa Peninsula and Golfo Dulce Region, Costa Rica (a, b). Osa pulchra, an endangered and scarce species from Costa Rica and Panama widely conserved ex situ (c). Desmopsis heteropetala, a species restricted to Costa Rica, is not conserved ex situ and has no data about seed germination or desiccation tolerance (d). Licania corniculata, a scarce, endangered species with recalcitrant seeds, endemic to the Osa Peninsula and not conserved ex situ (e). Terminalia amazonia, a widely distributed species, is very common in secondary forests and has orthodox seeds (f). Photos: b Daniel Davies, all other ECM
Species and seed traits database: Arboreal species were taken from the checklists from (Weber et al. 2001) and (Aguilar-Fernández et al. 2017) and were complemented with the field work results from (Morera-Beita et al. 2019). We selected the species catalogued as shrubs, palms, or trees with a reported height higher than 1.5 m. Taxonomic names were checked against “The World Flora Online” (WFO 2022) and based on the species descriptions available on this website, we recorded the seed size and plant height. Seeds were categorized as tiny (< 0,5 cm), small (0.55–0.99 cm), medium (1–1.99 cm), and large (> 2 cm) following (Cornejo and Janovec 2010). Based on maximum plant height, species were categorized as belonging to the understorey (< 10.0 m), subcanopy/canopy (10.01–45.0 m) or emergent (> 45.0 m). The average height was used for the species with overlapping height ranges to categorize between understorey or subcanopy/canopy strata. The seed and species traits collected for this database can be found in Table 1. When no data were available for the seed size, mode of dispersal, or height, additional searches were performed by reviewing the literature and online herbarium websites. However, that information was searched only for those species in which we found information about seed germination or desiccation tolerance. Seed size was also complemented by the work of Sandor (2002).
Information about desiccation tolerance was taken online from the seed storage predictor from Wyse and Dickie (2018) [available at: https://seedcollections.shinyapps.io/seed_storage_predictor/]. The authors provide a set of models that gives the probability of seed desiccation tolerance based on several traits but in which the taxonomy at the lowest level gives the highest reliability (see Wyse and Dickie 2018 for details). Values closer to 0 are likely orthodox, and values closer to one are expected to be recalcitrant. In our study, to increase the certainty, we classified as “possible orthodox” or “possible recalcitrant” only those species with values higher than 0.8 or lower than 0.2 for models run at the genus and family levels. Data from the mode of dispersal were collated from the database of the Royal Botanic Gardens, Kew (RBGK 2021). Due to the low number of records available on seed dispersal, we classified the dispersal mode based on what has been reported within the genus, since seed dispersal is a relatively conserved trait throughout phylogeny (Kuhlmann and Ribeiro 2016). Germination and dormancy data were searched in Baskin and Baskin (2014) and references therein and later in Google Scholar for all the species by using the scientific name plus “germination” and “seeds” as search terms. When available, we compiled data about specific pregermination treatments, percentage of germination (% G), mean length of germination (MLG), time to reach 50% germination (T50), and first and last day of germination. Based on these indices, we classified species as germination as uncertain (if not enough data were available), dormant or non-dormant (non-dormant if % G was higher than 40% and other variables on average shorter than 30 days) or if the species were previously cataloged as dormant or non-dormant.
Conservation parameters: We define the conservation priorities and scopes for each species based on their geographic distribution, abundance at the site, the IUCN conservation status, and presence at ex situ sites. Distribution was taken from the Tropicos website (Missouri Botanical Garden 2022) and categorized based on regions using Costa Rica and adjacent countries as the limit of the distribution to the north and south (A: Osa region, B: Costa Rica, C: Costa Rica and neighbouring countries, D: Mesoamerica to the south until Panama, E: Colombia up to Nicaragua, and F: Widespread, when the distribution was more significant than the previous categories). Abundance was obtained based on Morera-Beita et al. (2019). This work identified approximately 10,000 individuals with dbh > 10 cm found in 20 plots of one hectare located in 4 habitats (ridge, slope, ravine, secondary forest). According to the abundance in each habitat and plots, species were classified as follows: (a) oligarch: species that were found in at least half of the plots and contributed to the accumulated 50% of all individuals encountered; (b) common: species with more than one individual per hectare and with a frequency higher than 25% of the plots (excluding oligarchs); (c) rare: species with a density lower than 1 individual per hectare and/or with a frequency lower than 25% of the plots; (d) very rare: species which were found only once (Morera-Beita et al. 2019). We excluded understory species from the abundance data, as many of these species may have a diameter of less than 10 cm. Species with a reported height higher than 10 m with no records in the plots were classified as not recorded. However, for a conservation approach, we also considered these species to be scarce. We assessed the conservation status in The IUCN Red List of Threatened Species (IUCN 2021), where we also checked if the species are reported to be subject to ex situ conservation. In addition, we obtained the number of sites worldwide where each species is reported as preserved ex situ from the Botanic Gardens Conservation International database (BGCI 2021b). Trees were classified as a timber source or with other human uses based on Quesada-Monge and Fernández-Vega (2005). Finally, we developed a list of priority species for ex situ conservation by assigning one point when (1) the species is scarce in the area (rare or very rare); (2) it has a limited distribution (Nicaragua to Panama); (3) it is considered DD, EN or CR by the IUCN; or (4) it has recalcitrant seeds. For the latter category we assigned a half point for species with no information about seed desiccation behaviour. Species already recorded as conserved ex situ were excluded from the list, while higher priority was given to species with more points assigned. The whole database is available as Online Resource 1, while the priority species list with each category is provided as Online Resource 2.
Results
Storage behaviour, germination, and seed traits
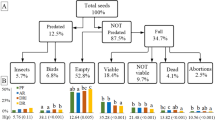
We reviewed information for 953 species distributed in 368 genera and 91 families. There are records of the storage behaviour for 160 (16.79%) species, 176 (47.82%) genera, and 78 (85.71%) families. We assigned 106 (11.1%) of the species as orthodox (O), 319 (33.5%) as possible orthodox (O?), 51 (5.4%) as recalcitrant, and 228 (23.9%) as possible recalcitrant (R?; Fig. 2a). When considering all the models disregarding the probability of desiccation, 375 (39.35%) of the species are considered recalcitrant. We found information about the germination behaviour for 286 species, with only 99 species having detailed germination studies (Table 2, Online Resource 1). We classified 117 species as nondormant (12.3%) and 111 as dormant (11.6%), of which only 28 had information available on how to break dormancy or improve germination (Table 2). In 58 species, it was not possible to determine the presence or absence of dormancy since there was not enough data available or there were different results between authors. Two hundred twenty-four (91.5%) species with records of % G have values higher than 40% (Table 2). However, it should be noted that only 146 species have a reported MLG, which ranges from 2 to 263 days (Online Resource 1). One hundred forty-eight (65.77%) species with whole trajectories available showed completion of germination longer than 30 days, including 32 classified as nondormant (Table 2, Online Resource 1).
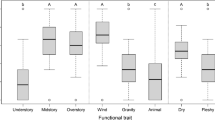
We found information about seed size for 675 species. Most of the surveyed species (704; 87.89%) are animal dispersed, and 59 (7.36%) are wind dispersed. All 26 wind-dispersed species with information on desiccation tolerance are orthodox (Fig. 2b). Similarly, all species with tiny seeds (47 from 47) and all dominant species from the secondary forest (11 from 11) with information available were also orthodox despite being mostly animal dispersed (Fig. 2). Species with small seeds (21 from 29), from the understorey (29 from 34), and emergent trees (9 from 11) also tended to be orthodox (Fig. 2 c,d). However, most of the species from the understorey with these traits belong to Melastomataceae or Rubiaceae, and half of the emergent trees were wind dispersed (Online Resource 1). All the above mentioned patterns were maintained when using species with extrapolated behaviour from the seed desiccation tolerance model. For instance, all additional 26 additional wind-dispersed species, 107 of 108 with tiny seeds and seven of eight species from the secondary forest were likely to be orthodox (Fig. 2). Seed size was not a suitable parameter to infer desiccation behaviour among medium and large seeds (Fig. 2c). Dormancy was not related to storage behaviour. It was distributed almost equally among all categories (Table 2). Of the 94 dormant species with information on desiccation tolerance, 19 and 28 were found to be recalcitrant or possibly recalcitrant (Table 2).
Conservation traits
Based on the abundance, 27 tree species from the region are considered oligarchs, 80 as common, 243 as rare, and 77 as very rare (Table 3). Two hundred thirty species reported to grow in the area and that belong to the subcanopy, or canopy were not found in any of the 20 one-hectare plots (Table 3). Additionally, only 30 species were considered emergent, and 11 species were exclusive to the coastal or mangrove areas. Secondary forests were dominated by 22 species (Online Resource 1). One hundred ninety-five species had a limited distribution; 40 are endemic to the Osa region, 48 to the country, and 107 are found only in Costa Rica and Nicaragua, or Panama (Table 3).
Six hundred seventy-eight species are recorded in the IUCN database, of which 99 are in a category other than Least Concern (Table 3). Four hundred sixty-five species are reported to be conserved ex situ, including almost half of the rare or scarce species in the area (Table 3). However, only 44 (22.56%) of the species with restricted distribution and 35 (35.35%) of the ones not considered under Least Concern are already conserved ex situ (Table 3). Additionally, more than half of the species (229) were recorded in just 1 or 2 sites in the BGCI, including 24 of the 32 species not in the Least Concern category (Online Resource 1). In contrast, of the 301 species reported to have human use, 219 (72.75%) are preserved ex situ in the BGCI, and 85 (28.24%) are already subject to ex situ conservation, according to the IUCN. Only three species with restricted distribution and five in a category other than Least Concern have data available about desiccation tolerance (Fig. 3a, b). However, more than half of the species in both categories in which the model was able to provide a result were catalogued as possible recalcitrant. Likewise, 183 of 412 (44.42%) scarce species and 13 of 20 oligarchs were classified as recalcitrant or possibly recalcitrant (Fig. 3c). In total, there are 362 species not yet reported as conserved ex situ and with at least one characteristic that makes them a priority for conservation (Online Resource 2): One hundred thirty-one have recalcitrant seeds, 276 are scarce in the area, 151 have a limited distribution and 25 are catalogued as a CR, EN or DD by the IUCN. Of these, 37 are the most vulnerable since they have 3 or 4 characteristics that make them a priority for conservation (Online Resource 2).
Discussion
Our study region is characterized by a high diversity dominated by a few species, the rest being very rare, a common trait of humid tropical forests. Additionally, we found that a high proportion of species are dispersed by animals, dormant, or recalcitrant; the latter especially among oligarch species, endangered species, or species with restricted distributions. Nonetheless, tiny and wind-dispersed seeds are usually orthodox and common among dominant secondary forest species. Additionally, understorey and smaller seeded species tend to be orthodox. We found that almost half of the species are already reported as being conserved ex situ. However, information on seed traits is still lacking for most species and is particularly deficient among species with higher levels of risk of extinction. Our results coincide with previous studies analysing these factors (Howe and Smallwood 1982; Baskin and Baskin 2005; Daws et al. 2005; Sautu et al. 2006, 2007; Wyse and Dickie 2018). However, they have rarely been discussed in the context of tropical humid forest restoration and conservation.
Seed desiccation tolerance
The practical goal of determining desiccation tolerance is establishing an approach for seed collection and ex situ storage. Seed types, in relation to moisture content and longevity, can differ drastically. However, the distinction between orthodox and recalcitrant seeds is an effective and practical way to ensure safe short-term storage conditions. Almost 60% of the species in our study region are orthodox. From them, the first group that is easy to target for seed banks are common species of secondary forests. It has been estimated that millions of currently unavailable seeds will be required to restore degraded areas (Merritt and Dixon 2011). Pioneer species play a fundamental role since they have an “R” strategy with a high production of small seeds and rapid growth, which can accelerate succession. As at our study site, most secondary forest species are expected to display orthodox behavior, as their seedlings tend to grow in open, degraded areas exposed to high levels of sunlight and dryness. Collecting such species in high quantities without affecting natural populations could be feasible. Moreover, information about seed germination requirements is widely available for several of them (e.g. Aud and Ferraz 2012; Pearson et al. 2002). Orthodox species with a wide distribution or abundant densities are also good candidates to promote seed collection efforts. Hyperdominant and oligarchic species play a fundamental role in tropical ecosystems (Fauset et al. 2015; Morera-Beita et al. 2019; Hofhansl et al. 2020); in addition, they are expected to have a broader niche or be better adapted to particular environments (Brousseau et al. 2021). This plasticity could become essential for restoring tropical forests under future unpredictable scenarios. Although information about seed traits for many of them is limited, obtaining seed material for testing and meeting adequate genetic variability should not be a significant challenge. It is necessary to increase the representation of scarce species of tropical forests in seed banks (O’Donnell and Sharrock 2017; Wyse and Dickie 2018). In our study area, one-third of the species catalogued as a priority for conservation are orthodox. As such, seed collections targeting those species could be prioritized. The third group of ecological characteristics target additional highly probable orthodox species in mature forests: tiny, wind-dispersed, and, to a lesser extent, small-seeded understorey species. Many of these would be easy to collect and store due to their small height and seed size. For example, some highly diverse, widespread, and well-known keystone taxa, such as Ficus or Melastomataceae show abundant small-seeded production with an orthodox behaviour.
In contrast, certain families are less likely to be in seed banks due to their low abundance or recalcitrant behaviour (Dickie and Pritchard 2002). This might be the case for more than one-third of the scarce species in the area belonging to abundant and diverse genera, such as Pouteria or Inga. For such species, considering both seed and conservation traits could provide a practical way to develop efficient strategies for their preservation. It is widely known that many tropical species are recalcitrant, while several potential bankable species are scarce, making successful seed collection challenging. For our study region, 710 arboreal species have traits that make them a priority for conservation. However, the list decreased to 363 when considering only those not reported to be conserved ex situ. Among them, only 131 are expected to be recalcitrant. Complementing in situ and ex situ actions can be fundamental to increasing the conservation of such a group of species (Volis 2015; Potter et al. 2017; Werden et al. 2020b). The propagation in restoration projects, the use of vegetative and micropropagation techniques, and the preservation and coordination from different botanical gardens could provide efficient tools to preserve most of these taxa (Werden et al. 2020b, a). For example, the very rare and endangered Osa pulchra, (Fig. 1c) restricted to a small region from Costa Rica and Panama, is already reported to be cultivated ex situ at 22 different sites. This case is promoted by the high ornamental potential of the species. Despite its orthodox behaviour, it still shows how focusing on targeted taxa could easily guarantee the ex situ preservation of most endangered species. The fact that most species with human uses have information about seed traits and are already conserved ex situ supports this idea. As efforts to protect the most vulnerable species increase, it will become easier to determine which species and for what reasons are more difficult to conserve [e.g., low seed production, recalcitrant behaviour or dormancy (Pence et al. 2022a, b)]. Developing specific programs for those exceptional species would be essential to prevent extinctions in the short term (Ma et al. 2013; Jestrow et al. 2018). Recently, there has been an increase in the development of techniques for the long-term storage of plant material from recalcitrant species (Pence et al. 2020). However, there are still several limitations to their use in tropical species (Normah et al. 2019). Such techniques could be prioritized for exceptional, highly endangered, extremely rare, recalcitrant and dormant species that are difficult to propagate with conventional methods (Ashmore et al. 2011; Sarasan et al. 2006; Pence et al. 2022a, b). Finally, it should be considered that several of our data come from model predictions and, as such, should be treated carefully. Although experimental testing is encouraged, this approach has already been shown to be highly reliable based on tropical tree species (Mattana et al. 2020). To date, this is the only option due to the lack of data for most species.
Dormancy
According to our data, dormancy and extended germination are common among arboreal species in the area. The general implications of dormancy for restoration have been recently discussed by Kildisheva et al. (2020) and in the context of tropical species by Sánchez et al. (2019). Here, we highlight three overlook aspects for practitioners. First, since almost one-third of nondormant species have seeds with extended germination, it is advisable to perform pregermination treatments that can make the germination process uniform. The selection of vigorous seeds in the nursery could turn into unwanted genetic selection, as already recorded in botanical garden collections (Ensslin et al. 2018). Our dataset reported simple methods, such as soaking seeds in water to reduce the MLG and synchronize germination in several nondormant species. Second, the fact that we found germination information for only one-third of the total species and pregermination treatments for only one-fifth of the dormant species emphasizes the vital need for research and protocols for most species. Indeed, many opportunities are available to run simple tests since more than half of the species are already reported as conserved ex situ, which means many of these plants have already been propagated. Simple treatments could improve germination up to 100% for many dormant species (Jayasuriya et al. 2013). For rarer or more difficult-to-germinate species, focusing on dormancy type would require few seeds and could quickly provide clues on improving germination (Kildisheva et al. 2020).
Additionally, dormancy is considered a highly conserved trait (Baskin and Baskin 2014). Finding treatments for related species from similar environments could provide a straightforward approach for improving germination. For example, two of the most diverse and abundant families in the tropics, Fabaceae and Arecaceae, are known to have primarily physical (Jayasuriya et al. 2013) and morphophysiological dormancy (Baskin and Baskin 2014). Treatments to improve germination for several species in both families are widely available. Finally, it should be noted that most of the studied species show high germination percentages. Therefore, obtaining seeds is probably the most significant barrier to the propagation of most rare species.
Conclusions
In this work, we showed how common seed traits could be easily used to develop ex situ plant conservation strategies in highly diverse tropical humid forests. For our study region, we found that several aspects could hinder plant conservation since a high proportion of species are scarce, recalcitrant, or dormant. Moreover, information on seed traits is still lacking for most species, although almost half of them are already reported as being conserved ex situ. In contrast, wind-dispersed, smaller seeds and species from the understorey and secondary forest are usually orthodox. Following this approach, we propose targeting common orthodox species for future long-scale restoration projects while prioritizing seed collections for the most endangered ones. For the recalcitrant species, decisions based on the distribution, abundance and current ex situ conservation status could help to improve conservation strategies and priorities. Our results are highly relevant, since as in our study area, most tropical areas have a high diversity dominated by rare and often highly threatened species. As such, analysis to target the most vulnerable species and develop efficient conservation strategies could be essential to protect plant species and prevent extinction (Ma et al. 2013; Pence et al. 2022a). Finally, we highlight the strong need to link conservation efforts with basic research to fill current gaps in both areas. More analysis and in-field work under this approach from other tropical communities and species would allow us to detect the main aspects hindering plant ex situ conservation of exceptional species and develop specific protocols for them (Pence et al. 2022a).
References
Abeli T, Dalrymple S, Godefroid S et al (2020) Ex situ collections and their potential for the restoration of extinct plants. Conserv Biol 34:303–313. https://doi.org/10.1111/cobi.13391
Aguilar-Fernández R, Cornejo X, Santamaría-Aguilar D, et al (2017) Vascular Plants of the Osa Peninsula, Costa Rica (http:sweetgum.nybg.org/osa/).
Ashmore SE, Hamilton KN, Offord CA (2011) Conservation technologies for safeguarding and restoring threatened flora: case studies from Eastern Australia. Vitr Cell Dev Biol Plant 47:99–109. https://doi.org/10.1007/s11627-010-9320-9
Aud FF, Ferraz IDK (2012) Seed size influence on germination responses to light and temperature of seven pioneer tree species from the central Amazon. An Acad Bras Cienc 84:759–766. https://doi.org/10.1590/S0001-37652012000300018
Barnosky AD, Matzke N, Tomiya S et al (2011) Has the Earth’s sixth mass extinction already arrived? Nature 471:51–57. https://doi.org/10.1038/nature09678
Baskin CC, Baskin JM (2005) Seed dormancy in trees of climax tropical vegetation types. Trop Ecol 46:17–28
Baskin CC, Baskin JM (2014) Seeds. ecology, biogeography, and, evolution of dormancy and germination, Second. Elsevier
BGCI (2021a) State of the world’s trees
BGCI (2021b) PlantSearch online database. https://tools.bgci.org/plant_search.php
Blakesley D, Elliott S, Kuarak C et al (2002) Propagating framework tree species to restore seasonally dry tropical forest: Implications of seasonal seed dispersal and dormancy. For Ecol Manage 164:31–38. https://doi.org/10.1016/S0378-1127(01)00609-0
Brousseau L, Fine PVA, Dreyer E et al (2021) Genomic and phenotypic divergence unveil microgeographic adaptation in the Amazonian hyperdominant tree Eperua falcata Aubl. (Fabaceae). Mol Ecol 30:1136–1154. https://doi.org/10.1111/mec.15595
CBD (2020) Aichi biodiversity targets
Chau MM, Chambers T, Weisenberger L et al (2019) Seed freeze sensitivity and ex situ longevity of 295 species in the native Hawaiian flora. Am J Bot 106:1248–1270. https://doi.org/10.1002/ajb2.1351
Cole RJ, Holl KD, Keene CL, Zahawi RA (2011) Direct seeding of late-successional trees to restore tropical montane forest. For Ecol Manage 261:1590–1597. https://doi.org/10.1016/j.foreco.2010.06.038
Cornejo F, Janovec J (2010) Seeds of Amazonian plants (princeton field guides). Princeton University Press, Princeton
Cornejo X, Mori SA, Aguilar R et al (2012) Phytogeography of the trees of the Osa Peninsula, Costa Rica. Brittonia 64:76–101
Da Costa MLMN, Wyse Jackson P, Fernandes RA, Peixoto AL (2018) Conservation of threatened plant species in botanic garden reserves in Brazil. Oryx 52:108–115. https://doi.org/10.1017/S0030605316000776
Daws MI, Garwood NC, Pritchard HW (2006) Prediction of desiccation sensitivity in seeds of woody species: a probabilistic model based on two seed traits and 104 species. Ann Bot 97:667–674. https://doi.org/10.1093/aob/mcl022
Daws MI, Garwood NC, Pritchard HW (2005) Traits of recalcitrant seeds in a semi-deciduous tropical forest in Panamá: Some ecological implications. Funct Ecol 19:874–885. https://doi.org/10.1111/j.1365-2435.2005.01034.x
De Santana DG, Wielewicki AP, Salomão AN (2012) Validation of quality tests for forest seed species. Seed Sci Res 22:S74–S79. https://doi.org/10.1017/S096025851100033X
Dickie JB, Pritchard HW (2002) Systematic and evolutionary aspects of desiccation tolerance in seeds. In: Black M, Pritchard HW (eds) Desiccation and survival in plants. CABI Publishing, Wallingford, UK, Drying without dying, pp 239–259
Ensslin A, Van de Vyver A, Vanderborght T, Godefroid S (2018) Ex situ cultivation entails high risk of seed dormancy loss on short-lived wild plant species. J Appl Ecol 55:1145–1154. https://doi.org/10.1111/1365-2664.13057
Fauset S, Johnson M, Gloor M et al (2015) Hyperdominance in Amazonian forest carbon cycling. Nat Commun 6:1–2
Galindo-Rodriguez C, Roa-Fuentes LL (2017) Seed desiccation tolerance and dispersal in tropical dry forests in Colombia: implications for ecological restoration. For Ecol Manage 404:289–293. https://doi.org/10.1016/j.foreco.2017.08.042
Garwood NC (1983) Seed germination in a seasonal tropical forest in panama: a community study. Ecol Monogr 53:159–181. https://doi.org/10.2307/1942493
Hofhansl F, Chacón-Madrigal E, Fuchslueger L et al (2020) Climatic and edaphic controls over tropical forest diversity and vegetation carbon storage. Sci Rep 10:1–11. https://doi.org/10.1038/s41598-020-61868-5
Hofhansl F, Chacón-Madrigal E, Silla F et al (2019) Diversity and composition of tropical forest plant communities in the Golfo Dulce region. Acta ZooBot Austria 156:31–46
Howe F, Smallwood J (1982) Ecology of seed dispersal. Annu Rev Ecol Syst 13:201–228. https://doi.org/10.1146/annurev.es.13.110182.001221
Hoyle GL, Steadman KJ, Good RB et al (2015) Seed germination strategies: an evolutionary trajectory independent of vegetative functional traits. Front Plant Sci 6:1–13. https://doi.org/10.3389/fpls.2015.00731
IUCN (2021) The IUCN Red List of Threatened Species. Version 2021-3. https://www.iucnredlist.org.
Jayasuriya KMGG, Wijetunga ASTB, Baskin JM, Baskin CC (2013) Seed dormancy and storage behaviour in tropical Fabaceae: a study of 100 species from Sri Lanka. Seed Sci Res 23:257–269. https://doi.org/10.1017/S0960258513000214
Jestrow B, Peguero B, Jiménez F et al (2018) A conservation framework for the Critically Endangered endemic species of the Caribbean palm Coccothrinax. Oryx 52:452–463. https://doi.org/10.1017/S0030605317000588
Kildisheva OA, Dixon KW, Silveira FAO et al (2020) Dormancy and germination: making every seed count in restoration. Restor Ecol 28:S256–S265. https://doi.org/10.1111/rec.13140
Kildisheva OA, Erickson TE, Merritt DJ, Dixon KW (2016) Setting the scene for dryland recovery: an overview and key findings from a workshop targeting seed-based restoration. Restor Ecol 24:S36–S42. https://doi.org/10.1111/rec.12392
Kuhlmann M, Ribeiro JF (2016) Evolution of seed dispersal in the Cerrado biome: ecological and phylogenetic considerations. Acta Bot Brasil 30:271–282
León-Lobos P, Way M, Aranda PD, Lima-Junior M (2012) The role of ex situ seed banks in the conservation of plant diversity and in ecological restoration in Latin America. Plant Ecol Divers 5:245–258. https://doi.org/10.1080/17550874.2012.713402
Lima MJ, Hong TD, Arruda YMBC et al (2014) Classification of seed storage behaviour of 67 Amazonian tree species. Seed Sci Technol 42:363–392
Liu U, Breman E, Cossu TA, Kenney S (2018) The conservation value of germplasm stored at the Millennium Seed Bank, Royal Botanic Gardens, Kew, UK. Biodivers Conserv 27:1347–1386. https://doi.org/10.1007/s10531-018-1497-y
Lughadha EN, Bachman SP, Leão TCC et al (2020) Extinction risk and threats to plants and fungi. Plants People Planet 2:389–408. https://doi.org/10.1002/ppp3.10146
Luna-Nieves AL, Meave JA, González EJ et al (2019) Guiding seed source selection for the production of tropical dry forest trees: Coulteria platyloba as study model. For Ecol Manage 446:105–114. https://doi.org/10.1016/j.foreco.2019.05.020
Ma Y, Chen G, Edward Grumbine R et al (2013) Conserving plant species with extremely small populations (PSESP) in China. Biodivers Conserv 22:803–809. https://doi.org/10.1007/s10531-013-0434-3
Mattana E, Peguero B, Di Sacco A et al (2020) Assessing seed desiccation responses of native trees in the Caribbean. New for 51:705–721. https://doi.org/10.1007/s11056-019-09753-6
Merritt DJ, Dixon KW (2011) Restoration seed banks. Science 332:21–22
Missouri Botanical Garden (2022) Tropicos. www.tropicos.org
Mondoni A, Probert RJ, Rossi G et al (2011) Seeds of alpine plants are short lived: implications for long-term conservation. Ann Bot 107:171–179. https://doi.org/10.1093/aob/mcq222
Morera-Beita A, Sánchez D, Wanek W et al (2019) Beta diversity and oligarchic dominance in the tropical forests of Southern Costa Rica. Biotropica 51:117–128. https://doi.org/10.1111/btp.12638
Mounce R, Smith P, Brockington S (2017) Ex situ conservation of plant diversity in the world’s botanic gardens. Nat Plants 3:795–802. https://doi.org/10.1038/s41477-017-0019-3
Normah MN, Sulong N, Reed BM (2019) Cryopreservation of shoot tips of recalcitrant and tropical species: advances and strategies. Cryobiology 87:1–14. https://doi.org/10.1016/j.cryobiol.2019.01.008
Núñez-Cruz A, Meave JA, Bonfil C (2018) Reproductive phenology and seed germination in eight tree species from a seasonally dry tropical forest of Morelos, Mexico: implications for community-oriented restoration and conservation. Trop Conserv Sci 11:1–14. https://doi.org/10.1177/1940082917749946
O’Donnell K, Sharrock S (2017) The contribution of botanic gardens to ex situ conservation through seed banking. Plant Divers 39:373–378. https://doi.org/10.1016/j.pld.2017.11.005
Pearson TRH, Burslem DFRP, Mullins CE, Dalling JW (2002) Germination ecology of neotropical pioneers: Interacting effects of environmental conditions and seed size. Ecology 83:2798–2807. https://doi.org/10.1890/0012-9658(2002)083[2798:GEONPI]2.0.CO;2
Pedrini S, Dixon KW (2020) International principles and standards for native seeds in ecological restoration. Restor Ecol 28:S286–S303. https://doi.org/10.1111/rec.13155
Pence VC, Ballesteros D, Walters C et al (2020) Cryobiotechnologies: tools for expanding long-term ex situ conservation to all plant species. Biol Conserv 250:108736. https://doi.org/10.1016/j.biocon.2020.108736
Pence VC, Bruns EB, Meyer A et al (2022a) Gap analysis of exceptional species—using a global list of exceptional plants to expand strategic ex situ conservation action beyond conventional seed banking. Biol Conserv 266:109439. https://doi.org/10.1016/J.BIOCON.2021.109439
Pence VC, Meyer A, Linsky J et al (2022b) Defining exceptional species: a conceptual framework to expand and advance ex situ conservation of plant diversity beyond conventional seed banking. Biol Conserv 266:109440. https://doi.org/10.1016/J.BIOCON.2021.109440
Potter KM, Jetton RM, Bower A et al (2017) Banking on the future: progress, challenges and opportunities for the genetic conservation of forest trees. New for 48:153–180. https://doi.org/10.1007/s11056-017-9582-8
Quesada-Monge R, Fernández-Vega, J. (2005) Actualización de listado de especies arbóreas de uso forestal y otros usos en Costa Rica. Rev. For. Mesoam. Kurú 2:44-87.
RBGK (2021) Seed Information Database (SID).
Ribeiro GVT, Teixido AL, Barbosa NPU, Silveira FAO (2016) Assessing bias and knowledge gaps on seed ecology research: implications for conservation agenda and policy. Ecol Appl 26:2033–2043. https://doi.org/10.1890/15-1852.1/suppinfo
Rivière S, Müller JV (2018) Contribution of seed banks across Europe towards the 2020 global strategy for plant conservation targets, assessed through the ENSCONET database. Oryx 52:464–470. https://doi.org/10.1017/S0030605316001496
Romero-Saritama JM, Pérez Ruiz C (2016) Seed morphological traits and their implication in the ex situ conservation of woody species in Tumbesian dry forests. Ecosistemas 25:59–65. https://doi.org/10.7818/ECOS.2016.25-2.07
Saldaña-Vázquez RA, Castaño JH, Baldwin J, Pérez-Torres J (2019) Does seed ingestion by bats enhance germination? A new meta-analysis 15 years later. Mamm Rev 49:201–209
Sánchez J, Torres Y, Pernús M, Barrios D (2019) Dormancia y germinación en semillas de árboles y arbustos de Cuba : implicaciones para la restauración ecológica. Acta Botánica Cuba 218:1–33
Sánchez JA, Montejo L, Gamboa A et al (2015) Germination and dormancy of shrubs and climbing plants of the evergreen forest of Sierra del Rosario. Cuba Pastures and Forages 38:95–110
Sandor ME (2002) Forest regeneration on the Osa Peninsula. University of Connecticut, Costa Rica
Santamaría-Aguilar D, Aguilar-Fernández R, van der Werff H (2021) Beilschmiedia osacola (Lauraceae) a new species from the Osa Peninsula, Costa Rica. Phytotaxa 498:197–204. https://doi.org/10.11646/phytotaxa.498.3.5
Sarasan V, Cripps R, Ramsay MM et al (2006) Conservation in vitro of threatened plants—progress in the past decade. Vitr Cell Dev Biol - Plant 423(42):206–214. https://doi.org/10.1079/IVP2006769
Sautu A, Baskin JM, Baskin CC et al (2007) Classification and ecological relationships of seed dormancy in a seasonal moist tropical forest, Panama, Central America. Seed Sci Res 17:127–140. https://doi.org/10.1017/S0960258507708127
Sautu A, Baskin JM, Baskin CC, Condit R (2006) Studies on the seed biology of 100 native species of trees in a seasonal moist tropical forest, Panama, Central America. For Ecol Manage 234:245–263. https://doi.org/10.1016/j.foreco.2006.07.006
Sharrock S (2012) Global strategy for plant conservation: a guide
Solberg SØ, Yndgaard F, Andreasen C et al (2020) Long-Term storage and longevity of orthodox seeds: a systematic review. Front Plant Sci 11:1007. https://doi.org/10.3389/fpls.2020.01007
Soltani E, Baskin CC, Baskin JM et al (2018) A meta-analysis of the effects of frugivory (endozoochory) on seed germination: role of seed size and kind of dormancy. Plant Ecol 219:1283–1294. https://doi.org/10.1007/s11258-018-0878-3
Sommerville KD, Errington G, Newby Z-J et al (2021) Assessing the storage potential of Australian rainforest seeds: a decision-making key to aid rapid conservation. Biodivers Conserv 30:3185–3218. https://doi.org/10.1007/s10531-021-02244-1
Tweddle JC, Dickie JB, Baskin CC, Baskin JM (2003) Ecological aspects of seed desiccation sensitivity. J Ecol 91:294–304. https://doi.org/10.1046/j.1365-2745.2003.00760.x
UICN (2020) The Bonn challenge. https://www.bonnchallenge.org/
Valverde-Rodríguez K, Morales CO, García EG (2019) Seed germination of Crescentia alata (Bignoniaceae) in different conditions of temperature, luminosity and storage. Rev Biol Trop 67:S120–S131. https://doi.org/10.15517/rbt.v67i2supl.37211
Volis S (2015) Species-targeted plant conservation: time for conceptual integration. Isr J Plant Sci 63:232–249. https://doi.org/10.1080/07929978.2015.1085203
Wade EM, Nadarajan J, Yang X et al (2016) Plant species with extremely small populations (PSESP) in China: a seed and spore biology perspective. Plant Divers 38:209–220. https://doi.org/10.1016/j.pld.2016.09.002
Wang JH, Xu GX, Chen W et al (2021) Impacts of growth form and phylogenetic relatedness on seed germination: a large-scale analysis of a subtropical regional flora. Ecol Evol 11:1280–1293. https://doi.org/10.1002/ece3.7132
Weber A, Huber W, Weissenhofer A et al (2001) An introductory field guide to the flowering plants of the Golfo Dulce rain forests, Costa Rica. Stapfia 78:1–569
Wehncke EV, Dalling JW (2005) Post-dispersal seed removal and germination selected tree species dispersed by Cebus capucinus on Barro Colorado Island, Panama. Biotropica 37:73–80. https://doi.org/10.1111/j.1744-7429.2005.03037.x
Weissenhofer A, Huber W, Mayer M et al (2008) Natural and cultural history of the Golfo Dulce Region, Costa Rica. Stapfia 88:1–768. https://doi.org/10.1017/S0960428611000199
Weissenhofer A, Picado-Zuñiga A, Ramírez WB et al (2019) Forest conservation and restoration in southwestern Costa Rica: the biological corridors COBIGA and AMISTOSA. Acta ZooBot Aust 156:47–60
Werden LK, Holl KD, Rosales JA et al (2020a) Effects of dispersal- and niche-based factors on tree recruitment in tropical wet forest restoration. Ecol Appl 30:1–16. https://doi.org/10.1002/eap.2139
Werden LK, Sugii NC, Weisenberger L et al (2020b) Ex situ conservation of threatened plant species in island biodiversity hotspots: A case study from Hawaii. Biol Conserv. https://doi.org/10.1016/j.biocon.2020.108435
WFO (2022) World Flora Online; Published on the Internet. http://www.worldfloraonline.org
Wyse SV, Dickie JB (2017) Predicting the global incidence of seed desiccation sensitivity. J Ecol 105:1082–1093. https://doi.org/10.1111/1365-2745.12725
Wyse SV, Dickie JB (2018) Taxonomic affinity, habitat and seed mass strongly predict seed desiccation response: a boosted regression trees analysis based on 17 539 species. Ann Bot 121:71–83. https://doi.org/10.1093/aob/mcx128
Wyse SV, Dickie JB, Willis KJ (2018) Seed banking not an option for many threatened plants. Nat Plants 4:848–850. https://doi.org/10.1038/s41477-018-0298-3
Aknowledgments
We want to dedicate this paper to the memory of Marvin López who passed away during the reviewing of this manuscript. His departure is a great loss for the conservation of the forests of the Osa Peninsula. We thank all the staff from the Department of Developmental Biology, at the Institute of Dendrology, Kórnik for their assistance during the preparation of this manuscript.
Funding
This work was supported by the Institute of Dendrology, Polish Academy of Sciences.
Author information
Authors and Affiliations
Contributions
JMLL: Conceptualization, Data Curation; Formal Analysis; Writing—Original Draft Preparation Writing–Review & Editing. MKW: Writing—Review & Editing; Visualization; Supervision. ECh-M: Writing—Review & Editing. PC: Writing—Review & Editing; Funding Acquisition. All authors gave final approval for publication.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no conflict of interest or competing interests.
Additional information
Communicated by Pedro Eisenlohr.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ley-López, J.M., Wawrzyniak, M.K., Chacón-Madrigal, E. et al. Seed traits and tropical arboreal species conservation: a case study of a highly diverse tropical humid forest region in Southern Costa Rica. Biodivers Conserv 32, 1573–1590 (2023). https://doi.org/10.1007/s10531-023-02565-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02565-3