Abstract
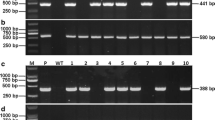
Rice stripe disease (RSD), caused by rice stripe virus (RSV), is a serious disease in temperate rice-growing areas. We have created an RNAi construct containing coat protein gene (CP) and disease specific protein gene (SP) sequences from RSV. The RNAi construct was transformed into two susceptible japonica varieties, Suyunuo and Guanglingxiangjing, to develop resistance against RSD. The homozygous progeny of rice plants in the T5 and T7 generations containing RNAi constructs, after self-fertilization were strongly resistant to viral infection. RT-PCR indicated that viral replication of SP and CP in the transgenic plants was significantly inhibited. There were no obvious morphological or developmental differences between the transgenic and wild-type plants from seedling stage to maturity. The excellent agronomic traits of these two varieties, such as high yield and good quality were maintained. Suppression of virus genes using RNAi is therefore a practical and effective strategy for controlling viral infection in crops.




Similar content being viewed by others
References
Abhary MK, Anfoka GH, Nakhla MK, Maxwell DP (2006) Post-transcriptional gene silencing in controlling viruses of the tomato yellow leaf curl virus complex. Arch Virol 151:2349–2363
Chuang CF, Meyerowitz EM (2000) Specific and heritable genetic interference by double-stranded RNA in Arabidopsis thaliana. Proc Natl Acad Sci USA 97:4985–4990
Ding XL, Jiang L, Liu SJ, Wan JM (2004) QTL analysis for rice stripe disease resistance gene using recombinant inbred lines derived from crossing of Kinmaze and Dv85. Acta Genet Sin 31:287–292
Fire A (1999) RNA-triggered gene silencing. Trends Genet 15:358–363
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Hayakawa T, Zhu YF, Itoh K, Kimura Y, Izawa T, Shmamoto K, Toriyama S (1992) Genetically engineered rice resistant to rice stripe virus, an insect-transmitted virus. Proc Natl Acad Sci USA 89:9865–9869
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium tumefaciens and sequence analysis of the boundaries of the T-DNA. Plant J 6:271–282
Kakutani T, Hayano Y, Hayashit T, Minobe Y (1990) Ambisense segment 4 of rice stripe virus: possible evolutionary relationship with phleboviruses and uukuviruses (Bunyaviridae). J Gen Virol 71:1427–1432
Kakutani T, Hayano Y, Hayashit T, Minobe Y (1991) Ambisense segment 3 of rice stripe virus: the first instance of a virus containing two ambisense segments. J Gen Virol 72:465–468
Lennefors BL, van Roggen PM, Yndgaard F, Savenkov EI, Valkonen JP (2008) Efficient dsRNA-mediated transgenic resistance to beet necrotic yellow vein virus in sugar beets is not affected by other soilborne and aphid-transmitted viruses. Trans Res 17:219–228
Lin Q, Lin H, Wu Z, Lin Q, Xie L (1998) Accumulations of coat protein and disease-specific protein of rice stripe virus in its host. J Fujian Agric Univ 27:257–260
Ma C, Ma BG, He JA, Hao QN, Lu XY, Wang L (2011a) Regulation of carotenoid content in tomato by silencing of lycopene beta/epsilon-cyclase genes. Plant Mol Biol Rep 29:117–124
Ma J, Song YZ, Wu B, Jiang MS, Li KD, Zhu CX, Wen FJ (2011b) Production of transgenic rice new germplasm with strong resistance against two isolations of rice stripe virus by RNA interference. Trans Res. doi:10.1007/s11248-011-9502-1
Mansoor S, Amin I, Hussain M, Zafar Y, Briddon RW (2006) Engineering novel traits in plants through RNA interference. Trends Plant Sci 11:559–565
Qi Y, Hannon GJ (2005) Uncovering RNAi mechanisms in plants: biochemistry enters the foray. FEBS Lett 579:5899–5903
Qin WQ, Gao DM, Chen SX (1994) Studies on techniques of rapid detecting rice stripe virus in Laodelphax striatellus. Acta Agric Zhejiangensis 6:226–229
Sharp PA, Zamore PD (2000) Molecular biology. RNA interference. Science 287:2431–2433
Shimizu T, Yoshii M, Wei T, Hirochika H, Omura T (2009) Silencing by RNAi of the gene for Pns12, a viroplasm matrix protein of rice dwarf virus, results in strong resistance of transgenic rice plants to the virus. Plant Biotechnol J 7:24–32
Shimizu T, Nakazono-Nagaoka E, Uehara-Ichiki T, Sasaya T, Omura T (2010) Targeting specific genes for RNA interference is crucial to the development of strong resistance to rice stripe virus. Plant Biotechnol J 9:503–512
Sunilkumar G, Campbell LM, Puckhaber L, Stipanovic RD, Rathore KS (2006) Engineering cottonseed for use in human nutrition by tissue-specific reduction of toxic gossypol. Proc Natl Acad Sci USA 103:18054–18059
Takahashi M, Toriyama S, Hamamatsu C, Ishihama A (1993) Nucleotide sequence and possible ambisense coding strategy of rice stripe virus RNA segment 2. J Gen Virol 74:769–773
Toriyama S, Takahashi M, Sano Y, Shimizu T, Ishihama A (1994) Nucleotide sequence of RNA 1, the largest genomic segment of rice stripe virus, the prototype of the tenuiviruses. J of Gen Virol 75:3569–3579
Tyagi H, Rajasubramaniam S, Rajam MV, Dasgupta I (2008) RNA-interference in rice against rice tungro bacilliform virus results in its decreased accumulation in inoculated rice plants. Trans Res 17:897–904
Washio O, Toriyama K, Ezuka A, Sakurai Y (1968) Studies on the breeding of rice varieties resistant to stripe disease II. Genetic study on resistance to stripe disease in Japanese upland rice. Jpn J Breed 18:96–101
Waterhouse PM, Graham MW, Wang MB (1998) Virus resistance and gene silencing in plants can be induced by simultaneous expression of sense and antisense RNA. Proc Natl Acad Sci USA 95:13959–13964
Wu SJ, Zhong H, Zhou Y, Zuo H, Zhou LH, Zhu JY, Ji CQ, Gu SL, Gu MH, Liang GH (2009) Identification of QTLs for the resistance to rice stripe virus in the indica rice variety Dular. Euphytica 165:557–565
Wu X, Zuo S, Chen Z, Zhang Y, Zhu J, Ma N, Tang J, Chu C, Pan X (2011) Fine mapping of qSTV11TQ, a major gene conferring resistance to rice stripe disease. Theor Appl Genet 122:915–923
Yang W, Wang X, Wang S, Yie Y, Tien P (1996) Infection and replication of a planthopper transmitted virus-rice stripe virus in rice protoplasts. J Virol Methods 59:57–60
Yin DM, Deng SZ, Zhan KH, Cui DQ (2007) High-oleic peanut oils produced by HpRNA-mediated gene silencing of oleate desaturase. Plant Mol Biol Rep 25:154–163
Zhang HG, Li B, Zhu ZB, Cui XF, Tang SZ, Liang GH, Gu MH (2009) Improving resistance of a good quality japonica variety Wuyujing 3 to rice stripe virus via molecular marker-assisted selection. Chin J Rice Sci 23:263–270
Zhang HG, Xu ZP, Li P, Li B, Liu C, Zhu BH, Xu JF, Niu ZY, Tang SZ, Liang GH, Gu MH (2011a) Improving the resistance of Wuyunjing 8 to rice stripe virus via molecular marker-assisted selection. Acta Agron Sin 37:745–754
Zhang YX, Wang Q, Jiang L, Liu LL, Wang BX, Shen YY, Cheng XN, Wan JM (2011b) Fine mapping of qSTV11(KAS), a major QTL for rice stripe disease resistance. Theor Appl Genet 122:1591–1604
Zhu Y, Hayakawa T, Toriyama S (1992) Complete nucleotide sequence of RNA 4 of rice stripe virus isolate T, and comparison with another isolate and with maize stripe virus. J Gen Virol 73:1309–1312
Acknowledgments
This study was financially supported by grants from the Ministry of Agriculture of China for Transgenic Research (2009ZX08001-019B, 2008ZX08009-003), the State Key Program of Basic Research of P. R. China (No. 2011CB100206), and the Key Program of the Bureau of Education of Jiangsu Province, P. R. China (10KJA210059).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, Y., Yuan, Y., Yuan, F. et al. RNAi-directed down-regulation of RSV results in increased resistance in rice (Oryza sativa L.). Biotechnol Lett 34, 965–972 (2012). https://doi.org/10.1007/s10529-012-0848-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-0848-0