Abstract
Information on the allergenic pollen season provides insight on the state of the environment of a region and facilitates allergy symptom management. We present a retrospective analysis of the duration and severity of the allergenic pollen season and the role of meteorological factors in Istanbul, Turkey. Aerobiological sampling from January 2013 to June 2016, pollen identification and counting followed current standard methodology. Pollen seasons were defined according to 95% of the Annual Pollen Integral (APIn) and the season start date was compared with the first day of 5 day consecutive non-zero records. Generalized additive models (GAMs) were created to study the effect of meteorological factors on flowering. The main pollen contributors were taxa of temperate and Mediterranean climates, and neophytic Ambrosia. Cupressaceae, Poaceae, Pinaceae, Quercus and Ambrosia had the greatest relative abundance. The pollen season defined on 95% of the APIn was adequate for our location with total APIns around 10.000 pollen*day*m−3. Woody taxa had generally shorter seasons than herbaceous taxa. In trees, we see precipitation as the main limiting factor for assimilate production prior to anthesis. A severe tree pollen season in 2016 suggests intense synchronous flowering across taxa and populations triggered by favourable water supply in the preceding year. GAM models can explain the effect of weather on pollen concentrations during anthesis. Under the climatic conditions over the study period, temperature had a negative effect on spring flowering trees, and a positive one on summer flowering weeds. Humidity, atmospheric pressure and precipitation had a negative effect on weeds. Our findings contribute to environmental and allergological knowledge in southern Europe and Turkey with relevancy in the assessment of impacts of climate change and the management of allergic disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Information on the allergenic pollen season and its severity provides insight on the state of the environment of a region and facilitates allergy symptom management (Karatzas et al., 2019; Gehrig et al., 2018). Both aspects are closely connected in aerobiological research. Climate change gives rise to increased respiratory allergy, due to its impact on the start and duration of the pollen season, the flowering intensity, and the presence of allergen content (D’Amato et al., 2020; Eguiluz‐Gracia et al., 2020; Damialis et al., 2019; Katelaris & Beggs, 2018; Jenerowicz et al., 2012). The megacity Istanbul with a population of close to 18 million has now an allergy and asthma incidence of 20% in adolescents and adults (Turkish Statistical Institute, 2020). Furthermore, it supports health practitioners in the choice of allergen extracts to test (Zemmer et al., 2021; Ansotegui et al., 2020; Heinzerling et al., 2013), in the timing for implementation of clinical trials (Bastl et al., 2019; Pfaar et al., 2017) and in the sound interpretation of clinical results (Werchan et al., 2018). Pollen types detected in aerobiological sampling differ in their clinical relevance between regions (Karatzas et al., 2019; D’Amato et al., 2007). Within a region, relevance may also change over time. In Milan, for example, Cupressaceae only recently have become important with regard to allergy (Asero et al., 2020). Regular updates on local pollen information are, thus, essential (Ansotegui et al., 2020). In the interpretation of pollen information cross-reactivity patterns of taxa have to be considered (Ansotegui et al., 2020). Some of the major proteins causing allergy are panallergens, common to many flowering plants (McKenna et al., 2016). The major birch allergen Bet v 1, for example, belonging to the protein family PR10, has homologues in hazel (Cor a 1) and alder (Aln g 1) within the families Betulaceae and Fagaceae as in beech (Fag s 1) in the order Fagales (Matricardi et al., 2016; Weber, 2008) and also in non-related taxa, such as apple (Mal d 1) (McKenna et al., 2016). Therefore, when presenting pollen information, you can address clinical needs by grouping taxa above species, genus, and even family level (Hoffmann et al., 2020).
In environmental terms, the local pollen spectrum depends on the natural vegetation, land use, ornamental flora in green urban spaces, and the ruderal urban flora (Werchan et al., 2018; Monroy-Colín et al., 2018; McInnes et al., 2017; Tosunoglu et al., 2015; Carinanos et al., 2014; Fernández-Rodríguez et al., 2014; Çeter et al., 2012), as well as on prevailing air currents (Alan et al., 2019; Rojo et al., 2015; Damialis et al., 2005). It can reveal the arrival of invasive plants, such as ragweed, when the presence of local sources is not yet known (Zemmer et al., 2012). Meteorological factors influence the flowering phenology, and thus, the duration and intensity of the pollen flow in anemophilous species (Dahl et al., 2013). So can accumulated temperature during the period when male catkins are initiated and formed, affect the amount of pollen released during flowering (Bogdziewicz et al., 2017; Dahl & Strandhede, 1996). Air humidity impacts pollen release. Dry air favours anther dehiscence in many anemophilous species (Dahl et al., 2013; Martin et al., 2010; Pacini, 2000; Bianchi et al., 1959). When air is humid, pollen grains absorb water and get heavier; some types like grass pollen will even burst as they cannot adjust to humidity (Dahl et al., 2013; Pacini, 2000). In dryer climates, water availability is a limiting factor affecting plant biomass accumulation (Szymczak et al., 2020) and flowering intensity (Velasco-Jiménez et al., 2020; Galán et al., 2016; García-Mozo et al., 2010; Peñuelas et al., 2004). The Mediterranean region, including southern Europe, will face increased arid conditions due to climate change (Hoegh-Guldberg et al., 2018). In Istanbul this trend has become evident in the shift from a moist-subhumid towards dry subhumid climate during the last twenty years (Turoğlu, 2014). The result may be spatial and temporal shifts of allergenic pollen sources (Damialis et al., 2019). Reports on phenology and flowering intensity of wind pollinated taxa of a region provide, therefore, important information on environmental changes over time (Velasco-Jiménez et al., 2020; Galán et al., 2016).
In this paper, we analyse atmospheric pollen monitored in Istanbul from the environmental perspective. The aims are to (1) define pollen seasons (2) report on the intensity of pollen concentrations (3) and on the effect of meteorological factors on the main taxa.
2 Materials and methods
2.1 Study area
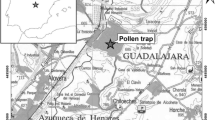
The monitoring was performed in Büyükçekmece at the western fringes of Greater Istanbul. The climate of Istanbul corresponds to the Köppen Geiger Csa-type (Turoğlu, 2014) that is somewhat between temperate and Mediterranean (Climate-Data.org, 1999). The Bosporus and the south of the city are influenced by the Marmara Sea, while the climate in the northern and western hinterland is more continental, as it is influenced by the Black Sea and the Balkans. The study area is, thus, characterized by a transitional climate from cool and wet conditions northwards into the Istranca mountain range, to a dry and warm situation in the south and around Bosporus (Baser, 2011; Fig. 1). The topography is hilly and the highest elevation is about 300 m above sea level. North-easterly winds prevail. The vegetation changes according to the climatic north–south gradient. In plant geographical terms, two floristic regions blend here into one another: the South-Eurosiberian and the Mediterranean (Schroeder, 1998). Most of the wood cover is situated upwind the trap in the Belgrad Forest, and features the following wind-pollinated deciduous thermophilus trees: Quercus frainetto, Q. petrea, Q. robur, Q. cerris, Fagus orientalis, Castanea sativa, Corylus avellana, Carpinus betulus, C. orientalis, Alnus glutinosa, Fraxinus angustifolia, F. ornus, Populus tremula, Erica arbora, E. manipuliflora, Ulmus minor, Salix caprea and S. cinerea (Çoban et al., 2016; Baser, 2011; Kavgaci et al., 2010). There are also areas of Pinus nigra plantations. Amidst patches of pseudomaquis with Quercus coccifera, Juniperus communis, Paliurus spina-christi and Pistacia terebinthus, grasslands turning dry in summer, and agricultural lands, villages emerge. The agricultural land is used for the growth of annual crops (sunflower, canola, cereals). The area has four watersheds, some of them functioning as fresh water reserves of the city with restrictions on urban development. Anemophilous urban flora on walls, wastelands and coasts feature herbaceous plants pertaining to Poaceae (i.e. Digitaria sanguinalis, Setaria ssp., Echinochloa ssp., Sorghum halepense), Asteraceae (i.e. Artemisia vulgaris, A. annua, A. absinthium, Xanthium spinosum, X. strumarium), Urticaceae (i.e. Urtica dioica, U. membranacea, U. pilulifera, U. urens, Parietaria judaica), Amaranthaceae (i.e. Amaranthus ssp., Chenopodium ssp., Atriplex ssp., Salicornia ssp.), Polygonaceae (Rumex ssp.), Plantaginaceae (i.e. Plantago lanceolata, P. major, P. coronopus, P. afra) (Bakis et al., 2020; Altay et al., 2010). Common anemophilous ornamental trees are Cupressus sempervirens, Cedrus libani, Plantanus x acerifolia, P. orientalis, Thuja orientalis, Morus ssp., Salix babylonica, Fraxinus angustifolia, Ulmus ssp., Celtis australis, Ailanthus altissima, Acer negundo, Pinus ssp. (Baser, 2011) and Olea europaea.
Study area (left) and climate diagram of Istanbul (right). The striped areas in the climate diagram denote humid conditions, and the dotted areas show when the conditions are arid (constructed according to Walter & Lieth, 1960)
2.2 Pollen monitoring
Pollen data include a 3½ year period from 28th January 2013 to 26th June 2016 sampled according to the current standard methodology with a 7 day volumetric Hirst-type pollen sampler (Hirst, 1952). Pollen monitoring, identification and counting followed the minimum requirements of the European Society of Aerobiology (EAS) (Galán et al., 2014) and are normed by EN 16868 (2019). Daily mean pollen concentrations are expressed in pollen (p)*m−3 air.
On 21st April 2014 the position of the trap had to be moved from three to 10 m height (ca. 200 m ENE from the original position) due to a construction project. We did not alter the master data set for the period prior to the change, based on findings that the height of the trap does not influence the time series significantly (Rojo et al., 2019a; Fernández-Rodríguez et al., 2014; Galán et al., 1995). We verified this assumption following the theory that most pollen is shed in the vicinity of the trap (Adams-Groom et al., 2017). To do so, we studied the distribution of pollen concentrations of Poaceae and Plantago, growing in the immediate surrounding of the trap, and of Olea and Pinaceae growing at about 40 m distance during their peak flowering period (pre-peak in case of Plantago); all of them flowering during May (Fig. 2).
Pollen concentrations of taxa growing in the surrounding of the trap during May in a four-year comparison. The boxes include 50% of the data, the interquartile range (IQR) between the first quartile (Q1), and the third quartile (Q3); the line in the box is the median, the whiskers show the data range (Q1 − 1.5 * IQR, Q3 + 1.5 * IQR); dots are outliers and indicate peak concentrations
2.3 Meteorological data
Meteorological data on temperature, humidity and pressure were obtained from the open source www.weatherunderground.com, recorded at 41.14° N, 28.46° E Istanbul Hezarfen Airfield Station located 6 km northwest of the trap location. Precipitation data were purchased from the State Meteorological Service (www.mgm.gov.tr). We tested the effect of maximum (Tmax) and minimum daily temperature (Tmin), mean daily humidity (HumMean), mean daily pressure (PressMean) and precipitation (Prec) on the main pollen taxa. Precipitation of the year 2012 was included to study the effect of this factor on trees a year ahead of anthesis when flowers are initiated (Dahl and Strandhede, 1996).
2.4 Data analysis
The dataset was homogenised to obtain equally 31 taxa for all years by removing pollen taxa with negligible Annual Pollen Integral (APIn), for example, Arecaceae, Mercurialis, Typha, Iva. Single pollen out of season attributed to resuspension or flowering out of season (for example, Ulmus) were removed. Next, a quality check was performed to identify missing data. In 2013 and 2015 sampling was discontinued during January for technical reasons. We interpolated the missing data with the moving mean function because it adapted better to our data than linear regression did in preliminary tests. We confirm that for aerobiological data, the moving mean interpolation performs better as compared to other methods as suggested by Picornell et al. (2021).
We present the weekly pollen concentration average of our studied years, not considering this presentation a pollen calendar for which at least five years of data are required (Galán et al., 2017). For this figure, we chose a scale of 10 colour gradients from light to dark: light colour standing for low and dark for higher pollen concentrations.
We used descriptive statistics for the analysis of phenology, represented by the timing of the pollen season, and the flowering intensity, represented by the Seasonal Pollen Integral (SPIn) and APIn. For the retrospective analysis of our aerobiological data the percent method was deemed appropriate (Bastl et al., 2018). The pollen seasons were, thus, defined according to the 95% method (Andersen, 1991): the season starts when 2.5% of the APIn is reached and ends on the day with 97.5% of the APIn. The result thereof can be used to calculate the SPIn. To assess the validity of this method for the season start date, the 1st day of 5 non-zero concentrations was determined. Aerobiological data were mainly analysed with the AeRobiology package (Rojo et al., 2019b) in RStudio version 3.6.3.
To analyse the effect of meteorological factors on pollen seasons, we run generalized additive models (GAMs) based on the assumption of a nonlinear relationship between meteorological factors and pollen concentrations. These models estimate the effect on the change of pollen concentrations with each added unit of independent variable (Ravindra et al., 2019). The model can automatically fit nonlinear dependencies and is adequate for short term and long-term time series (Ravindra et al., 2019). For the model we used the quasi-Poisson function and a spline function on time. GAMs were created with the nlme package (Pinheiro et al., 2021) in R.
3 Results
3.1 Trap position change
Herbaceous Poaceae and Plantago displayed pronouncedly higher pollen concentrations in May 2013 compared to the other years and peak concentrations were over three times higher. In woody Olea and Pinaceae, the bulk of the data was similar over the studied years. Peak concentrations, however, were twice higher in 2013 than in the other years. Effects on pollen intensities of other taxa were negligible (compare SPIn and peak values in Tables 1, 2). When reading diagrams this bias should be considered.
3.2 Flowering phenology
We calculated numerical details on seasonal parameters of 31 woody and herbaceous taxa (Tables 1, 2).
The start (2.5%) and end date (97.5%) of the APIn of the pollen seasons reflecting the flowering phenophase of anemophilous plants varied between years (Fig. 3, Tables 1, 2). The longest seasons were generally observed in herbaceous plants, for example, Urticaceae (> 60 days difference between years), Amaranthaceae and Poaceae, but also in Carpinus (about 50 days difference between years). Variations for the end date of the seasons was pronounced in herbaceous Plantago, Amaranthaceae and Urticaceae, and for some trees, especially in Pinaceae (over 100 days difference between years), Carpinus, and to a lesser extent in Fraxinus.
We compared the 95% method to define the season start with the date of the 1st day of 5 non− zero concentrations for the main pollen contributing taxa to assess the feasibility of this method at our location. Cupressaceae, Fraxinus, Pinaceae, Platanus, Poaceae, and Plantago had a standard deviation < 5 in the dates of the season start during the study period. The differences in the season start dates were low also for Olea (except in 2016) and Betula (except in 2013).
3.3 Pollen intensity
The relative abundance of the 15 main pollen types is reported in Fig. 4. The summarization of Betulaceae and Fagaceae in the analysis is made to follow the rationale of cross-reactions in the Fagales order (Matricardi et al., 2016). Alnus and Carpinus were the main pollen contributors within Betulaceae and Quercus in the Fagaceae.
The details on the phenology and relative abundancies of all taxa pertaining to the Fagales including Corylus, Fagus and Castanea sativa, all three with < 0.5% of the APIn, are presented as violin plots in Fig. 5. Violin plots can show the entire distribution of the data emphasising peak periods. Wider parts in the plot contain observations with higher density than in thinner parts (Hintze & Nelson, 1998). Corylus, Quercus and Castanea had a shorter pre-peak period than Alnus, which can be inferred from the positive skewness of the distribution. Alnus displayed a short and intense flowering period. Fagus and Betula showed a more uniform distribution without a pronounced peak.
Variation in the interannual flowering intensity was pronounced for most species. We observed remarkable differences in the SPIn of single years (Tables 1, 2 and Fig. 4). In 2013, the SPIn of Poaceae was about four times (2426 pollen*day*m−3) and of Plantago (666 pollen*day*m−3), two times higher than the mean concentrations of the other three years observed. Olea had the highest SPIn and peak value in 2013. Comparably higher SPIns and peak concentrations were observed also in Salix both in 2013 and 2014. In 2014 the SPIn of Corylus (67 pollen*day*m−3) and Alnus (223 pollen*day*m−3) was over two times higher than the mean. The SPIn of Carpinus was five times higher in 2015 (234 pollen*day*m−3) and nine times higher in 2016 (429 pollen*day*m−3) than the mean SPIn of the two preceding years 2013 and 2014 (48 pollen*day*m−3). All in all, seven woody taxa (Pinaceae, Platanus, Fraxinus, Cupressaceae, Betula, Quercus and Castanea) out of 20 showed an obvious increase in the SPIn in 2016. The SPIn of Ambrosia increased steadily from 476 pollen*day*m−3 in 2013, to 539 pollen*day*m−3 in 2014 and 600 pollen*day*m−3 in 2015.
Weekly mean pollen concentrations of the 15 main pollen contributing taxa recorded in Büyükcekmece are shown in Fig. 6. The highest pollen concentrations were found in the months April and May. Table 3 provides an overview of taxa with concentrations > = 100 pollen*m−3. Over the entire study period the highest pollen loads were recorded in April 2016. The APIn in 2013 was 9591 pollen*day*m−3; in 2014: 6695 pollen*day*m−3; in 2015: 7458 pollen*day*m−3. Interestingly, till the 26th June 2016 already more pollen (10790 pollen*day*m−3) was shed than in all the other years monitored.
Fraxinus and Pinaceae displayed pronounced bimodal flowering periods (Fig. 6). Low pollen concentrations of Cupressaceae were recorded irregularly in autumn, mostly in October and November and in the last week of December. Urtica and Plantago pollen were not continuously recorded during the season.
3.4 The effect of meteorological factors
We observed an increased flowering intensity in spring flowering trees in 2015 and even more so in 2016. This was most likely elicited by abundant and evenly distributed rains throughout spring 2014 and the second half of 2014 including the winter months, which continued into spring 2015 (Table 4). As a result, the APIn in 2015 was higher than in 2014. In Istanbul’s winters, humidity is higher than it is in summer (Table 4) with pronounced daily fluctuations due to frequent precipitation events during this season. Water was sufficiently available during the vegetative growth period in winter and early spring during the years studied. The dry period in Istanbul lasts from May to September (Fig. 1). We investigated the effect of meteorological factors on the 15 main woody and herbaceous taxa with generalized additive models (GAMs) (Table 5).
In GAMs including the entire study period, we observed the following patterns (Table 5): The effect of temperature on pollen concentrations was significantly negative for tree taxa mainly flowering in winter and spring within April (Table 1 and Fig. 6). On Poaceae and Olea, with peak flowering in May, the effect faded. For the weeds with flowering phenophase in summer (Plantago, Amaranthaceae and Ambrosia), the effect of temperature turned to be the opposite. This applies also to Urticaceae, which have their peak in summer (Table 2, Fig. 6). Relative humidity had a positive effect on winter and spring flowering Cupressaceae, Fraxinus, Betulaceae and Salix, and in Fagaceae. Significances for relative humidity, pressure, and precipitation were mostly negative for taxa with a pollen season in summer.
4 Discussion
4.1 Flowering phenology
Airborne pollen can be used to infer the timing of flowering of taxa that are relevant in allergological terms. The intensity of the pollen season depends on the magnitude of the flowering, as well as on weather and climate (Dahl et al., 2013). The duration of the season, however, is bound to how it is defined. The 95% method based on the APIn (Andersen, 1991), can be suitable for low pollen concentrations (Bastl et al., 2018), as it is the case at the trap location in Büyükcekmece/Istanbul with APIns < 10.000 pollen*day*m−3 during 2013–2015. To compare, Kastamonu, located in the mountainous Turkish Black Sea Region with dense forest cover, has APIns > 100.000 pollen*day*m−3 (Çeter et al., 2012); Antalya, on the Mediterranean coast on the foot of the Taurus mountains has > 20.000 pollen*day*m−3 (Tosunoglu et al., 2015). The disadvantage of the percentual method is that the delineation of seasons depends on the flowering intensity, on missing data in the database, and can only be done retrospectively (Bastl et al., 2018; Grundström et al., 2019). The inherent bias for an earlier start of the season for years with lower intensities was seen, for example, in Poaceae and Ambrosia. Alternatively, with the 1st day of 5 non-zero concentrations, the start date of the season was generally delayed, except for taxa with high pollen production, such as Cupressaceae or Poaceae. For Fraxinus and Platanus, but also Pinaceae, Poaceae, and Plantago the season start date using both methods were very similar. In South Spain the pollen season start for Fraxinus and Populus was defined with 1 pollen grain *m−3 plus 5 non-zero concentrations (Velasco-Jiménez et al., 2020) suggesting that this method could possibly work also in Istanbul for some taxa. The five consecutive non-zero concentrations are not suitable for all taxa at our location because the start date would fall far into the main flowering phase of the taxon and even trespass the first peak. Grundström et al (2019), who analysed several methods to calculate the oak pollen season across Europe found that the cumulative sum method of three consecutive days with 5 p * m−3 delayed the start 10–12 days into the oak season as compared to the cumulative sum of 50 pollen grains. Further investigation targeted to individual taxa would be needed to delineate the most suitable method to define the pollen season in Istanbul. Other factors determine the duration of the pollen season. First, the taxonomic level of identification of a taxon under the light microscope, whether it consists of a species (i.e. Olea europaea), genus (i.e. Quercus) or family (i.e. Cupressaceae, Poaceae). Second, if conspecific plants flower at the same time (synchronically) or continuously staggered (asynchronically) and how many flowers are open per day (the flowering rate). The third important factor is weather conditions (Dahl et al., 2013). The pollen season start and end of herbaceous plants, for example, Urticaceae, showed considerable variations over three pollen seasons and a long flowering duration. In Urticaceae we can observe asynchronous flowering with the contribution of Parietaria judaica to the pollen spectrum of this family. Parietaria judaica, a perennial herb, grows and flowers all year long in shaded, ruderal urban (wall) communities of the city (Altay et al., 2010). Continuous growth ensures reproduction when availability of resources is unpredictable (Fotiou et al., 2011). The course of the Urticaceae season in Istanbul was comparable to the Urticaceae season in Thessaloniki (Fotiou et al., 2011), with continuous records from May to August and irregular ones stretching into November. In contrast, in Krakow, in an analysis of pollen season dynamics on a 17 year long data series, pollen seasons of herbaceous plants were reported to have generally low variability with regard to the season start and end date (Myszkowska et al., 2011). We explain this difference with summer drought in Mediterranean regions forcing herbaceous plants to an opportunistic flowering behaviour (Dahl et al., 2013). In Krakow, a typical city with warm temperate cfb climates of Europe, June, July and August are the wettest months (Climate-Data.org, 1999), while they are the driest in Istanbul. The pollen season start and end dates were less variable for grasses. The onset of the grass pollen season in Istanbul is preceded by regular equinoctial (winter) rains facilitating growth during the developmental phase (Dahl et al., 2013). This leads to rather short and intense flowering peaks. When abundant rains occur during anthesis, the grass pollen season can be prolonged (Dahl et al., 2013). We observed this phenomenon during 2014, when grass flowering coincided with persistent rainfalls in spring.
Woody taxa had generally shorter pollen seasons than herbaceous ones, which was also observed by Werchan et al. (2018) and Myszkowska et al. (2011). The season end of Pinaceae, however, was prolonged due to the bi-modal flowering of this taxon. Tosunoglu et al. (2015) identified a distinct pollen season for Cedrus in Antalya, from the end of September to mid-December, with highest concentrations in October. It could be envisioned to record Cedrus-pollen separately from the rest of the Pinaceae family also in Istanbul to better delineate the pollen season of this taxon. Cupressaceae, Carpinus and Fraxinus had long seasons, too. In the case of ash, for example, Fraxinus angustifolia flowers in early spring, but Fraxinus ornus in April. While Fraxinus angustifolia is a common ornamental tree, F. ornus grows in the submediterranean woods in the hinterland of the city. The consequence is an ash season that can possibly stretch from the last week of January into the beginning of May. We observed pollen of Carpinus rather irregularly, with two months difference in the start and the end date of pollen seasons in four years of observations. The Carpinus/Ostrya type in Split (Croatia) varied also six weeks in the season start (Puljak et al., 2016). Eurosiberian vegetation is found mostly about 80 km upwind and at ca. 15 km downwind of our trap location. Temporal shifts in the beginning of flowering due to temperature differences at the different growth locations, and atmospheric transport can explain the long season of this taxon.
4.2 Flowering intensity
Relative abundances and the SPIn of pollen are measures of flowering intensity. Differences in the magnitudes have to be discussed in the light of annual variations and the bias of the trap height for some taxa in 2013 and early 2014. Annual variation depends on meteorological conditions and species inherent physiological flowering dynamics in woody perennial plants. The weather determines the amount of resources (i.e. assimilates in form of carbohydrates, the resorption of nutrients) that a plant can accumulate (Bogdziewicz et al., 2017; Dahl et al., 2013; Dahl & Strandhede, 1996). In 2016, conspicuously high intensities of Cupressaceae, Quercus, Fraxinus, Pinaceae, Platanus and Carpinus seem to be the result of favourable environmental conditions for the accumulation of resources acting across families. We suspect a masting phenomenon across taxa and populations. Masting, in fact, does not occur independently of environmental factors (Bogdziewicz et al., 2017). At time intervals plant populations in a wider area spend these resources in highly synchronous mass reproduction (Koenig et al., 2015). Masting is typical for many wind-pollinated woody species (Pearse et al., 2016) including Betula, Quercus, and Fraxinus (Bogdziewicz et al., 2020; Dahl et al., 2013; Dahl & Strandhede, 1996; Tapper, 1992). The correlation of weather with synchronous fluctuations in pollination success of isolated populations (known as pollination Moran effect) acts on individuals of the same taxon but also across taxa and ecosystems (Hansen et al., 2020; Pearse et al., 2016). Interestingly, in 2016 the highest birch pollen concentrations since the start of the records were reported for Poland and connected to long distance transport from Southeast Europe early in the birch pollen season in April (Myszkowska et al., 2021).
An increased magnitude of pollen with pronouncedly higher daily peak values of Poaceae and Plantago growing within 10 m of the trap location was detected when the trap was positioned at three m height, than when it was positioned at 10 m height. Also, higher peak values of Olea, Salix and possibly Pinaceae in 2013 could result from stands in the vicinity. Conversely, several Platanus trees grow about 250 m (N)NE of the original trap location but pollen concentrations were not increased in comparison to other years. Pollen concentration, in fact, is highest in the immediate vicinity of the source (Adams-Groom et al., 2017). All in all, the change of the trap height did not constitute an apparent effect on most pollen type concentrations monitored, which is consistent with Rojo et al. (2019a).
The main contributing pollen taxa in Istanbul observed during the study period are very similar to the taxa reported for Thessaloniki (in order of magnitude: Cupressaceae, Quercus, Urticaceae, Oleaceae, Pinaceae, Poaceae, Platanus, Corylus, Amaranthaceae, Asteraceae, Populus and Plantaginaceae) (Gioulekas et al., 2004). The city is located at a similar geographical latitude as Istanbul and has warm-temperate cfa climate with more precipitation in summer (Climate-Data.org, 1999). Cupressaceae typically domineers the pollen spectrum of Mediterranean cities in the winter and spring (Puljak et al., 2016; Tosunoglu et al., 2015; Martínez-Bracero et al., 2015; Gioulekas et al., 2004). Cupressaceae pollen can be responsible for pollinosis early in the year (Asero et al., 2020; Charpin et al., 2019; Sposato & Scalese, 2013). However, sensitization to Cupressaceae allergen in western Istanbul seems low (3%) (Zemmer et al., 2021). Comparing data from our trap location in the western suburbs with data from Central Istanbul obtained between 2005 and 2006 (Celenk et al., 2010), relative abundances for Cupressaceae (34%), Fraxinus (3%), and Moraceae (2%) were similar. Abundances of Poaceae, Pinaceae, Quercus, Olea and Salix pollen, in contrast, were considerably higher at our site, while Platanus (24%) and Urticaceae pollen (13%) appeared to be more important in the city centre. In highly urbanised Central Istanbul ornamentals like Platanus and Fraxinus are frequent. In the historic peninsula there are still many Platanus trees originating from the Ottoman Empire (Baser, 2011) which are very large in size. Besides, Parietaria judaica is the most common species on the ancient stone walls of the city (Altay et al., 2010). In contrast, the hinterland of Istanbul features grass- and woodlands. Pollen from taxa of the Fagales order reached considerable concentrations at our study site, due to north-easterly air currents. Considering Fagales as a group facilitates the practitioner as well as self-empowered patients to identify periods of increased incidence of this allergenic pollen. Highly allergenic Ambrosia has become the main weed pollen contributor in western Istanbul with proven clinical relevance (Zemmer et al., 2021). A clinical pollen season (Pfaar et al., 2017) of ragweed identified on the base of pollen data from 2018 (Hoffmann et al., 2020), endorses this finding.
4.3 Meteorological factors
An important limiting factor for flowering intensity in the Mediterranean is precipitation (Galán et al., 2016; Dahl et al., 2013). Prior to anthesis, in the example of Betula, catkin initiation depends on the availability of assimilates during the vegetation period previous to flowering (Dahl & Strandhede, 1996). In summer-dry Istanbul, water availability plays an important role in this process (Table 4).
The flowering intensity of herbaceous plants such as grasses and weeds is influenced by meteorological conditions during vegetative growth (Dahl et al., 2013). The peak flowering period of grasses in Istanbul is May. Water was sufficiently available during the vegetative growth period in winter and early spring during the years studied, necessary for the induction of flowering shoots (Dahl et al., 2013). Drought, as a limiting growth factor in the study area, might have affected the intensity of the Urticaceae pollen season in 2013. This taxon has the peak period in summer, but flowers all year so that water deprivation might have reduced the vigour of the plants and the ability to produce flowers.
To assess the effect of meteorological factors during anthesis we ran GAMs. In aerobiological studies, the relationship between meteorological factors and pollen concentrations is often discussed on the base of correlations (Kluska et al., 2020; Bruffaerts et al., 2018) and regressions (Gioulekas et al., 2004) assuming a linear dependency (Le, 2003). The effects of meteorological factors studied in a GAM provide insight on flowering behaviour and can be part of predictive models (Cordero et al., 2021). Significances in the GAMs reflected direct impacts of meteorological factors on the pollen curve during peak flowering periods. If the factor units follow the pollen curve, the effect will be positive, otherwise negative. Temperature models were significantly positive when pollen concentrations rose with rising temperatures, as in the case of weeds. Summer rains – often torrential – reduce temperatures. As temperatures rise again and humidity deceases when skies clear, dehiscence occurs (Dahl et al., 2013). The effect turns negative when the pollen curve falls as temperatures are still on the rise as seen in spring flowering trees. Grasses and Olea flower at the onset of summer when temperatures increase and precipitation becomes less frequent. This may explain why the GAM model did not show a significant effect in temperature.
Humidity models were positive for early spring (winter) flowering trees, as Cupressaceae and Fraxinus. In Istanbul’s winters humidity is higher than in summer with high daily fluctuations due to frequent precipitation events during this season. Rising pollen curves must have coincided with rising mean humidity levels, when dehiscence can still occur at sunny spells during a day. In 2015, mean humidity was higher in February than in January and may have had an effect on the global model. Negative humidity signals for summer flowering weeds could be eighter linked to both short term precipitation effects as in Amaranthaceae or Ambrosia or to a seasonal decrease in humidity after the peak period in the course the pollen seasons as in Plantago.
In weeds, the signals of atmospheric pressure were the same as in the humidity models. The moist parameters humidity, pressure and precipitation had a similar negative effect, as they are interconnected (Brenner, 2004). This was not evident in spring flowering trees due to daily weather fluctuations.
Precipitation generally lowers pollen concentrations (Paschalidou et al., 2020). This phenomenon occurs due to wash out (Tormo-Molina et al., 2010), whereby 5 mm * h−1 are necessary for a pronounced effect (Kluska et al., 2020) and is seen also in correlation analysis (Bruffaerts et al., 2018). In Poaceae, for example, an extended period of rain during the last week of April and first decade of May in 2014 may have caused a decrease in airborne pollen concentrations and explain the negative effect in the GAM.
GAMs run over a time series of several years provide a more general picture on the effect of the factors studied on the pollen seasons, than seasonal or annual GAMs. A higher resolution of the effects on the pollen curve can be obtained with short term GAMs and used in forecasts (Cordero et al., 2021; Ravindra et al., 2019).
5 Conclusion
The main pollen contributing taxa were Cupressaceae, Poaceae, Pinaceae, Quercus, Carpinus, Olea, Fraxinus, Urticaceae, Salix, Amaranthaceae, Plantago, Morus, Acer, Platanus and neophytic ragweed. The definition of the pollen season, referring to the period when 95% of the APIn is dispersed, seems adequate due to low concentrations of airborne pollen. For Pinaceae, Platanus, Fraxinus, Cupressaceae, as well as for Poaceae and Plantago, the method of consecutive 5 non-zero daily concentrations could be an alternative to the percent-method. Further investigations on single taxa are needed to define a pollen season start detached from the percentual method. High pollen concentrations observed in 2016 indicate that weather cues prior to anthesis can trigger intense flowering across tree populations. We possibly witnessed a masting phenomenon. Further research on pollen seasons of neighbouring regions in 2016 would provide insight on the truth of this hypothesis. A change in how high the pollen trap was positioned mainly affected pollen concentrations of taxa growing in the immediate surrounding. Differences in the importance of pollen contributing taxa between the city centre and the suburbs suggest that one trap in the city centre and one each at the western and eastern outskirts of the city would assure representable pollen information for Istanbul. GAMs provide causative explanations on the effect of weather on pollen concentrations during anthesis. Meteorological factors showed contrasting effects on spring and summer flowering taxa.
Data availability
Data spreadsheets used for analysis are provided upon request.
References
Adams-Groom, B., Skjøth, C. A., Baker, M., & Welch, T. E. (2017). Modelled and observed surface soil pollen deposition distance curves for isolated trees of Carpinus betulus, Cedrus atlantica Juglans Nigra and Platanus Acerifolia. Aerobiologia, 33(3), 407–416. https://doi.org/10.1007/s10453-017-9479-1
Alan, Ş, Sarışahin, T., Acar Şahin, A., Kaplan, A., & Pınar, N. M. (2019). An assessment of ragweed pollen and allergen loads in an uninvaded area in the western black sea region of Turkey. Aerobiologia. https://doi.org/10.1007/s10453-019-09620-z
Altay, V., Özyiğit, I. I., & Yarci, C. (2010). Urban ecological characteristics and vascular wall flora on the Anatolian side of Istanbul, Turkey. Maejo International Journal of Science and Technology, 4(03), 483–495.
Andersen, T. B. (1991). A model to predict the beginning of the pollen season. Grana, 30(1), 269–275. https://doi.org/10.1080/00173139109427810
Ansotegui, I. J., Melioli, G., Canonica, G. W., Caraballo, L., Villa, E., Ebisawa, M., Passalacqua, G., Savi, E., Ebo, D., Gómez, R. M., Luengo Sánchez, O., Oppenheimer, J. J., Jensen-Jarolim, E., Fischer, D. A., Haahtela, T., Antila, M., Bousquet, J. J., Cardona, V., Chiang, W. C., & Zuberbier, T. (2020). IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. World Allergy Organization Journal, 13(2), 100080. https://doi.org/10.1016/j.waojou.2019.100080
Asero, R., Ceriotti, V., & Bonini, M. (2020). Cypress pollen allergy in milan: The story of an ongoing growth. European Annals of Allergy and Clinical Immunology. https://doi.org/10.23822/EurAnnACI.1764-1489.155
Bakis, Y., Babac, M., & Uslu, E. (2020). Turkish Plants Data Service (TÜBİVES). http://www.tubives.com/
Baser, B. (2011). Natural and cultural traces of the flora of Istanbul. Turkish Academy of Sciences Journal of Cultural Inventory, 9, 155–170.
Bastl, K., Bastl, M., Bergmann, K.-C., & Berger, U. (2019). How to do a clinical trial? Recommendations from the aerobiological point of view. World Allergy Organization Journal, 12(4), 100020. https://doi.org/10.1016/j.waojou.2019.100020
Bastl, K., Kmenta, M., & Berger, U. E. (2018). Defining pollen seasons: Background and recommendations. Current Allergy and Asthma Reports, 18(12), 73. https://doi.org/10.1007/s11882-018-0829-z
Bianchi, D. E., Schwemmin, D. J., & Wagner, W. H. (1959). Pollen release in the common ragweed (ambrosia artemisiifolia). Botanical Gazette, 120(4), 235–243.
Bogdziewicz, M., Fernández-Martínez, M., Bonal, R., Belmonte, J., & Espelta, J. M. (2017). The moran effect and environmental vetoes: Phenological synchrony and drought drive seed production in a mediterranean oak. Proceedings of the Royal Society B: Biological Sciences, 284(1866), 20171784. https://doi.org/10.1098/rspb.2017.1784
Bogdziewicz, M., Pesendorfer, M., Crone, E. E., Pérez-Izquierdo, C., & Bonal, R. (2020). Flowering synchrony drives reproductive success in a wind-pollinated tree. Ecology Letters, 23(12), 1820–1826. https://doi.org/10.1111/ele.13609
Brenner, I. S. (2004). The relationship between meteorological parameters and daily summer rainfall amount and coverage in west-central Florida. Weather and Forecasting, 19(2), 286–300. https://doi.org/10.1175/1520-0434(2004)019%3c0286:TRBMPA%3e2.0.CO;2
Bruffaerts, N., De Smedt, T., Delcloo, A., Simons, K., Hoebeke, L., Verstraeten, C., Van Nieuwenhuyse, A., Packeu, A., & Hendrickx, M. (2018). Comparative long-term trend analysis of daily weather conditions with daily pollen concentrations in Brussels Belgium. International Journal of Biometeorology, 62(3), 483–491. https://doi.org/10.1007/s00484-017-1457-3
Cariñanos, P., Casares-Porcel, M., & Quesada-Rubio, J.-M. (2014). Estimating the allergenic potential of urban green spaces: A case-study in Granada, Spain. Landscape and Urban Planning, 123, 134–144. https://doi.org/10.1016/j.landurbplan.2013.12.009
Celenk, S., Bicakci, A., Tamay, Z., Guler, N., Altunoglu, M. K., Canitez, Y., Malyer, H., Sapan, N., & Ones, U. (2010). Airborne pollen in European and Asian parts of Istanbul. Environmental Monitoring and Assessment, 164(1–4), 391–402. https://doi.org/10.1007/s10661-009-0901-1
EN 16868. (2019). Ambient air—Sampling and analysis of airborne pollen grains and fungal spores for networks related to allergy—Volumetric hirst method.
Çeter, T., Pinar, N. M., Güney, K., Yildiz, A., Aşcı, B., & Smith, M. (2012). A 2 year aeropalynological survey of allergenic pollen in the atmosphere of Kastamonu Turkey. Aerobiologia, 28(3), 355–366. https://doi.org/10.1007/s10453-011-9240-0
Charpin, D., Pichot, C., Belmonte, J., Sutra, J.-P., Zidkova, J., Chanez, P., Shahali, Y., Sénéchal, H., & Poncet, P. (2019). Cypress pollinosis: From tree to clinic. Clinical Reviews in Allergy & Immunology, 56(2), 174–195. https://doi.org/10.1007/s12016-017-8602-y
Climate-Data.org. (1999). Climate data for cities worldwide. https://en.climate-data.org/
Çoban, S., Bayraktar, S., & Akgül, M. (2016). Forest vegetation maps and its development in turkey: A case study from istanbul-belgrade forest. Forest Review, 47(7), 7–16.
Cordero, J. M., Rojo, J., Gutiérrez-Bustillo, A. M., Narros, A., & Borge, R. (2021). Predicting the Olea pollen concentration with a machine learning algorithm ensemble. International Journal of Biometeorology, 65(4), 541–554. https://doi.org/10.1007/s00484-020-02047-z
D’Amato, G., Cecchi, L., Bonini, S., Nunes, C., Annesi-Maesano, I., Behrendt, H., Liccardi, G., Popov, T., & van Cauwenberge, P. (2007). Allergenic pollen and pollen allergy in Europe. Allergy, 62(9), 976–990. https://doi.org/10.1111/j.1398-9995.2007.01393.x
D’Amato, G., Chong-Neto, H. J., Monge Ortega, O. P., Vitale, C., Ansotegui, I., Rosario, N., Haahtela, T., Galán, C., Pawankar, R., Murrieta-Aguttes, M., Cecchi, L., Bergmann, C., Ridolo, E., Ramon, G., Gonzalez Diaz, S., D’Amato, M., & Annesi-Maesano, I. (2020). The effects of climate change on respiratory allergy and asthma induced by pollen and mold allergens. Allergy, 75(9), 2219–2228. https://doi.org/10.1111/all.14476
Dahl, Å., Galán, C., Hajkova, L., & Pauling, A. (2013). The onset, course and intensity of the pollen season. In M. Sofiev & B. C. Bergmann (Eds.), Allergenic pollen: A review of the production, release, distribution and health impacts (pp. 29–70). Springer.
Dahl, Å., & Strandhede, S.-O. (1996). Predicting the intensity of the birch pollen season. Aerobiologia, 12(2), 97–106. https://doi.org/10.1007/BF02446601
Damialis, A., Gioulekas, D., Lazopoulou, C., Balafoutis, C., & Vokou, D. (2005). Transport of airborne pollen into the city of Thessaloniki: The effects of wind direction, speed and persistence. International Journal of Biometeorology, 49(3), 139–145. https://doi.org/10.1007/s00484-004-0229-z
Damialis, A., Traidl-Hoffmann, C., & Treudler, R. (2019). Climate change and pollen allergies. In M. R. Marselle, J. Stadler, H. Korn, K. N. Irvine, & A. Bonn (Eds.), Biodiversity and health in the face of climate change (pp. 47–66). Springer.
Eguiluz-Gracia, I., Mathioudakis, A. G., Bartel, S., Vijverberg, S. J. H., Fuertes, E., Comberiati, P., Cai, Y. S., Tomazic, P. V., Diamant, Z., Vestbo, J., Galán, C., & Hoffmann, B. (2020). The need for clean air: The way air pollution and climate change affect allergic rhinitis and asthma. Allergy, 75(9), 2170–2184. https://doi.org/10.1111/all.14177
Fernández-Rodríguez, S., Skjøth, C. A., Tormo-Molina, R., Brandao, R., Caeiro, E., Silva-Palacios, I., Gonzalo-Garijo, Á., & Smith, M. (2014). Identification of potential sources of airborne Olea pollen in the southwest Iberian Peninsula. International Journal of Biometeorology, 58(3), 337–348. https://doi.org/10.1007/s00484-012-0629-4
Fotiou, C., Damialis, A., Krigas, N., Halley, J. M., & Vokou, D. (2011). Parietaria judaica flowering phenology, pollen production, viability and atmospheric circulation, and expansive ability in the urban environment: Impacts of environmental factors. International Journal of Biometeorology, 55(1), 35–50. https://doi.org/10.1007/s00484-010-0307-3
Galán, C., Alcázar, P., Oteros, J., García-Mozo, H., Aira, M. J., Belmonte, J., Diaz de la Guardia, C., Fernández-González, D., Gutierrez-Bustillo, M., Moreno-Grau, S., Pérez-Badía, R., Rodríguez-Rajo, J., Ruiz-Valenzuela, L., Tormo, R., Trigo, M. M., & Domínguez-Vilches, E. (2016). Airborne pollen trends in the Iberian Peninsula. Science of the Total Environment, 550, 53–59. https://doi.org/10.1016/j.scitotenv.2016.01.069
Galán, C., Alcázar-Teno, P., Domínguez-Vilches, E., Villamandos de la Torre, F., & Garcia-Pantaleon, F. I. (1995). Airborne pollen grain concentrations at two different heights. Aerobiologia, 11(2), 105–109. https://doi.org/10.1007/BF02738275
Galán, C., Ariatti, A., Bonini, M., Clot, B., Crouzy, B., Dahl, A., Fernandez-González, D., Frenguelli, G., Gehrig, R., Isard, S., Levetin, E., Li, D. W., Mandrioli, P., Rogers, C. A., Thibaudon, M., Sauliene, I., Skjoth, C., Smith, M., & Sofiev, M. (2017). Recommended terminology for aerobiological studies. Aerobiologia, 33(3), 293–295. https://doi.org/10.1007/s10453-017-9496-0
Galán, C., Smith, M., Thibaudon, M., Frenguelli, G., Oteros, J., Gehrig, R., Berger, U., Clot, B., & Brandao, R. (2014). Pollen monitoring: Minimum requirements and reproducibility of analysis. Aerobiologia, 30(4), 385–395. https://doi.org/10.1007/s10453-014-9335-5
García-Mozo, H., Mestre, A., & Galán, C. (2010). Phenological trends in southern Spain: A response to climate change. Agricultural and Forest Meteorology, 150(4), 575–580. https://doi.org/10.1016/j.agrformet.2010.01.023
Gehrig, R., Maurer, F., & Schwierz, C. (2018). Designing new automatically generated pollen calendars for the public in Switzerland. Aerobiologia, 34(3), 349–362. https://doi.org/10.1007/s10453-018-9518-6
Gioulekas, D., Balafoutis, C., Damialis, A., Papakosta, D., Gioulekas, G., & Patakas, D. (2004). Fifteen years’ record of airborne allergenic pollen and meteorological parameters in Thessaloniki Greece. International Journal of Biometeorology, 48(3), 128–136. https://doi.org/10.1007/s00484-003-0190-2
Grundström, M., Adams-Groom, B., Pashley, C. H., Dahl, Å., Rasmussen, K., de Weger, L. A., Thibaudon, M., Fernández-Rodríguez, S., Silva-Palacios, I., & Skjøth, C. A. (2019). Oak pollen seasonality and severity across Europe and modelling the season start using a generalized phenological model. Science of the Total Environment, 663, 527–536. https://doi.org/10.1016/j.scitotenv.2019.01.212
Hansen, B. B., Grøtan, V., Herfindal, I., & Lee, A. M. (2020). The moran effect revisited: Spatial population synchrony under global warming. Ecography, 43(11), 1591–1602. https://doi.org/10.1111/ecog.04962
Heinzerling, L., Mari, A., Bergmann, K.-C., Bresciani, M., Burbach, G., Darsow, U., Durham, S., Fokkens, W., Gjomarkaj, M., Haahtela, T., Bom, A. T., Wöhrl, S., Maibach, H., & Lockey, R. (2013). The skin prick test – European standards. Clinical and Translational Allergy, 3, 1. https://doi.org/10.1186/2045-7022-3-3
Hintze, J. L., & Nelson, R. D. (1998). Violin plots: A box plot-density trace synergism. The American Statistician, 52(2), 181–184. https://doi.org/10.1080/00031305.1998.10480559
Hirst, J. M. (1952). An automatic volumetric spore trap. Annals of Applied Biology, 39(2), 257–265. https://doi.org/10.1111/j.1744-7348.1952.tb00904.x
Hoegh-Guldberg, O., Jacob, D., Taylor, M., Bindi, M., Brown, S., Camilloni, A., Diedhiou, A., Daljante, R., Ebi, K. L., Engelbrecht, F., & et al. (2018). Impacts of 1.5°C global warming on natural and human systems. In Global warming of 1.5°C. An IPCC special report on the impacts of global warming of 1.5°C above pre-industrial levels and related global greenhouse gas emission athways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty.
Hoffmann, T. M., Acar Şahin, A., Aggelidis, X., Arasi, S., Barbalace, A., Bourgoin, A., Bregu, B., Brighetti, M. A., Caeiro, E., Caglayan Sozmen, S., Caminiti, L., Charpin, D., Couto, M., Delgado, L., Di Rienzo Businco, A., Dimier, C., Dimou, M. V., Fonseca, J. A., Goksel, O., & Dramburg, S. (2020). Whole versus fragmented approach to EAACI pollen season definitions: A multicenter study in six Southern European cities. Allergy, 75(7), 1659–1671. https://doi.org/10.1111/all.14153
Jato, V., Rodríguez-Rajo, F. J., Fernandez-González, M., & Aira, M. J. (2015). Assessment of Quercus flowering trends in NW Spain. International Journal of Biometeorology, 59(5), 517–531. https://doi.org/10.1007/s00484-014-0865-x
Jenerowicz, D., Wojciech, S., Dańczak-Pazdrowska, A., & Polańska, A. (2012). Environmental factors and allergic diseases. Annals of Agricultural and Environmental Medicine, 19(3), 475–481.
Karatzas, K., Tsiamis, A., Charalampopoulos, A., Damialis, A., & Vokou, D. (2019). Pollen season identification for three pollen taxa in Thessaloniki, Greece: A 30 year retrospective analysis. Aerobiologia, 35(4), 659–669. https://doi.org/10.1007/s10453-019-09605-y
Katelaris, C. H., & Beggs, P. J. (2018). Climate change: Allergens and allergic diseases. Internal Medicine Journal, 48(2), 129–134. https://doi.org/10.1111/imj.13699
Kavgaci, A., Carni, A., Tecimeni, B., & Özalp, G. (2010). Diversity and ecological differentiation of oak forests in NW Thrace (Turkey). Archives of Biological Sciences, 62(3), 705–718. https://doi.org/10.2298/ABS1003705K
Kluska, K., Piotrowicz, K., & Kasprzyk, I. (2020). The impact of rainfall on the diurnal patterns of atmospheric pollen concentrations. Agricultural and Forest Meteorology, 291, 108042. https://doi.org/10.1016/j.agrformet.2020.108042
Koenig, W. D., Knops, J. M. H., Carmen, W. J., & Pearse, I. S. (2015). What drives masting? The phenological synchrony hypothesis. Ecology, 96(1), 184–192. https://doi.org/10.1890/14-0819.1
Le, C. T. (2003). Introductory biostatistics. Wiley-Interscience.
Martin, M. D., Chamecki, M., & Brush, G. S. (2010). Anthesis synchronization and floral morphology determine diurnal patterns of ragweed pollen dispersal. Agricultural and Forest Meteorology, 150(9), 1307–1317. https://doi.org/10.1016/j.agrformet.2010.06.001
Martínez-Bracero, M., Alcázar, P., Díaz de la Guardia, C., González-Minero, F. J., Ruiz, L., Trigo Pérez, M. M., & Galán, C. (2015). Pollen calendars: A guide to common airborne pollen in Andalusia. Aerobiologia, 31(4), 549–557. https://doi.org/10.1007/s10453-015-9385-3
Matricardi, P. M., Kleine-Tebbe, J., Hoffmann, H. J., Valenta, R., Hilger, C., Hofmaier, S., Aalberse, R. C., Agache, I., Asero, R., Ballmer-Weber, B., Barber, D., Beyer, K., Biedermann, T., Bilò, M. B., Blank, S., Bohle, B., Bosshard, P. P., Breiteneder, H., Brough, H. A., & Ollert, M. (2016). EAACI Molecular allergology user’s guide. Pediatric Allergy and Immunology, 27, 1–250. https://doi.org/10.1111/pai.12563
McInnes, R. N., Hemming, D., Burgess, P., Lyndsay, D., Osborne, N. J., Skjøth, C. A., Thomas, S., & Vardoulakis, S. (2017). Mapping allergenic pollen vegetation in UK to study environmental exposure and human health. Science of the Total Environment, 599–600, 483–499. https://doi.org/10.1016/j.scitotenv.2017.04.136
McKenna, O. E., Asam, C., Araujo, G. R., Roulias, A., Goulart, L. R., & Ferreira, F. (2016). How relevant is panallergen sensitization in the development of allergies? Pediatric Allergy and Immunology, 27(6), 560–568. https://doi.org/10.1111/pai.12589
Monroy-Colín, A., Silva-Palacios, I., Tormo-Molina, R., Maya-Manzano, J. M., Rodríguez, S. F., & Gonzalo-Garijo, Á. (2018). Environmental analysis of airborne pollen occurrence, pollen source distribution and phenology of Fraxinus angustifolia. Aerobiologia, 34(3), 269–283. https://doi.org/10.1007/s10453-018-9512-z
Myszkowska, D., Piotrowicz, K., Ziemianin, M., Bastl, M., Berger, U., Dahl, Å., Dąbrowska-Zapart, K., Górecki, A., Lafférsová, J., Majkowska-Wojciechowska, B., Malkiewicz, M., Nowak, M., Puc, M., Rybnicek, O., Saarto, A., Šaulienė, I., Ščevková, J., Kofol Seliger, A., Šikoparija, B., & Czarnobilska, E. (2021). Unusually high birch (Betula spp.) pollen concentrations in Poland in 2016 related to long-range transport (LRT) and the regional pollen occurrence. Aerobiologia, 37(3), 543–559. https://doi.org/10.1007/s10453-021-09703-w
Myszkowska, D., Jenner, B., Stępalska, D., & Czarnobilska, E. (2011). The pollen season dynamics and the relationship among some season parameters (start, end, annual total, season phases) in Kraków, Poland, 1991–2008. Aerobiologia, 27(3), 229–238. https://doi.org/10.1007/s10453-010-9192-9
Pacini, E. (2000). From anther and pollen ripening to pollen presentation. Plant Systematics and Evolution, 222(1/4), 19–43.
Paschalidou, A. K., Psistaki, K., Charalampopoulos, A., Vokou, D., Kassomenos, P., & Damialis, A. (2020). Identifying patterns of airborne pollen distribution using a synoptic climatology approach. Science of the Total Environment, 714, 136625. https://doi.org/10.1016/j.scitotenv.2020.136625
Pearse, I. S., Koenig, W. D., & Kelly, D. (2016). Mechanisms of mast seeding: Resources, weather, cues, and selection. New Phytologist, 212(3), 546–562. https://doi.org/10.1111/nph.14114
Peñuelas, J., Filella, I., Zhang, X., Llorens, L., Ogaya, R., Lloret, F., Comas, P., Estiarte, M., & Terradas, J. (2004). Complex spatiotemporal phenological shifts as a response to rainfall changes. New Phytologist, 161(3), 837–846. https://doi.org/10.1111/j.1469-8137.2004.01003.x
Pfaar, O., Bastl, K., Berger, U., Buters, J., Calderon, M. A., Clot, B., Darsow, U., Demoly, P., Durham, S. R., Galán, C., Gehrig, R., Gerth van Wijk, R., Jacobsen, L., Klimek, L., Sofiev, M., Thibaudon, M., & Bergmann, K. C. (2017). Defining pollen exposure times for clinical trials of allergen immunotherapy for pollen-induced rhinoconjunctivitis—An EAACI position paper. Allergy, 72(5), 713–722. https://doi.org/10.1111/all.13092
Picornell, A., Oteros, J., Ruiz-Mata, R., Recio, M., Trigo, M. M., Martínez-Bracero, M., Lara, B., Serrano-García, A., Galán, C., García-Mozo, H., Alcázar, P., Pérez-Badia, R., Cabezudo, B., Romero-Morte, J., & Rojo, J. (2021). Methods for interpolating missing data in aerobiological databases. Environmental Research, 200, 111391. https://doi.org/10.1016/j.envres.2021.111391
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D., & R Core Team. (2021). nlme: Linear and nonlinear mixed effects models (R package version 3.1–152).
Puljak, T., Mamić, M., Mitić, B., Hrga, I., & Hruševar, D. (2016). First aerobiological study in mediterranean part of Croatia Dalmatia: Pollen spectrum and seasonal dynamics in the air of split. Aerobiologia, 32(4), 709–723. https://doi.org/10.1007/s10453-016-9444-4
Ravindra, K., Rattan, P., Mor, S., & Aggarwal, A. N. (2019). Generalized additive models: Building evidence of air pollution, climate change and human health. Environment International, 132, 104987. https://doi.org/10.1016/j.envint.2019.104987
Rojo, J., Oteros, J., Pérez-Badia, R., Cervigón, P., Ferencova, Z., Gutiérrez-Bustillo, A. M., Bergmann, K.-C., Oliver, G., Thibaudon, M., Albertini, R., Rodríguez-De la Cruz, D., Sánchez-Reyes, E., Sánchez-Sánchez, J., Pessi, A.-M., Reiniharju, J., Saarto, A., Calderón, M. C., Guerrero, C., Berra, D., & Buters, J. (2019a). Near-ground effect of height on pollen exposure. Environmental Research, 174, 160–169. https://doi.org/10.1016/j.envres.2019.04.027
Rojo, J., Picornell, A., & Oteros, J. (2019b). Aerobiology: The computational tool for biological data in the air. Methods in Ecology and Evolution, 10(8), 1371–1376. https://doi.org/10.1111/2041-210X.13203
Rojo, J., Rapp, A., Lara, B., Fernández-González, F., & Pérez-Badia, R. (2015). Effect of land uses and wind direction on the contribution of local sources to airborne pollen. Science of the Total Environment, 538, 672–682. https://doi.org/10.1016/j.scitotenv.2015.08.074
Schroeder, F.-G. (1998). Lehrbuch der Pflanzengeographie. Quelle u. Meyer.
Sposato, B., & Scalese, M. (2013). Prevalence and real clinical impact of Cupressus sempervirens and Juniperus communis sensitisations in Tuscan Maremma Italy. Allergologia Et Immunopathologia, 41(1), 17–24. https://doi.org/10.1016/j.aller.2011.08.001
Szymczak, S., Häusser, M., Garel, E., Santoni, S., Huneau, F., Knerr, I., Trachte, K., Bendix, J., & Bräuning, A. (2020). How do mediterranean pine trees respond to drought and precipitation events along an elevation gradient? Forests, 11(7), 758. https://doi.org/10.3390/f11070758
Tapper, P.-G. (1992). Irregular fruiting in fraxinus excelsior. Journal of Vegetation Science, 3(1), 41–46. https://doi.org/10.2307/3235996
Tormo-Molina, R., Gonzalo-Garijo, M., Silva-Palacios, I., & Muñoz-Rodríguez, A. (2010). General trends in airborne pollen production and pollination periods at a mediterranean site (Badajoz, Southwest Spain). Journal of Investigational Allergology and Clinical Immunology, 20(7), 567–574.
Tosunoglu, A., Altunoglu, M. K., Bicakci, A., Kilic, O., Gonca, T., Yilmazer, I., Saatcioglu, G., Akkaya, A., Celenk, S., Canitez, Y., Malyer, H., & Sapan, N. (2015). Atmospheric pollen concentrations in Antalya South Turkey. Aerobiologia, 31(1), 99–109. https://doi.org/10.1007/s10453-014-9350-6
Turkish Statistical Institute (TUIK). (2020). Statistics by topic. Turkey health survey - period: 2019. www.tuik.gov.tr
Turoğlu, H. (2014). Detection of changes on temperature and precipitation features in Istanbul (Turkey). Atmospheric and Climate Sciences, 4, 549–562.
Velasco-Jiménez, M. J., Alcázar, P., Díaz de la Guardia, C., Trigo, M. D. M., de Linares, C., Recio, M., & Galán, C. (2020). Pollen season trends in winter flowering trees in South Spain. Aerobiologia, 36(2), 213–224. https://doi.org/10.1007/s10453-019-09622-x
Walter, H., & Lieth, H. (1960). Klimadiagramm-Weltatlas climate diagram world atlas. Fischer.
Weber, R. W. (2008). Guidelines for using pollen cross-reactivity in formulating allergen immunotherapy. Journal of Allergy and Clinical Immunology, 122(1), 219–221. https://doi.org/10.1016/j.jaci.2008.05.034
Werchan, M., Werchan, B., & Bergmann, K.-C. (2018). German pollen calendar 4.0 – update based on 2011–2016 pollen data. Allergo Journal International, 27(3), 69–71. https://doi.org/10.1007/s40629-018-0055-1
Zemmer, F., Cenk, E., Dahl, A., Galán, C., & Ozkaragoz, F. (2021). A multidisciplinary approach of outdoor aeroallergen selection for skin prick testing in the geographical area of greater Istanbul. European Annals of Allergy and Clinical Immunology. https://doi.org/10.23822/EurAnnACI.1764-1489.188
Zemmer, F., Karaca, F., & Ozkaragoz, F. (2012). Ragweed pollen observed in Turkey: Detection of sources using back trajectory models. Science of the Total Environment, 430, 101–108. https://doi.org/10.1016/j.scitotenv.2012.04.067
Acknowledgements
The data collection for this work was facilitated by Prof. Fatih Ozkaragoz, MD.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement of Universidad de Córdoba / CBUA with Springer Nature. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by FZ. The first draft of the manuscript was written by FZ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Conceptualization: FZ, CG, ÅD; Methodology: FZ, ÅD; Formal analysis and investigation: FZ; Writing—original draft preparation: FZ; Writing—review and editing: FZ, CG, ÅD; Supervision: CG, ÅD.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known conflict of interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zemmer, F., Dahl, Å. & Galán, C. The duration and severity of the allergenic pollen season in Istanbul, and the role of meteorological factors. Aerobiologia 38, 195–215 (2022). https://doi.org/10.1007/s10453-022-09742-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10453-022-09742-x