Abstract
Purpose
To investigate the utility of selected pattern electroretinogram (PERG) parameters—including N95 amplitude and N95/P50 ratio, and a BL-N95 amplitude—in the analysis of visual function(s) and for predicting changes in retinal ganglion cell structures in traumatic optic neuropathy.
Study design
A retrospective, observational case series performed at a single center.
Methods
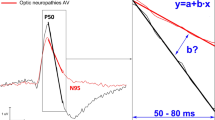
Forty-four eyes from 36 patients diagnosed with optic neuropathy were included. A BL-N95 amplitude was defined as the amplitude measured from baseline to the trough of N95. PERG and pattern visual evoked potential (pVEP) measures were acquired within 1 week after onset of optic neuropathies. To compare functional and anatomical changes, mean temporal peripapillary retinal nerve fiber layer (pRNFL) and average and minimum ganglion cell-inner plexiform layer (GC-IPL) thicknesses were measured using optical coherence tomography.
Results
Thirty-six patients (20 men, 16 women; mean age 37.5 ± 17.6 years) were evaluated. The BL-N95 amplitude was significantly smaller than the N95 amplitude (1.01 ± 0.56 μV and 2.45 ± 1.02 μV, respectively; p < 0.0001). Both the N95 (r = − 0.38, p = 0.010) and BL-N95 r = − 0.32, p = 0.029) amplitudes were significantly correlated with visual acuity. Although P100 latency was not correlated with all PERG parameters, the N95 (r = 0.32, p = 0.032) and BL-N95 (r = 0.41, p = 0.005) amplitudes demonstrated a positive correlation with P100 amplitude in pVEP. PERG parameters, including the N95 and BL-N95 amplitudes, and N95/P50 ratio, were not correlated with pRNFL thickness in optical coherence tomography. Only the BL-N95 amplitude demonstrated a significant correlation with GC-IPL.
Conclusion
The BL-N95 amplitude―measured from baseline to the trough of N95―was valuable in the analysis of visual function(s) and for predicting changes in retinal ganglion cell structures in traumatic optic neuropathy.



Similar content being viewed by others
References
Ostrin LA, Choh V, Wildsoet CF. The pattern ERG in chicks—stimulus dependence and optic nerve section. Vision Res. 2016;128:45–52.
Viswanathan S, Frishman LJ, Robson JG. The uniform field and pattern ERG in macaques with experimental glaucoma: removal of spiking activity. Invest Ophthalmol Vis Sci. 2000;41:2797–810.
Bach M, Brigell MG, Hawlina M, Holder GE, Johnson MA, McCulloch DL, et al. ISCEV standard for clinical pattern electroretinography (PERG): 2012 update. Doc Ophthalmol. 2013;126:1–7.
Holder GE. Pattern electroretinography (PERG) and an integrated approach to visual pathway diagnosis. Prog Retinal Eye Res. 2001;20:531–61.
Odom JV, Bach M, Brigell M, Holder GE, McCulloch DL, Mizota A, et al. ISCEV standard for clinical visual evoked potentials: (2016 update). Doc Ophthalmol. 2016;133:1–9.
Atilla H, Tekeli O, Ornek K, Batioglu F, Elhan AH, Eryilmaz T. Pattern electroretinography and visual evoked potentials in optic nerve diseases. J Clin Neurosci. 2006;13:55–9.
Holder GE. The incidence of abnormal pattern electroretinography in optic nerve demyelination. Electroencephalogr Clin Neurophysiol. 1991;78:18–26.
Berninger TA, Heider W. Pattern electroretinograms in optic neuritis during the acute stage and after remission. Graefes Arch Clin Exp Ophthalmol. 1990;228:410–4.
Lowitzsch K. Welt R [Pattern-reversal visual evoked potentials and electroretinography in the early diagnosis of chronic simple glaucoma]. EEG EMG Z Elektroenzephalogr Elektromyogr Verwandte Geb. 1991;22:217–23 in German.
Benedek K, Pálffy A, Bencsik K, Fejes I, Rajda C, Tuboly G, et al. Combined use of pattern electroretinography and pattern visual evoked potentials in neuroophthalmological practice. Ideggyl Sz. 2008;61:33–41 In Hungarian.
Ikejiri M, Adachi-Usami E, Mizota A, Tsuyama Y, Miyauchi O, Suehiro S. Pattern visual evoked potentials in traumatic optic neuropathy. Ophthalmologica. 2002;216:415–9.
Kanamori A, Nakamura M, Yamada Y, Negi A. Longitudinal study of retinal nerve fiber layer thickness and ganglion cell complex in traumatic optic neuropathy. Arch Ophthalmol. 2012;130:1067–9.
Yukita M, Machida S, Nishiguchi KM, Tsuda S, Yokoyama Y, Yasuda M, et al. Molecular, anatomical and functional changes in the retinal ganglion cells after optic nerve crush in mice. Doc Ophthalmol. 2015;130:149–56.
Porciatti V. Electrophysiological assessment of retinal ganglion cell function. Exp Eye Res. 2015;141:164–70.
Sehi M, Pinzon-Plazas M, Feuer WJ, Greenfield DS. Relationship between pattern electroretinogram, standard automated perimetry, and optic nerve structural assessments. J Glaucoma. 2009;18:608–17.
Bowd C, Tafreshi A, Zangwill LM, Medeiros FA, Sample PA, Weinreb RN. Pattern electroretinogram association with spectral domain-OCT structural measurements in glaucoma. Eye. 2011;25:224–32.
Goyal JL, Ghosh B, Sangit V, Kumar S, Jain P, Veerwal V, et al. Pattern ERG in central serous retinopathy. Doc Ophthalmol. 2015;130:141–7.
Berninger T, Arden GB. The Pattern Electroretinogram. In: Heckenlively JR, Arden GB, editors. Principles and practice of clinical electrophysiology of vision. Cambridge: MIT press; 2006. p. 291–300.
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
K. H. Kim, None; U. S. Kim, None.
Informed consent
For this type of study informed consent was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding author: Ungsoo Samuel Kim
About this article
Cite this article
Kim, K.H., Kim, U.S. Efficacy of N95 amplitude of pattern electroretinogram measured from baseline to N95 trough in the traumatic optic neuropathy. Jpn J Ophthalmol 63, 284–288 (2019). https://doi.org/10.1007/s10384-019-00664-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-019-00664-9