Abstract
As an essential nutrient, Nitrogen (N) availability is fundamental in evaluating forest productivity, and as such, understanding the effects of changing atmospheric N inputs in forest ecosystems is of high significance. While most field experiments have been employing ground fertilization as a method to simulate N deposition, two experimental forest sites in Italy have adopted the more advanced canopy N application approach. Here we present findings from a case study of wood core analyses of predominantly pure, even aged, Sessile oak (Quercus petraea L.) and European beech (Fagus sylvatica L.) forest stands, treated with either below or above canopy N fertilization, comparing between the two simulation pathways of increased N deposition. The potential effects of elevated N availability on total ring width, mean ring density, and their corresponding earlywood and latewood fractions are examined. Our results indicate inconclusive effects of the treatments on the ring width traits of both Q. petraea or F. sylvatica, although basal area increment patterns appeared to be affected divergently between the species and treatments. Mean and earlywood, but not latewood, densities on the other hand, exhibited a decrease in certain years of the treatment period in Q. petraea as result of the above canopy N application only, whereas F. sylvatica wood density showed no clear response to any of the treatments. Thus, we are describing distinct reactions of the two broadleaved species to the different experimental N deposition approaches, discussing potential growth patterns under increased N availability, and emphasizing the importance of considering wood density in assessments of tree biomass accumulation and essentially Carbon storage capacities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With ongoing anthropogenic meddling in the natural Nitrogen (N) cycle, elevation in atmospheric N deposition has been fairly steady in the past decades. Despite of existing evidence suggesting a decline in the total averaged European N deposition rate, the trends are spatially divergent, where certain areas still experience an upsurge (Schmitz et al. 2019). On a global scale, rates of inorganic N deposition were shown to increase by 8% between 1984–2016 from 86.6 to 93.6 Tg N yr−1 (Ackerman et al. 2019). Thus, propelling the interest of investigating its function in forests worldwide (Galloway et al. 2008; Bobbink et al. 2010; Etzold et al. 2020), in an attempt to uncover and define its underlying mechanisms (Khaine and Woo 2015; Carter et al. 2017), and consequent effects on Carbon (C) storage (Högberg 2007; Janssens et al. 2010; Quinn Thomas et al. 2010).
When examining plant C and N dynamics, there are multiple aspects to consider. The nutrimental condition of a habitat as a whole, and its inherent shifts, are reflected in the development of different tree compartments (Jiang et al. 2015), promptly and visibly at times (Guan and Wen 2011; Ward et al. 2013; Helm et al. 2023), yet may be latent and obscure as well (Wright et al. 1995; de Vries et al. 2014; Gagen et al. 2019). As such, the woody tissue may hold valuable information on the tree’s health and productivity (Helama et al. 2009; Poorter et al. 2010; Gagen et al. 2019; Cherubini et al. 2021), allowing for the provision of growth patterns and prediction models for fitness and mortality, based on tree core characteristics (Baker et al. 2004; Bigler et al. 2004; Cailleret et al. 2017; Liang et al. 2021). Various measures of tree rings have been studied within the reality of climate shifts (Ferretti et al. 2002; Scharnweber et al. 2011; St. George and Ault 2014; Borghetti et al. 2017). In that, wood density has been shown to alter under varying N inputs with divergent responses (Beets et al. 2001; Bucci et al. 2006; Pivovaroff et al. 2016), whilst Pretzsch et al. (2018) reported a general declining trend in wood density in Central European forests, alongside an increase in wood volume growth, over the recent century, suggesting a plausible link to rising N deposition rates over the same period.
It is evident that the tree canopy and atmospheric N hold a direct interaction where some canopy interception may occur with a consequent change in the amount and form of deposited N that actually reaches the ground, resulting in a considerable regulation of N distribution within the system (Sparks 2009). It has been demonstrated that as much as 80% of total reactive N descending from the atmosphere may be retained by the canopy before eventually arriving at the forest floor (Klopatek et al. 2006; Lovett and Lindberg 2011; Fenn et al. 2013; Houle et al. 2015), some of which may be immobilized or up-taken by the tree and its associated organisms, as well as being volatilized back as gaseous N (Bryan Dail et al. 2009; Nair et al. 2016). As experimental designs are innately bound by frequent constraints, a major culprit in studying forest ecosystems under increased N deposition, through means of experimental N addition, resides amongst others in the choice of fertilizer, application intervals, and administration method, and may limit favorable examination of hypotheses. While the majority of past studies investigating the effects of experimental N deposition in forests have implemented the traditional ground fertilization methods (Bebber 2021), an apt approach of above tree canopy N application (ACN), which may better mimic the actual deposition pathway than below canopy N application (BCN), has been adopted in more advanced research. In their thorough reviews, Bortolazzi et al. (2021) and Guerrieri et al. (2021b) addressed the matter, accentuating the eminent tree canopy-deposition relationship that is often overlooked, and presented many of the studies that incorporated the more appropriate approach of above canopy N administration.
Several ACN experiments were previously established in various forests and continents. In subalpine Engelmann spruce growing in Southwestern US, it was shown that N applied from above the tree crown is, similarly to ambient N deposition, mostly retained by the canopy, contributing to increased photosynthetic processes and foliar growth (Sievering et al. 2007; Tomaszewski and Sievering 2007). In a low-elevation red spruce and Eastern hemlock dominated forest in Northeastern US, the significance of the N-canopy interaction was further confirmed through ACN fertilization (Bryan Dail et al. 2009). A large-scale experiment was established in China (Zhang et al. 2015), examining different levels of ACN and BCN treatments in two separate forests dominated by native broadleaved species, where multiple investigations were performed and published since, presenting an array of species and habitat specific responses when measuring stem wood related affects (Yu et al. 2019; Guo et al. 2020; Zhang et al. 2022). An acid mist spray experiment in Scotland compared the effects of N and Sulphur (S) foliar applications in young managed Sitka spruce in Scotland, and detected a positive effect on stem radial growth when combining N + S during four years of treatment, with a minor insignificant negative effect of N alone (Sheppard et al. 2001), which later turned significantly positive after five years (Sheppard et al. 2004). In mature black spruce, growing in a Canadian boreal forest, limited effects of ACN treatment on xylem phenology and anatomy were found after three years (Lupi et al. 2012), which later faded after six years (Dao et al. 2015), to eventually conclude no significant effects. Both these latter spruce studies emphasized the importance of long-term monitoring for more constructive deductions in the context of wood traits, a tree tissue that may lag in immediate and/or obvious response.
The interrelation between N deposition and wood density for both Sessile oak (Quercus petraea) and European beech (Fagus sylvatica), two of the more ecologically and economically prominent and valuable broadleaved species in Europe (Mölder et al. 2019; Baumbach et al. 2019), has been reported in several observational and modelling studies (Braun et al. 1999, 2010, 2017,; Laubhann et al. 2009; Kint et al. 2012; Vannoppen et al. 2018; Hess et al. 2018). To the best of our knowledge, fewer studies have examined effects of experimental N application on mature oak and beech wood traits (Becker et al. 1996; Elhani et al. 2005; Ponton et al. 2019; Durand et al. 2020). Only one experiment in two Swiss forest sites used actual direct N application to oak (Q. cerris and Q. pubescens), and beech (F. sylvatica) branches, over a short period, and examined photosynthetic efficiency, to find an increase in the oak’s foliar photosynthetic measures, and an indeterminate decrease in beech (Wortman et al. 2012).
With the intention to contribute in filling some of these information voids, we perform an assessment of tree rings in two separate pure and mature Sessile oak and European beech forests. The objective here is to explore intrinsic growth traits in the form of tree ring widths and densities as measures for radial tree expansion and wood production, in forest stands subjected to two different elevated N treatment methods i.e., ACN and BCN. Our main hypotheses in this context are: i) while we expect no significant differences between the untreated experimental units in the period prior to N application, tree ring width and density development may be modified under increased experimental N inputs; ii) the two different N deposition simulation methods, may result in distinct effects on the examined tree ring characteristics; iii) and finally, the entire treatment period trend might reveal sperate inferences than that of the single inter-annual rings, which may reflect temporary time point responses.
Methods
Experimental sites
The oak site is located in the temperate forest of Monticolo, South-Tyrol, Italy (46°25′35'' N; 11°17′55'' E, 530 m elevation), with a mean annual temperature of 11.5 °C and mean annual precipitation of 835 mm. The soil is an acid brown loam, Cambisol (topsoil pH 4.8) overlaying porphyritic quartz rock. The site is dominated by Sessile oak (Quercus petraea (Matt.) Liebl.; ~ 96%), largely even-age averaged at 65 years, with a stand density of 1120 trees ha−1, mean height of 13.3 ± 0.1 m, and mean diameter at breast height (DBH) of 18.2 ± 0.2 cm. Recorded atmospheric bulk N deposition was estimated at ~ 6.5 kg N ha−1 yr−1 between 1996–1999. Initially managed for thinning, the forest stand has been unmanaged in the past decades and is part of a natural reserve, where no previous experiments were conducted.
The beech site is situated on the periphery of the sub-Alpine plateau of Pian del Cansiglio, Italy (46°3′19’’ N; 12°22′51’’ E, 1100 m elevation). With an oceanic climate, the mean annual temperature is 6.2 °C and mean annual precipitation is 2150 mm. The soil is formed on a karst bedrock with a carbonate rock matrix, Haplic Luvisol (topsoil pH 4.3). The site is exclusively dominated by mature European beech (Fagus sylvatica L.), even-age averaged at 135 years, with a stand density of 170 trees ha−1, mean height of 29.4 ± 0.1 m and mean DBH of 45.5 ± 1.1 cm. Historical total N deposition averaging around 17.5 kg N ha−1 yr−1. Previously managed as a high forest, it is currently recognized as a natural reserve. Last intervention of low intensity thinning was performed in early 2000, and no experimental history prior to the current research.
Fertilization treatments
The Monticolo experimental design is detailed and illustrated by Giammarchi et al. (2019). Briefly, the site consists of three treatments: 20 kg N ha−1 yr−1 applied below the tree canopy directly to the forest floor (BCN), 20 kg N ha−1 yr−1 applied above the canopy (ACN), and unfertilized control. Each treatment is replicated in three circular plots (12 m radius), for a total of nine experimental plots, arranged in a completely randomized design with a minimum of 10 m buffer zone to prevent contamination. N fertilizer, as NH4NO3 solution (4.3 g N L−1), has been applied five times during the growing season, from May to September, since 2015 and annually thereafter, until this day and ongoing. The BCN treatment is applied through manual spraying, ensuring uniform distribution. For ACN, rotating sprinklers mounted on telescopic masts are utilized, providing a largely uniform spray radius of 12 m above the tree canopy, from the center of the plot. The water supplied with the fertilized treatments (210 L H2O plot−1 yr−1) is equivalent to a negligible 0.46 mm of precipitation, compared to the region's average annual precipitation. No intervention was performed in the control plots. The conservative application rate of 20 kg ha−1 yr−1, roughly over three times the background atmospheric N deposition, was chosen to simulate feasible future scenarios of N deposition increases.
The Cansiglio experimental design is detailed by Teglia et al. (2022). Briefly, the site contains four treatments: an ACN treatment with 30 kg N ha−1 yr−1, BCN treatment with 30 kg N ha−1 yr−1, a double-dose BCN treatment with 60 kg N ha−1 yr−1 (denoted as BCN60), and unfertilized control. Each treatment was replicated in three plots using a randomized block design, for a total of twelve experimental plots. The control and both below canopy fertilization treatment plots are square (30 × 30 m), while the ACN plots are circular (20 m radius). N has been administered as NH4NO3 solution three times annually during the growing season, June to September, since 2015 and annually thereafter. Similarly to the Monticolo site, below canopy fertilization was sprayed manually, while above canopy N was supplied via rotating sprinkler towers from the center of the plots, with a conservative N application rate of two to three times the background atmospheric N deposition.
Tree core sampling and analysis
Due to pre-existing wood sampling of past projects in the sites since 2015, and inclination towards minimizing disturbances of the protected forests, limitations in the current sampling were enforced. One 5 cm long wood core sample per tree was collected with an increment borer (5 mm diameter) at breast height (~ 1.3 m) from six trees on average in each plot (in each site) to ensure a minimum of 18 tree repetitions per treatment. The cores from Monticolo were collected in early 2022 and the cores of Cansiglio in the end of the same year. In each site healthy trees with DBH around the stand average were chosen. DBH measurements of the cored trees were obtained accordingly. In the BCN60 treatment of the Cansiglio site two wood cores were damaged during the analysis process, leaving 16 cores instead of 18 for this treatment.
After air drying, the cores were sanded and analyzed for ring width and wood density via the high-frequency densitometry LIGNOSTATION™ method, considered to provide relative density measurements (Schinker et al. 2003; Shchupakivskyy et al. 2014; Koutsianitis and Barboutis 2017). The density analysis is based on the interaction between continuous electromagnetic waves emitted from a transmitter–receiver link of a small electrode system and the dielectric properties of the wood. The instrument simultaneously measures ring width through optically scanning the core path with a high-resolution camera. Analysis with the LIGNOSTATION™ allows for distinguishing between the seasonal sections of the total ring, providing separate earlywood and latewood density and width values.
Data and statistical analyses
The analysis of tree cores was performed on rings dating back two decades up to the year 2000. While the focus of the study was the examination of the N application period (2015 and onward), an additional 15-year period prior to the beginning of the experiment was chosen as a satisfactory time frame baseline for representing the state of the stands prior to any experimental manipulation, for comparing between the two periods of pre- and post-treatment. The most recent ring of each core (2021 for Monticolo and 2022 for Cansiglio) was omitted from the analysis to avoid potentially misreading non-fully consolidated rings. Tree rings were cross-dated with the CDendro software. No age-related trends or other confounding patterns were detected for the analyzed period, thus not warranting for detrending or standardization of the data, and eliminating concerns of potentially loosing conceivable N treatment signals. Rather, the entire raw data was used with no manipulation or any removal of data points, and all the presented graphical plots are based on treatment averages of the observed values.
Basal area increment (BAI) was estimated as an extended measure for stem radial expansion and annual tree growth. BAI was computationally reconstructed using the analyzed total ring widths and the coinciding current year DBH measurements. DBH values for past years were back-calculated using total ring widths under the rough assumption of an even total width around a circular stem area section. Furthermore, to assume the power and necessity of including wood density in tree C storage assessments, an interaction between radial growth and density was to be evaluated. For this purpose, we followed previous studies who used the biomass area increment measure (IAB; Vannoppen et al. 2018; Van Den Berge et al. 2021), or BAIden as it is referred to by Giberti et al. (2022) and referenced here, produced by combining BAI with the annual mean ring density. An overview of the mean values of all the above-mentioned measures and computed response variables is given in Table 1.
A linear mixed effects model was chosen as a robust approach for statistical analysis of the longitudinal tree ring time series. Best model fits were chosen according to the research objectives for the fixed effects and optimization of random effects, while considering the Akaike Information Criterion (AIC) and using Maximum Likelihood (ML) and Restricted Maximum Likelihood (REML) approximations when appropriate. Considering the use of the raw data for analyses, and to address potential temporal dependence and autocorrelation between consecutive tree rings, an autoregressive term of type AR1 for the year variable within the tree was incorporated in all the models, which significantly reduced the AIC:
The term describes the correlation between the residual errors (\({\varepsilon }_{ij}\) and \({\varepsilon }_{{i}{\prime}j}\)) for two consecutive observations (i and i′) within the same tree (j). Where \(\rho\) is the autoregressive parameter representing the correlation between the observations in the same tree across different years. \({\sigma }^{2}\) is the residual variance.
To account for the tree core repeated measurements and between individual variability in each treatment level, the subject unit of ‘tree’ was set as a random effect. The ‘plot’ unit (as the subplot) was included as a random effect in the initial model, yet it was found non-constructive to the model fit and outcome, and therefore was not included in the final model. For potential within individual variability, a random slope was additionally set in the random ‘tree’ term, which further improved the model. For each response variable a different random slope was indicated according to the best model fit.
As an initial approach for distinguishing between the two periods of prior and post the experiment onset a dummy variable named ‘onset’ was defined with two levels: ‘pre_2015’ representing the pre-experiment period of the years 2000 until 2015, and ‘post_2015’ representing the post-experiment period for the years after 2015. ‘treatment’ and ‘onset’ were set as fixed effects with an interaction to investigate the average effects of treatment in each period. ‘tree’ was set as a random effect. The best fitted model formula was specified as follows:
where \({RT}_{ij}\) represents a given ring trait response variable for the i-th observation in the j-th tree. \({\beta }_{0}\) is the intercept. \({\beta }_{1}\), \({\beta }_{2}\), and \({\beta }_{3}\) are the fixed-effect coefficients for the treatment, onset, and the interaction, respectively. \({u}_{0i}\) and \({u}_{1i}\) represent the random intercept of the tree and random slope term (either onset or DBH, according to the better fit) for each tree, respectively. \({\varepsilon }_{ij}\) is the residual error term.
Next, for a further investigation of potential treatment effects on the ring traits within each individual annual ring, ‘treatment’ and ‘year’ were set as fixed effects with an interaction, where ‘year’ was set as a categorical variable. ‘tree’ was set as a random effect. The best fitted model formula was specified as follows:
where \({RT}_{ij}\) represents a given ring trait response variable for the i-th observation in the j-th tree. \({\beta }_{0}\) is the intercept. \({\beta }_{1}\), \({\beta }_{2}\), and \({\beta }_{3}\) are the fixed-effect coefficients for treatment, year, and the interaction, respectively. \({u}_{0i}\) and \({u}_{1i}\) represent the random intercept for the tree and a random slope term of year, respectively. \({\varepsilon }_{ij}\) is the residual error term.
Finally, for BAI and BAIden, an evaluation of the overall patterns across the total analyzed periods divided into the two periods of prior and post the experiment was performed, while keeping ‘year’ as a numerical predictor, to produce slope trend values. Here, an addition of ‘DBH’ as a fixed factor proved to improve the models, but not the random slope term. For the pre-treatment period, a general trend for the entire stand was derived for the experimental units combined, while for the post-treatment period the treatments were kept separate as per the previous models:
Equation 4 is for the pre-treatment period, and Eq. 5 is for the post-treatment period. Where \({RT}_{ij}\) represents either the BAI or BAIden response variables for the i-th observation in the j-th tree. \({\beta }_{0}\) is the intercept. \({\beta }_{1}\), \({\beta }_{2}\), \({\beta }_{3}\), and \({\beta }_{4}\) are the fixed-effect coefficients for treatment, year, the interaction of year and treatment, and DBH, respectively. \({u}_{0i}\) represents the random intercept for the tree. \({\varepsilon }_{ij}\) is the residual error term.
After deriving an initial inference from the linear mixed models, particular significance determination was performed for the differences between each treatment pair using the Tukey post-hoc test for multiple pairwise comparisons (Tables S1-S28). A Z-test was performed for determining significant differences between the BAI and BAIden slope values within the individual treatments (Table S29).
All computation was carried out with R software: BAI reconstruction with the ‘dlpR’ package; linear modeling with the ‘nlme’ package; and post hoc analysis with the ‘emmeans’ package (Bunn 2008; Pinheiro and Bates 2023; Lenth et al. 2023; R Core Team 2023).
Results
Oak ring width and wood density
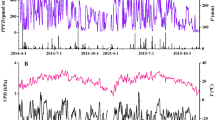
Ring widths of Q. petraea showed marked inter-annual oscillations through the entire analysis period, where total, earlywood, and latewood ring widths all followed a parallel trend (Fig. 1a,b,c). No overall differences were determined between any of the treatments in any of the ring width traits at any point in time by the ‘onset’ model (Table 2). With that said, when testing for differences between the treatments in each individual year (Tables S7-S9), a significant decrease in the earlywood width of ACN compared to the control was found by the post-hoc analysis, in 2015 (Fig. 1b; Table S8, p = 0.008), as well as in 2009, prior to the experiment onset (Table S8, p = 0.045).
Monticolo ring data of total (a) earlywood (b), and latewood (c) widths, and mean (d), earlywood (e), and latewood (f) densities, between 2000 and 2020 for Q. petraea. General averages for each annual growth ring (n = 18, SE). Note the y axis for densities is not set to zero. Dashed line indicates the experiment onset in 2015, with the light gray area thereafter indicating the post-treatment fertilization period. Asterisks indicate significant post-hoc pairwise comparison differences (p < 0.05)
Ring wood density exhibited lower inter-annual variability, and similarly to width, mean, earlywood, and latewood ring densities all followed a comparable and similar trend (Fig. 1d,e,f). Non of the ring density measures presented an overall effect of any treatment in any period as indicated by the ‘onset’ model (Table 2). This is despite a possible decreasing trend that was visually suggested in the mean density of the ACN treatment from 2017 (on the third year after the beginning of fertilization) and onwards, while BCN stayed closely uniform with the control (Fig. 1d). Indeed, this seeming decrease became meaningful in 2018 as was indicated by the mixed model results of the individual annual rings (Table S10), and the post-hoc analyses confirmed that ACN had a significantly lower mean density than BCN (Table S10, p = 0.018), yet not compared with the control (Table S10, p = 0.068). The difference between ACN and BCN was reduced in 2019 (Table S10, p = 0.062), while becoming significant between ACN and the control (Table S10, p = 0.043). In the final analyzed year, the extent of reduction between ACN and the other treatments was further diminished (Table S10, p = 0.097, p = 0.141). Earlywood density (Fig. 1e), which appeared visibly more variable than mean density was indeed slightly more impacted by N addition in both 2018 and 2019 (Table S11, p = 0.011 and p = 0.033, for ACN vs. BCN in 2018 and 2019, respectively, and p = 0.046 for ACN vs. control in 2019). For latewood density (Fig. 1f), no significant treatment response was determined (Table S12). None of the density measures were found significantly different in any individual year prior to N application (pre-treatment period). All mixed model and post-hoc results for the treatment effects in each individual annual tree ring, and full onset models, for Q. petraea may be found in Online Resource 1.
Beech ring width and wood density
For F. sylvatica growing in the Cansiglio forest, all width measures followed a rather unison trend for all treatments (Fig. 2a,b,c), with notable inter-annual fluctuations. Similarly to the case of Q. petraea, no treatment effects were found by the ‘onset’ model for any of the ring width measures in either of the two time periods (Table 3), yet the statistical analysis of the individual annual rings revealed several significant differences by both the mixed model and post-hoc tests (Tables S19-S21), here in each of the ring width traits. For total width, significantly lower in ACN compared to BCN60 in 2016 (Table S19, p = 0.022) and nearly significant in 2009 (Table S19, p = 0.053; Fig. 2a). For earlywood width, significant differences appeared in 2015, lower in ACN compared to BCN (Table S20, p = 0.002), and compared to BCN60 in 2016 (Table S20, p = 0.04; Fig. 2b). For latewood width, BCN60 was found significantly higher compared to all other treatments in 2006 (Table S21, p = 0.023, p = 0.036, p = 0.042), and compared to ACN in 2017 (Table S21, p = 0.028; Fig. 2c).
Cansiglio ring data of total (a) earlywood (b), and latewood (c) widths, and mean (d), earlywood (e), and latewood (f) densities, between 2000 and 2021 for F. sylvatica. General averages for each annual growth ring per treatment (n = 16–18, SE). Note the y axis for densities is not set to zero. Dashed line indicates the experiment onset in 2015, with the light gray area thereafter indicating the post-treatment fertilization period. Asterisks indicate significant post-hoc pairwise comparison differences (p < 0.05)
Ring density of F. sylvatica displayed a high intra-annual variability (Fig. 2d,e,f). No significant overall impacts of any treatment were indicated by the ‘onset’ analysis for any of the density traits, prior nor post N application (Table 3). While, the mixed model test of the individual years suggested some significant differences in each of the three density measures (Tables S22-24), none were reaffirmed by the subsequent post-hoc analysis. All mixed model and post-hoc results for the treatment effects in each individual annual tree ring, and the full onset models, for F. sylvatica may be found in Online Resource 2.
BAI and BAIden trends
After establishing no significant differences in total ring widths and mean densities between the treatments prior to the experiment, BAI and BAIden trends for the pre-treatment period were computed for all experimental units combined to present the overall temporal trends in the entire forest stand. The BAI trend for both oak and beech trees was generally negative in the two decades since 2000 (Fig. 3a,c; Table 4), and although extremely minimal when looking at the years prior to the experiments, the downward trend appeared to notably increase in the years after 2015. During the period of N fertilization, the ACN treatment seemed to somewhat attenuate the decline in the negative BAI trend of the two species, as indicated by the milder slopes of ACN compared to the control. While in Monticolo the BCN treatment was well paralleled with the control, in Cansiglio both BCN and BCN60 treatments had the opposite effect of ACN, inducing a greater decline in the BAI trend, which was closely significant for BCN compared to ACN (Table 4, p = 0.052), but not nearly as much compared to the control (p = 0.385).
Basal area increment (BAI) and biomass area increment (BAIden) trends for Q. petraea in Monticolo (a, b), and F. Sylvatica in Cansiglio (c, d). General patterns for pre-experiment years (n = 54–60), and patterns separated by treatment for post treatments years (n = 16–18). Dashed line indicates the experiment onset in 2015, with the light gray area thereafter indicating the post-treatment fertilization period
The BAIden trends in the pos-treatment period did not display markedly different patterns than that of BAI as a whole, generally slightly decreasing the observed BAI slope (Fig. 3b,d; Table 4). Yet only in the ACN treatment of Q. petraea, the incorporation of density in BAI to produce BAIden seemed to somewhat amend the treatment effect that was observed in the BAI trend (Fig. 3a,b), resulting in a larger slope. An outcome that might be expected by the corresponding decreased mean density (Fig. 1d). In F. sylvatica, although no concrete effects of N on mean density was concluded, when considering it within the BAIden trend, here the impact of density appeared to be prominent in both of the below canopy treatments (Fig. 3c,d; Table 4), as the decline that was observed in their BAI trends was significantly contracted for BCN and nearly significant for BCN60 (Table S29, p = 0.027 and p = 0.059, respectively). Full mixed model and post-hoc results for BAI and BAIden of both species may be found in Online Resource 3.
Discussion
It is evident that the majority of existing N application studies in oak species reveal a positive relation between N availability and radial growth (Jennings et al. 2016; Becker et al. 1996). In Q. petraea in particular, an increase in ring width expansion was detected following administration of an excessive 220 kg N ha−1 yr−1 (along with Ca fertilization) to the forest floor during two consecutive years (Ponton et al. 2019; Durand et al. 2020). The positive effects on radial growth were immediate, yet diminished after five years of monitoring, while no effects on wood density were observed at all. While the effects of N amendment in trees may manifest either promptly or only some years after N application (Sheppard et al. 2001, 2004), in our Q. petraea, after six years of consecutive, and moderate, annual N fertilization, neither of the treatments induced changes in ring widths compared to the control.
We observed a general high inter-annual variability in the ring widths of Q. petraea in Monticolo, and an overall negative radial growth trend over the past two decades. Inter-annual variability in ring widths of Q. petraea was previously linked to various climate variables, where precipitation frequently enhanced radial growth and temperature had a negative effect (Cedro 2007; Arend et al. 2011; Härdtle et al. 2013; Delpierre et al. 2016), dependant on the seasonal dynamics, of the current and previous years. Nevertheless, oaks do exhibit high plasticity in their reaction, often displaying marked resilience to environmental stress (Friedrichs et al. 2009; Mérian et al. 2011; Lebourgeois et al. 2013; Trouvé et al. 2017). Although we show a significant decrease in the earlywood of ACN compared to the control in 2015, it is doubtful to conclude that the effect of the N application is involved since the formation of earlywood ends in Q. petraea in adjacency to the beginning of the growing season (addressed later), and thus in 2015 ended prior to the very first fertilization event of the experiment. Together with the fact that in 2009 a similar significant difference was found, these results may be rather attributed to incidental divergences stemming from the higher overall variance in earlywood traits and the general susceptibility of ring width towards climatic conditions.
Mean ring wood density of our oaks, in contrast to ring width, seemed to experience a decline already on the third year for the ACN treatment, to become significant in reduction on the fourth year, compared to the BCN treated trees which remained rather consistent with the untreated control. Decrease in wood density is not uncommon in N application experiments (Ross et al. 1979; Hättenschwiler et al. 1996; Beets et al. 2001; Raymond and Muneri 2000; Kostiainen et al. 2004), and is possibly related to changes in stem hydraulic architecture and water use efficiency. Wood density has a direct link to transport system performance and vigor of the tree stem, where thicker cell walls are made to endure larger negative water pressure stress, and are consequently more resistant to cell implosion and organ cavitation (Hacke et al. 2001, 2005; Zanne et al. 2010). Lower wood densities on the other hand, have been shown to go in accord with increased stem conductivity (Hacke et al. 2010), while associated with higher foliar N at the same time (Bucci et al. 2006; Pivovaroff et al. 2016).
When comparing to reported literature values for Q. petraea (Zhang et al. 1993; Bergès et al. 2000, 2008; Cedro 2007; Vavrcik and Gryc 2012), our oaks seem to present general average wood densities on the higher end of the species spectrum, whilst total ring widths being lower. Thus, the relatively prompt apparent response of mean ring density to the direct contact of applied N with the foliage of the ACN treated trees, despite our conservative amount of added N, may be due to a sufficient leeway in the form of robust woody tissue to spare, for a worthwhile compromise towards improved conductivity (Pérez-de-Lis et al. 2018). Once the tree canopy experienced an elevated N supply, a shift could have been triggered in an aim to support the increased essential nutrient inflow. The additional N availability could have promoted growth processes where the decrease in density may allow for a potentially more efficient tree stem transport system.
According to our results of a moderately higher significance in the decrease of earlywood density than that of the mean density in the ACN treatment, in conjunction with no significant effects on latewood density, it is evident that the earlywood might be the more sensitive fraction of the ring and the determining factor in the overall mean ring density of Q. petraea (that, in spite of the latewood section having a slightly higher proportion of the total ring). This proposition is contrary to the findings of Pretzsch et al. (2018) who argued the opposite, namely that latewood is the main determinant in the mean ring density. With that said, the role of other environmental variables i.e., CO2, temperature, and growing season prolongations, where not excluded from their analyses, therefore these may come as a reasoning for the distinction in findings. Pretzsch et al. (2018) did however attribute a century long general decrease in wood density to concomitant N deposition increases in Central European forests, which is in line with the density reduction that we observed.
Earlywood is largely of lower density than latewood and contains a higher percentage of larger transport vessels providing the main pathway for water and nutrient flux during the growing season (Feuillat et al. 1997; Umebayashi et al. 2008). Its’ development begins and consolidates rather early in ring-porous species, even prior to full leaf formation (Kitin and Funada 2016), and earlywood to latewood transition in Q. petraea in particular may commence as soon as the month of May (Michelot et al. 2012b). At first glance, this might appear counterintuitive when considering our fertilization regimen, beginning right after leaf maturation (around June in the Monticolo forest). Nonetheless, it has been demonstrated that the earlywood often bears an imprint of the previous years’ (as well as the pre-growing season) conditions (González and Eckstein 2003; Fonti and García-González 2004; Fonti et al. 2007; González-González et al. 2014). In Q. petraea, earlywood and its vessel area composition have been linked to the precipitation rate of the preceding growing seasons in certain environments (Fonti and García-González 2008), while ring density as a whole was impacted by various previous year climate variables (Vannoppen et al. 2018). It has been suggested that earlywood formation originates in the over-wintered cambial cells of the prior growing season (Gričar and Čufar 2008). Further, earlywood development depends on past non-structural C reserves (Barbaroux and Bréda 2002; Kimak and Leuenberger 2015). For both C and N, allocation to different compartments of the tree is spread out along the annum, where N transportation ensues later in the vegetative season, peaking around senescence with leaf N resorption (El Zein et al. 2011a, 2011b; Bazot et al. 2013; Gilson et al. 2014). Therefore, one may assume that increased direct foliage N uptake, during the growing season period, between June–September in our case, might propel an altered forthcoming-season earlywood production, of presumably wider transport vessels together with a less dense fibrous vicinity, to support likely another anticipated nutrient rich season.
In regard to the BCN treated trees in Monticolo, which did not seem to experience this new nutrient balance, we may suggest that the distinction could arise from the rather conservative amount of ground N fertilization, that might have been initially intercepted by the thick perennial leaflitter layer in our forest stands prior to even reaching the soil, and possibly up-taken by lower organisms thereafter, and/or being volatilized in part at the same time. Indeed, Da Ros et al. (2023) found a significant difference in the N recovery between the two treatments in our site after a 15N tracer fertilization event during the 2016 growth season in Monticolo, where the vast majority of labeled N in the BCN treatment was retained by the litterfall layer and soil within 4 months post fertilization, twice as much as in the ACN treatment. In contrast, tree N recovery, albeit detected in rather low amounts altogether, was twice as much in ACN than in BCN.
In the case of the beech trees in the Cansiglio forest site, lower earlywood width was found in the ACN treatment compared to both below canopy N treatments in the first two years of the experiment. In the latewood width, the ACN treatment was also shown as significantly lower than that of BCN60 only in 2017, while being significantly higher in BCN60 compared to the rest of the treatments in 2006. At the same time, the total ring width of ACN was significantly lower than in BCN60 only, in 2016. These differences may plausibly imply that above canopy N application may cause a mild decrease in the ring width of F. sylvatica, while below canopy fertilization may somewhat increase it, yet when taking into account the entire time series, it does not appear as a consistent tendency considering significant difference determined in the pre-experimental year. Therefore, at this point no definitive statements can be confidently made regarding the effects of any type of N application on the ring with of our beech trees. The contrasting results of the linear mixed model and post-hoc tests for the density traits, ultimately showing no significant effects of any treatment in this regard, reinforce the possibility that the supposed responses seen in the ring widths may essentially be due to a high inter-subject variability of the individual trees, resulting in random signals.
In other studies relating N availability with beech tree ring characteristics, Guerrieri et al. (2021a) found a positive correlation of soil and leaf N to BAI and a negative link with wood density. A similar scale negative effect of N deposition on wood density was reported by Vannoppen et al. (2018). In ~ 80 old F. sylvatica stands fertilized with either N or NPKCa (100 kg N h−1a yr−1 during two consecutive years), radial growth increase was determined for the NPKCa treatment but not for N alone (Elhani et al. 2005). BAI based growth models concerning N deposition in European forest monitoring plots showed significant positive effects on Scots Pine, Norway Spruce, and oaks, yet for beech significance was only nearly achieved (Laubhann et al. 2009). A three-decade monitoring period of BAI revealed a decline in F. sylvatica between 1987–2014, along with an inversive relation with N deposition where a positive effect was observed up to 26 kg N h−1a yr−1, and turned negative thereafter (Braun et al. 2017). This was reinforced by the models of Kint et al., (2012), who showed a corresponding inversive trend where the negative effect ensued around 21 kg N ha−1 yr−1 (and 28 kg N ha−1 y−1 for Pedunculate Oak for that matter). Hess et al. (2018), likewise found both favorable and opposing effects on beech ring widths and radial growth with increasing N deposition (of up to 14 kg N h−1a yr−1), depending on interactions with temperature, precipitation, and tree age. It is thus apparent that N loads beyond certain thresholds may not act agreeably for overall tree productivity and may farther turn antagonistic (Fleischer et al. 2013; Flechard et al. 2020a, 2020b). The ambient N deposition of ~ 17.5 kg N ha−1 yr−1 in the Cansiglio forest surrounds the critical N loads for beech suggested above, and considering the additional supplied amount of 30 kg N ha−1 yr−1 (more so in the double-dose BCN treatment), exceeds the presumable threshold considerably. While our beech trees seemed to remain supposedly indifferent after seven years of fertilization in terms of the measured ring traits, the BAI results did point to a possible adverse effect of the BCN treatments after all. Indeed, the general declining trend in BAI appeared to be enhanced in both BCN treatments compared to the control, suggesting that a limited undertone response to the added N might have existed nonetheless. The implied opposite effect of ACN on the BAI trend further supports this notion, and at the same time indicates that the mode of N application may in fact matter here as well, and could induce opposing radial growth effects in F. sylvatica. For the oak trees in Monticolo, growing under ~ 6.5 kg N ha−1 yr−1 of ambient N deposition, total ring width also did not change with any of the added 20 kg N ha−1 yr−1 treatments, while the declining BAI trend seemed to be similarly mitigated by the ACN treatment, although with no evident impact of BCN in this case.
Climate impacts on beech stem growth and ring development have been studied extensively (Leuschner et al. 2001a; Bouriaud et al. 2004, 2005; Scharnweber et al. 2011; Michelot et al. 2012a). Although beech appears often as the more sensitive to environmental conditions altogether between the two species (Campioli et al. 2012; Michelot et al. 2012a), it was shown to prevail as the dominant one in mixed forests with oak (Leuschner et al. 2001b; Petritan et al. 2012, 2021). It may be argued that the beech in Cansiglio might endure larger N loads than in other habitats, and with a seemingly lower susceptibility than the oak in Monticolo. It is important to note here as well the age difference between the two species, where the mature beech is at ~ 135 years while the oaks are at the verge of maturity at ~ 65, which might contribute to an overall increased resilience towards environmental stresses, and a possibly higher resistance of the beech trees to N overloads in particular.
Contrary to the prevailing findings, where ring width and density alter in parallel under a changing environment, this was not the case in our oaks. Alterations in transport vessel size or area do not always manifest in conjunction with changes in ring width and may be affected by different factors to different extents (Fonti and García-González 2008). A similar lack of accord between external radial expansion and internal wood anatomy variations, was presented in two of the few existing ACN vs. BCN experiments (performed in broadleaf dominated mixed Chinese forests; Jiang et al. 2018; Zhang et al. 2022), emphasizing that wood related phenological shifts may develop beneath the surface, here in the context of varying N availabilities. Stressing yet again the importance of including measures such as wood density when investigating C sequestration. Indeed, further intrinsic wood traits may undergo morphological changes, where inner vessels may contract or expand (Spannl et al. 2016), along with changes in fibrous tissue, while on the macro scale total ring size may not budge. Thus, the differentiation of tree rings to the early and late wood fractions is one step forward in uncovering these phenomena, yet farther discrimination may be required for a deeper grasp of the trees’ actual state (Carrer et al. 2015; García-González et al. 2016).
Moreover, changes in wood density, especially when not accompanied with complimenting radial growth, may further take a substantial portion in the C stocks estimations, as suggested by our biomass area increment trends. When considering the density measure with BAI in BAIden in the ACN treatment of Q. petraea, albeit with a subtle effect, the mild upturn of the BAI trend (compared to the control and BCN), was lost, in accordance with the density reductions. Furthermore, even though density did not appear to be altered in F. sylvatica as consequence of any N treatment, the decrease that was visible in the BAI trend of both below canopy N treatments, was reduced with the inclusion of wood density when presenting BAIden. The effect of density here was as such, that there was a significant difference between the BAI and BAIden slopes of the BCN treatment, and nearly significant for BCN60. It is thus notable that wood functional traits, hereby tree rings and their physiology, are of major importance for both ecological and practical reasons, and considering the possible trade-offs between the ring traits, their significance in deeply understanding the trees’ productivity and C storage capacity with relation to its environment calls for a continuous and thorough investigation of as many measures as admissible (Ferretti et al. 2002; Chave et al. 2009; Njana et al. 2016; Giberti et al. 2023).
Conclusions
In the current study we utilized two N application approaches for simulating elevated N deposition in Q. petraea and F. sylvatica, the conventional ground N fertilization, and the more befitting above canopy N application which mimics closer the interaction of the tree with atmospheric deposition. The ring widths of both species were not conclusively affected by any of the treatments, yet when translating the ring widths to BAI, some patterns emerged where ACN appeared to have a positive effect while BCN had the opposite on the temporal trend. When looking at wood densities on the annual tree ring scale, Q. petraea was the more receptive between the two species, exhibiting decreases in mean density under the above canopy N application only, accentuating the established importance of the relationship between the tree canopy and atmospheric N deposition, and emphasizing the significance in choosing the right N application method for experimental research. The earlywood density fraction of Q. petraea underwent an additional decrease, while latewood was not affected, pointing to earlywood being the sensitive section of the ring towards N inputs. F. sylvatica did not show concrete changes in ring wood density, yet when evaluating biomass area increment (BAIden) through incorporating density in BAI, the previously observed negative effects of both below canopy N treatments on the BAI patterns were weakened.
Since our outcomes are based on a rather limited number of trees, and cores per tree, it would be encouraged to further substantiate the findings on a larger sample size. In addition, we determined significance of the N application on wood density of Q. petraea in middle years of the treatment period but not the last year. Considering past studies that observed reactions in wood traits to experimental treatments only some years down the monitoring periods, or rather responses in early years which diminished thereafter, a repeated analysis of our trees in upcoming years might be advised, as the experiment persists. Finally, in our field experiment, N was the singular factor differentiated in the system. Despite the advantages in focusing on one particular aspect of the environment for a deeper grasp of its role, it is clear that the interaction with other factors is substantial, thus further investigations including additional elements such as climate variables and age-related effects may be valuable.
With our current demonstration of the species-specific reactions to potential anticipated scenarios of increased N deposition rates, we aimed to contribute to a better understanding of possible consequences of increased N availability on the growth and wood formation in Q. petraea and F. sylvatica, whilst presenting the unique responses to different N application methodologies and utilizing restrained fertilizer dosages to encourage careful experimental designs with a long-term aptitude in similar prospective studies.
References
Ackerman D, Millet DB, Chen X (2019) Global estimates of inorganic nitrogen deposition across four decades. Global Biogeochem Cycles 33(1):100–107. https://doi.org/10.1029/2018GB005990
Arend M, Kuster T, Günthardt-Goerg MS, Dobbertin M (2011) Provenance-specific growth responses to drought and air warming in three European oak species (Quercus robur, Q. petraea and Q pubescens). Tree Physiol 31(3):287–297. https://doi.org/10.1093/treephys/tpr004
Baker TR, Phillips OL, Malhi Y, Almeida S, Arroyo L, Di Fiore A, Erwin T, Killeen TJ, Laurance SG, Laurance WF, Lewis SL, Lloyd J, Monteagudo A, Neill DA, Patiño S, Pitman NCA, M. Silva JN, Vásquez Martínez R, (2004) Variation in wood density determines spatial patterns inAmazonian forest biomass. Glob Change Biol 10(5):545–562. https://doi.org/10.1111/j.1365-2486.2004.00751.x
Barbaroux C, Bréda N (2002) Contrasting distribution and seasonal dynamics of carbohydrate reserves in stem wood of adult ring-porous sessile oak and diffuse-porous beech trees. Tree Physiol 22(17):1201–1210. https://doi.org/10.1093/treephys/22.17.1201
Baumbach L, Niamir A, Hickler T, Yousefpour R (2019) Regional adaptation of European beech (Fagus sylvatica) to drought in Central European conditions considering environmental suitability and economic implications. Reg Environ Change 19(4):1159–1174. https://doi.org/10.1007/s10113-019-01472-0
Bazot S, Barthes L, Blanot D, Fresneau C (2013) Distribution of non-structural nitrogen and carbohydrate compounds in mature oak trees in a temperate forest at four key phenological stages. Trees 27(4):1023–1034. https://doi.org/10.1007/s00468-013-0853-5
Bebber DP (2021) The gap between atmospheric nitrogen deposition experiments and reality. Sci Total Environ 801:149774. https://doi.org/10.1016/j.scitotenv.2021.149774
Becker M, Lévy G, Lefèvre Y (1996) Radial growth of mature pedunculate and sessile oaks in response to drainage, fertilization and weeding on acid pseudogley soils. Ann for Sci 53(2–3):585–594. https://doi.org/10.1051/forest:19960237
Beets PN, Gilchrist K, Jeffreys MP (2001) Wood density of radiata pine: effect of nitrogen supply. For Ecol Manage 145(3):173–180. https://doi.org/10.1016/S0378-1127(00)00405-9
Bergès L, Dupouey J-L, Franc A (2000) Long-term changes in wood density and radial growth of Quercus petraea Liebl. in northern France since the middle of the nineteenth century. Trees 14(7):398–408. https://doi.org/10.1007/s004680000055
Bergès L, Nepveu G, Franc A (2008) Effects of ecological factors on radial growth and wood density components of sessile oak (Quercus petraea Liebl.) in Northern France. Forest Ecol Manage 255(3):567–579. https://doi.org/10.1016/j.foreco.2007.09.027
Bigler C, Gričar J, Bugmann H, Čufar K (2004) Growth patterns as indicators of impending tree death in silver fir. For Ecol Manage 199(2):183–190. https://doi.org/10.1016/j.foreco.2004.04.019
Bobbink R, Hicks K, Galloway J, Spranger T, Alkemade R, Ashmore M, Bustamante M, Cinderby S, Davidson E, Dentener F, Emmett B, Erisman J-W, Fenn M, Gilliam F, Nordin A, Pardo L, De Vries W (2010) Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecol Appl 20(1):30–59. https://doi.org/10.1890/08-1140.1
Borghetti M, Gentilesca T, Leonardi S, van Noije T, Rita A, Mencuccini M (2017) Long-term temporal relationships between environmental conditions and xylem functional traits: a meta-analysis across a range of woody species along climatic and nitrogen deposition gradients. Tree Physiol 37(1):4–17. https://doi.org/10.1093/treephys/tpw087
Bortolazzi A, Da Ros L, Rodeghiero M, Tognetti R, Tonon G, Ventura M (2021) The canopy layer, a biogeochemical actor in the forest N-cycle. Sci Total Environ 776:146024. https://doi.org/10.1016/j.scitotenv.2021.146024
Bouriaud O, Bréda N, Le Moguédec G, Nepveu G (2004) Modelling variability of wood density in beech as affected by ring age, radial growth and climate. Trees 18(3):264–276. https://doi.org/10.1007/s00468-003-0303-x
Bouriaud O, Bréda N, Dupouey J-L, Granier A (2005) Is ring width a reliable proxy for stem-biomass increment? A case study in European beech. Can J for Res 35(12):2920–2933. https://doi.org/10.1139/x05-202
Braun S, Rihm B, Schindler C, Flückiger W (1999) Growth of mature beech in relation to ozone and nitrogen deposition: an epidemiological approach. Water Air Soil Pollut 116(1):357–364. https://doi.org/10.1023/A:1005209831728
Braun S, Thomas VFD, Quiring R, Flückiger W (2010) Does nitrogen deposition increase forest production? Role Phosphorus Environ Pollut 158(6):2043–2052. https://doi.org/10.1016/j.envpol.2009.11.030
Braun S, Schindler C, Rihm B (2017) Growth trends of beech and Norway spruce in Switzerland: the role of nitrogen deposition, ozone, mineral nutrition and climate. Sci Total Environ 599–600:637–646. https://doi.org/10.1016/j.scitotenv.2017.04.230
Bryan Dail D, Hollinger DY, Davidson EA, Fernandez I, Sievering HC, Scott NA, Gaige E (2009) Distribution of nitrogen-15 tracers applied to the canopy of a mature spruce-hemlock stand, Howland, Maine, USA. Oecologia 160(3):589–599. https://doi.org/10.1007/s00442-009-1325-x
Bucci SJ, Scholz FG, Goldstein G, Meinzer FC, Franco AC, Campanello PI, Villalobos-Vega R, Bustamante M, Miralles-Wilhelm F (2006) Nutrient availability constrains the hydraulic architecture and water relations of savannah trees. Plant, Cell Environ 29(12):2153–2167. https://doi.org/10.1111/j.1365-3040.2006.01591.x
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26(2):115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Cailleret M, Jansen S, Robert EMR, Desoto L, Aakala T, Antos JA, Beikircher B, Bigler C, Bugmann H, Caccianiga M, Čada V, Camarero JJ, Cherubini P, Cochard H, Coyea MR, Čufar K, Das AJ, Davi H, Delzon S, Dorman M, Gea-Izquierdo G, Gillner S, Haavik LJ, Hartmann H, Hereş A-M, Hultine KR, Janda P, Kane JM, Kharuk VI, Kitzberger T, Klein T, Kramer K, Lens F, Levanic T, Linares Calderon JC, Lloret F, Lobo-Do-Vale R, Lombardi F, López Rodríguez R, Mäkinen H, Mayr S, Mészáros I, Metsaranta JM, Minunno F, Oberhuber W, Papadopoulos A, Peltoniemi M, Petritan AM, Rohner B, Sangüesa-Barreda G, Sarris D, Smith JM, Stan AB, Sterck F, Stojanović DB, Suarez ML, Svoboda M, Tognetti R, Torres-Ruiz JM, Trotsiuk V, Villalba R, Vodde F, Westwood AR, Wyckoff PH, Zafirov N, Martínez-Vilalta J (2017) A synthesis of radial growth patterns preceding tree mortality. Glob Change Biol 23(4):1675–1690. https://doi.org/10.1111/gcb.13535
Campioli M, Vincke C, Jonard M, Kint V, Demarée G, Ponette Q (2012) Current status and predicted impact of climate change on forest production and biogeochemistry in the temperate oceanic European zone: review and prospects for Belgium as a case study. J for Res 17(1):1–18. https://doi.org/10.1007/s10310-011-0255-8
Carrer M, von Arx G, Castagneri D, Petit G (2015) Distilling allometric and environmental information from time series of conduit size: the standardization issue and its relationship to tree hydraulic architecture. Tree Physiol 35(1):27–33. https://doi.org/10.1093/treephys/tpu108
Carter TS, Clark CM, Fenn ME, Jovan S, Perakis SS, Riddell J, Schaberg PG, Greaver TL, Hastings MG (2017) Mechanisms of nitrogen deposition effects on temperate forest lichens and trees. Ecosphere 8(3):e01717. https://doi.org/10.1002/ecs2.1717
Cedro A (2007) Tree-ring chronologies of downy oak (Quercus Pubescens), pedunculate oak (Q. Robur) and Sessile Oak (Q. Petraea) in the bielinek nature reserve: comparison of the climatic determinants of tree-ring width. Geochronometria 26:39–45. https://doi.org/10.2478/v10003-007-0005-2
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12(4):351–366. https://doi.org/10.1111/j.1461-0248.2009.01285.x
Cherubini P, Battipaglia G, Innes JL (2021) Tree vitality and forest health: can tree-ring stable isotopes be used as indicators? Curr Forestry Rep 7(2):69–80. https://doi.org/10.1007/s40725-021-00137-8
Da Ros L, Rodeghiero M, Goodale CL, Trafoier G, Panzacchi P, Giammarchi F, Tonon G, Ventura M (2023) Canopy 15N fertilization increases short-term plant N retention compared to ground fertilization in an oak forest. For Ecol Manage 539:121001. https://doi.org/10.1016/j.foreco.2023.121001
Dao MCE, Rossi S, Walsh D, Morin H, Houle D (2015) A 6-Year-Long Manipulation with Soil Warming and Canopy Nitrogen Additions does not Affect Xylem Phenology and Cell Production of Mature Black Spruce. Frontiers in Plant Science 6. https://doi.org/10.3389/fpls.2015.00877
de Vries W, Du E, Butterbach-Bahl K (2014) Short and long-term impacts of nitrogen deposition on carbon sequestration by forest ecosystems. Curr Opinion Environ Sustain 9–10:90–104. https://doi.org/10.1016/j.cosust.2014.09.001
Delpierre N, Berveiller D, Granda E, Dufrêne E (2016) Wood phenology, not carbon input, controls the interannual variability of wood growth in a temperate oak forest. New Phytol 210(2):459–470. https://doi.org/10.1111/nph.13771
Durand M, Rose C, Dupouey J-L, Legout A, Ponton S (2020) Do tree rings record changes in soil fertility? Results from a Quercus petraea fertilization trial. Sci Total Environ 712:136148. https://doi.org/10.1016/j.scitotenv.2019.136148
El Zein R, Bréda N, Gérant D, Zeller B, Maillard P (2011a) Nitrogen sources for current-year shoot growth in 50-year-old sessile oak trees: an in situ 15N labeling approach. Tree Physiol 31(12):1390–1400. https://doi.org/10.1093/treephys/tpr118
El Zein R, Maillard P, Bréda N, Marchand J, Montpied P, Gérant D (2011b) Seasonal changes of C and N non-structural compounds in the stem sapwood of adult sessile oak and beech trees. Tree Physiol 31(8):843–854. https://doi.org/10.1093/treephys/tpr074
Elhani S, Guehl J-M, Nys C, Picard J-F, Dupouey J-L (2005) Impact of fertilization on tree-ring δ15N and δ13C in beech stands: a retrospective analysis. Tree Physiol 25(11):1437–1446. https://doi.org/10.1093/treephys/25.11.1437
Etzold S, Ferretti M, Reinds GJ, Solberg S, Gessler A, Waldner P, Schaub M, Simpson D, Benham S, Hansen K, Ingerslev M, Jonard M, Karlsson PE, Lindroos A-J, Marchetto A, Manninger M, Meesenburg H, Merilä P, Nöjd P, Rautio P, Sanders TGM, Seidling W, Skudnik M, Thimonier A, Verstraeten A, Vesterdal L, Vejpustkova M, de Vries W (2020) Nitrogen deposition is the most important environmental driver of growth of pure, even-aged and managed European forests. For Ecol Manage 458:117762. https://doi.org/10.1016/j.foreco.2019.117762
Fenn ME, Ross CS, Schilling SL, Baccus WD, Larrabee MA, Lofgren RA (2013) Atmospheric deposition of nitrogen and sulfur and preferential canopy consumption of nitrate in forests of the Pacific Northwest, USA. For Ecol Manage 302:240–253. https://doi.org/10.1016/j.foreco.2013.03.042
Ferretti M, Innes JL, Jalkanen R, Saurer M, Schäffer J, Spiecker H, von Wilpert K (2002) Air pollution and environmental chemistry – what role for tree-ring studies? Dendrochronologia 20(1):159–174. https://doi.org/10.1078/1125-7865-00014
Feuillat F, Dupouey J-L, Sciama D, Keller R (1997) A new attempt at discrimination between Quercus petraea and Quercus robur based on wood anatomy. Can J for Res 27(3):343–351. https://doi.org/10.1139/x96-174
Flechard CR, Ibrom A, Skiba UM, de Vries W, van Oijen M, Cameron DR, Dise NB, Korhonen JFJ, Buchmann N, Legout A, Simpson D, Sanz MJ, Aubinet M, Loustau D, Montagnani L, Neirynck J, Janssens IA, Pihlatie M, Kiese R, Siemens J, Francez A-J, Augustin J, Varlagin A, Olejnik J, Juszczak R, Aurela M, Berveiller D, Chojnicki BH, Dämmgen U, Delpierre N, Djuricic V, Drewer J, Dufrêne E, Eugster W, Fauvel Y, Fowler D, Frumau A, Granier A, Gross P, Hamon Y, Helfter C, Hensen A, Horváth L, Kitzler B, Kruijt B, Kutsch WL, Lobo-do-Vale R, Lohila A, Longdoz B, Marek MV, Matteucci G, Mitosinkova M, Moreaux V, Neftel A, Ourcival J-M, Pilegaard K, Pita G, Sanz F, Schjoerring JK, Sebastià M-T, Tang YS, Uggerud H, Urbaniak M, van Dijk N, Vesala T, Vidic S, Vincke C, Weidinger T, Zechmeister-Boltenstern S, Butterbach-Bahl K, Nemitz E, Sutton MA (2020a) Carbon–nitrogen interactions in European forests and semi-natural vegetation – Part 1: Fluxes and budgets of carbon, nitrogen and greenhouse gases from ecosystem monitoring and modelling. Biogeosciences 17(6):1583–1620. https://doi.org/10.5194/bg-17-1583-2020
Flechard CR, van Oijen M, Cameron DR, de Vries W, Ibrom A, Buchmann N, Dise NB, Janssens IA, Neirynck J, Montagnani L, Varlagin A, Loustau D, Legout A, Ziemblińska K, Aubinet M, Aurela M, Chojnicki BH, Drewer J, Eugster W, Francez A-J, Juszczak R, Kitzler B, Kutsch WL, Lohila A, Longdoz B, Matteucci G, Moreaux V, Neftel A, Olejnik J, Sanz MJ, Siemens J, Vesala T, Vincke C, Nemitz E, Zechmeister-Boltenstern S, Butterbach-Bahl K, Skiba UM, Sutton MA (2020b) Carbon–nitrogen interactions in European forests and semi-natural vegetation – Part 2: untangling climatic, edaphic, management and nitrogen deposition effects on carbon sequestration potentials. Biogeosciences 17(6):1621–1654. https://doi.org/10.5194/bg-17-1621-2020
Fleischer K, Rebel KT, van der Molen MK, Erisman JW, Wassen MJ, van Loon EE, Montagnani L, Gough CM, Herbst M, Janssens IA, Gianelle D, Dolman AJ (2013) The contribution of nitrogen deposition to the photosynthetic capacity of forests. Global Biogeochem Cycles 27(1):187–199. https://doi.org/10.1002/gbc.20026
Fonti P, García-González I (2004) Suitability of chestnut earlywood vessel chronologies for ecological studies. New Phytol 163(1):77–86. https://doi.org/10.1111/j.1469-8137.2004.01089.x
Fonti P, García-González I (2008) Earlywood vessel size of oak as a potential proxy for spring precipitation in mesic sites. J Biogeogr 35(12):2249–2257. https://doi.org/10.1111/j.1365-2699.2008.01961.x
Fonti P, Solomonoff N, García-González I (2007) Earlywood vessels of Castanea sativa record temperature before their formation. New Phytol 173(3):562–570. https://doi.org/10.1111/j.1469-8137.2006.01945.x
Friedrichs DA, Büntgen U, Frank DC, Esper J, Neuwirth B, Löffler J (2009) Complex climate controls on 20th century oak growth in Central-West Germany. Tree Physiol 29(1):39–51. https://doi.org/10.1093/treephys/tpn003
Gagen M, Matthews N, Denman S, Bridge M, Peace A, Pike R, Young G (2019) The tree ring growth histories of UK native oaks as a tool for investigating chronic oak decline: an example from the Forest of Dean. Dendrochronologia 55:50–59. https://doi.org/10.1016/j.dendro.2019.03.001
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science. https://doi.org/10.1126/science.1136674
García-González I, Souto-Herrero M, Campelo F (2016) Ring porosity and earlywood vessels: a review on extracting environmental information through time. IAWA J/int Assoc Wood Anatom 37:295–314. https://doi.org/10.1163/22941932-20160135
George SS, Ault TR (2014) The imprint of climate within Northern Hemisphere trees. Quaternary Sci Rev 89:1–4. https://doi.org/10.1016/j.quascirev.2014.01.007
Giammarchi F, Panzacchi P, Ventura M, Tonon G (2019) Tree growth and water-use efficiency do not react in the short term to artificially increased nitrogen deposition. Forests 11(1):47. https://doi.org/10.3390/f11010047
Giberti GS, Wellstein C, Giovannelli A, Bielak K, Uhl E, Aguirre-Ráquira W, Giammarchi F, Tonon G (2022) Annual carbon sequestration patterns in trees: a case study from scots pine monospecific stands and mixed stands with sessile oak in Central Poland. Forests 13(4):582. https://doi.org/10.3390/f13040582
Giberti GS, von Arx G, Giovannelli A, du Toit B, Unterholzner L, Bielak K, Carrer M, Uhl E, Bravo F, Tonon G, Wellstein C (2023) The admixture of Quercus sp. in Pinus sylvestris stands influences wood anatomical trait responses to climatic variability and drought events. Front Plant Sci. https://doi.org/10.3389/fpls.2023.1213814
Gilson A, Barthes L, Delpierre N, Dufrêne É, Fresneau C, Bazot S (2014) Seasonal changes in carbon and nitrogen compound concentrations in a Quercus petraea chronosequence. Tree Physiol 34(7):716–729. https://doi.org/10.1093/treephys/tpu060
González IG, Eckstein D (2003) Climatic signal of earlywood vessels of oak on a maritime site. Tree Physiol 23(7):497–504. https://doi.org/10.1093/treephys/23.7.497
González-González BD, Rozas V, García-González I (2014) Earlywood vessels of the sub-Mediterranean oak Quercus pyrenaica have greater plasticity and sensitivity than those of the temperate Q. petraea at the Atlantic-Mediterranean boundary. Trees 28(1):237–252. https://doi.org/10.1007/s00468-013-0945-2
Gričar J, Čufar K (2008) Seasonal dynamics of phloem and xylem formation in silver fir and Norway spruce as affected by drought. Russ J Plant Physiol 55(4):538–543. https://doi.org/10.1134/S102144370804016X
Guan L-L, Wen D-Z (2011) More nitrogen partition in structural proteins and decreased photosynthetic nitrogen-use efficiency of Pinus massoniana under in situ polluted stress. J Plant Res 124(6):663–673. https://doi.org/10.1007/s10265-011-0405-2
Guerrieri R, Correia M, Martín-Forés I, Alfaro-Sánchez R, Pino J, Hampe A, Valladares F, Espelta JM (2021a) Land-use legacies influence tree water-use efficiency and nitrogen availability in recently established European forests. Funct Ecol 35(6):1325–1340. https://doi.org/10.1111/1365-2435.13787
Guerrieri R, Templer P, Magnani F (2021b) Canopy exchange and modification of nitrogen fluxes in forest ecosystems. Curr Forestry Rep 7(3):115–137. https://doi.org/10.1007/s40725-021-00141-y
Guo X, Huang J-G, Li J, Liang H, Yu B, Ma Q, Jiang S, Lu X, Fu S, Ye Q, Zhao P, Cai X (2020) Nitrogen addition to the canopy of Castanopsis chinensis (Sprengel) Hance promoted xylem formation in a subtropical forest in China. Ann for Sci 77(2):1–11. https://doi.org/10.1007/s13595-020-00962-0
Hacke UG, Sperry JS, Pockman WT, Davis SD, McCulloh KA (2001) Trends in wood density and structure are linked to prevention of xylem implosion by negative pressure. Oecologia 126(4):457–461. https://doi.org/10.1007/s004420100628
Hacke UG, Sperry JS, Pittermann J (2005) 16 - Efficiency Versus Safety Tradeoffs for Water Conduction in Angiosperm Vessels Versus Gymnosperm Tracheids. In: Holbrook NM, Zwieniecki MA (eds) Vascular Transport in Plants. Academic Press, Burlington, pp 333–353
Hacke UG, Plavcová L, Almeida-Rodriguez A, King-Jones S, Zhou W, Cooke JEK (2010) Influence of nitrogen fertilization on xylem traits and aquaporin expression in stems of hybrid poplar. Tree Physiol 30(8):1016–1025. https://doi.org/10.1093/treephys/tpq058
Härdtle W, Niemeyer T, Assmann T, Aulinger A, Fichtner A, Lang A, Leuschner C, Neuwirth B, Pfister L, Quante M, Ries C, Schuldt A, von Oheimb G (2013) Climatic responses of tree-ring width and δ13C signatures of sessile oak (Quercus petraea Liebl.) on soils with contrasting water supply. Plant Ecol 214(9):1147–1156. https://doi.org/10.1007/s11258-013-0239-1
Hättenschwiler S, Schweingruber FH, Körner Ch (1996) Tree ring responses to elevated CO2 and increased N deposition in Picea abies. Plant, Cell Environ 19(12):1369–1378. https://doi.org/10.1111/j.1365-3040.1996.tb00015.x
Helama S, Läänelaid A, Raisio J, Tuomenvirta H (2009) Oak decline in Helsinki portrayed by tree-rings, climate and soil data. Plant Soil 319(1):163–174. https://doi.org/10.1007/s11104-008-9858-z
Helm J, Muhr J, Hilman B, Kahmen A, Schulze ED, Trumbore S, Hartmann H (2023) Carbon dynamics in long-term starving poplar trees—the importance of older carbohydrates and a shift to lipids during survival. Tree Physiol 1:13. https://doi.org/10.1093/treephys/tpad135
Hess C, Niemeyer T, Fichtner A, Jansen K, Kunz M, Maneke M, von Wehrden H, Quante M, Walmsley D, von Oheimb G, Härdtle W (2018) Anthropogenic nitrogen deposition alters growth responses of European beech (Fagus sylvativa L.) to climate change. Environ Pollut 233:92–98. https://doi.org/10.1016/j.envpol.2017.10.024
Högberg P (2007) Nitrogen impacts on forest carbon. Nature 447(7146):781–782. https://doi.org/10.1038/447781a
Houle D, Marty C, Duchesne L (2015) Response of canopy nitrogen uptake to a rapid decrease in bulk nitrate deposition in two eastern Canadian boreal forests. Oecologia 177(1):29–37. https://doi.org/10.1007/s00442-014-3118-0
Janssens IA, Dieleman W, Luyssaert S, Subke J-A, Reichstein M, Ceulemans R, Ciais P, Dolman AJ, Grace J, Matteucci G, Papale D, Piao SL, Schulze E-D, Tang J, Law BE (2010) Reduction of forest soil respiration in response to nitrogen deposition. Nature Geosci 3(5):315–322. https://doi.org/10.1038/ngeo844
Jennings KA, Guerrieri R, Vadeboncoeur MA, Asbjornsen H (2016) Response of Quercus velutina growth and water use efficiency to climate variability and nitrogen fertilization in a temperate deciduous forest in the northeastern USA. Tree Physiol 36(4):428–443. https://doi.org/10.1093/treephys/tpw003
Jiang Y, Zang R, Lu X, Huang Y, Ding Y, Liu W, Long W, Zhang J, Zhang Z (2015) Effects of soil and microclimatic conditions on the community-level plant functional traits across different tropical forest types. Plant Soil 390(1):351–367. https://doi.org/10.1007/s11104-015-2411-y
Khaine I, Woo SY (2015) An overview of interrelationship between climate change and forests. For Sci Technol 11(1):11–18. https://doi.org/10.1080/21580103.2014.932718
Kimak A, Leuenberger M (2015) Are carbohydrate storage strategies of trees traceable by early–latewood carbon isotope differences? Trees 29(3):859–870. https://doi.org/10.1007/s00468-015-1167-6
Kint V, Aertsen W, Campioli M, Vansteenkiste D, Delcloo A, Muys B (2012) Radial growth change of temperate tree species in response to altered regional climate and air quality in the period 1901–2008. Clim Change 115(2):343–363. https://doi.org/10.1007/s10584-012-0465-x
Kitin P, Funada R (2016) Earlywood vessels in ring-porous trees become functional for water transport after bud burst and before the maturation of the current-year leaves. Iawa J 37(2):315–331. https://doi.org/10.1163/22941932-20160136
Klopatek J, Barry M, Johnson D (2006) Potential canopy interception of nitrogen in the Pacific Northwest, USA. For Ecol Manage 234:344–354. https://doi.org/10.1016/j.foreco.2006.07.019
Kostiainen K, Kaakinen S, Saranpää P, Sigurdsson BD, Linder S, Vapaavuori E (2004) Effect of elevated [CO2] on stem wood properties of mature Norway spruce grown at different soil nutrient availability. Glob Change Biol 10(9):1526–1538. https://doi.org/10.1111/j.1365-2486.2004.00821.x
Koutsianitis D, Barboutis I (2017) Application of High frequency densitometry to determine wood density and ring width of beech trees. Pro Ligno 13(4):78–83
Laubhann D, Sterba H, Reinds GJ, De Vries W (2009) The impact of atmospheric deposition and climate on forest growth in European monitoring plots: An individual tree growth model. For Ecol Manage 258(8):1751–1761. https://doi.org/10.1016/j.foreco.2008.09.050
Lebourgeois F, Cousseau G, Ducos Y (2013) Climate-tree-growth relationships of Quercus petraea Mill. stand in the Forest of Bercé (“Futaie des Clos”, Sarthe, France). Ann for Sci 61:361–372. https://doi.org/10.1051/forest:2004029
Lenth RV, Bolker B, Buerkner P, Giné-Vázquez I, Herve M, Jung M, Love J, Miguez F, Riebl H, Singmann H (2023) emmeans: Estimated Marginal Means, aka Least-Squares Means.
Leuschner C, Backes K, Hertel D, Schipka F, Schmitt U, Terborg O, Runge M (2001a) Drought responses at leaf, stem and fine root levels of competitive Fagus sylvatica L. and Quercus petraea (Matt.) Liebl trees in dry and wet years. Forest Ecol Manage 149(1):33–46. https://doi.org/10.1016/S0378-1127(00)00543-0
Leuschner C, Hertel D, Coners H, Büttner V (2001b) Root competition between beech and oak: a hypothesis. Oecologia 126(2):276–284. https://doi.org/10.1007/s004420000507
Liang X, Ye Q, Liu H, Brodribb TJ (2021) Wood density predicts mortality threshold for diverse trees. New Phytol 229(6):3053–3057. https://doi.org/10.1111/nph.17117
Lovett GM, Lindberg SE (2011) Atmospheric deposition and canopy interactions of nitrogen in forests. Can J for Res. https://doi.org/10.1139/x93-200
Lupi C, Morin H, Deslauriers A, Rossi S, Houle D (2012) Increasing nitrogen availability and soil temperature: effects on xylem phenology and anatomy of mature black spruce. Canadian J for Res 42(7):1277–1288. https://doi.org/10.1139/x2012-055
Mérian P, Bontemps JD, Bergès L, Lebourgeois F (2011) Spatial variation and temporal instability in climate-growth relationships of sessile oak (Quercus petraea [Matt.] Liebl.) under temperate conditions. Plant Ecol 212(11):1855–1871. https://doi.org/10.1007/s11258-011-9959-2
Michelot A, Bréda N, Damesin C, Dufrêne E (2012a) Differing growth responses to climatic variations and soil water deficits of Fagus sylvatica, Quercus petraea and Pinus sylvestris in a temperate forest. For Ecol Manage 265:161–171. https://doi.org/10.1016/j.foreco.2011.10.024
Michelot A, Simard S, Rathgeber C, Dufrêne E, Damesin C (2012b) Comparing the intra-annual wood formation of three European species (Fagus sylvatica, Quercus petraea and Pinus sylvestris) as related to leaf phenology and non-structural carbohydrate dynamics. Tree Physiol 32(8):1033–1045. https://doi.org/10.1093/treephys/tps052
Mölder A, Meyer P, Nagel R-V (2019) Integrative management to sustain biodiversity and ecological continuity in Central European temperate oak (Quercus robur, Q. petraea) forests: an overview. For Ecol Manage 437:324–339. https://doi.org/10.1016/j.foreco.2019.01.006
Nair RKF, Perks MP, Weatherall A, Baggs EM, Mencuccini M (2016) Does canopy nitrogen uptake enhance carbon sequestration by trees? Glob Change Biol 22(2):875–888. https://doi.org/10.1111/gcb.13096
Njana MA, Meilby H, Eid T, Zahabu E, Malimbwi RE (2016) Importance of tree basic density in biomass estimation and associated uncertainties: a case of three mangrove species in Tanzania. Ann for Sci 73(4):1073–1087. https://doi.org/10.1007/s13595-016-0583-0
Pérez-de-Lis G, Rozas V, Vázquez-Ruiz RA, García-González I (2018) Do ring-porous oaks prioritize earlywood vessel efficiency over safety? Environmental effects on vessel diameter and tyloses formation. Agric for Meteorol 248:205–214. https://doi.org/10.1016/j.agrformet.2017.09.022
Petritan AM, Biris IA, Merce O, Turcu DO, Petritan IC (2012) Structure and diversity of a natural temperate sessile oak (Quercus petraea L.) – European Beech (Fagus sylvatica L.) forest. For Ecol Manage 280:140–149. https://doi.org/10.1016/j.foreco.2012.06.007
Petritan AM, Petritan IC, Hevia A, Walentowski H, Bouriaud O, Sánchez-Salguero R (2021) Climate warming predispose sessile oak forests to drought-induced tree mortality regardless of management legacies. For Ecol Manage 491:119097. https://doi.org/10.1016/j.foreco.2021.119097
Pinheiro JP, Bates D (2023) nlme: Linear and Nonlinear Mixed Effects Models.
Pivovaroff AL, Santiago LS, Vourlitis GL, Grantz DA, Allen MF (2016) Plant hydraulic responses to long-term dry season nitrogen deposition alter drought tolerance in a Mediterranean-type ecosystem. Oecologia 181(3):721–731. https://doi.org/10.1007/s00442-016-3609-2
Ponton S, Bornot Y, Bréda N (2019) Soil fertilization transiently increases radial growth in sessile oaks but does not change their resilience to severe soil water deficit. For Ecol Manage 432:923–931. https://doi.org/10.1016/j.foreco.2018.10.027
Poorter L, McDonald I, Alarcón A, Fichtler E, Licona J-C, Peña-Claros M, Sterck F, Villegas Z, Sass-Klaassen U (2010) The importance of wood traits and hydraulic conductance for the performance and life history strategies of 42 rainforest tree species. New Phytol 185(2):481–492. https://doi.org/10.1111/j.1469-8137.2009.03092.x
Pretzsch H, Biber P, Schütze G, Kemmerer J, Uhl E (2018) Wood density reduced while wood volume growth accelerated in Central European forests since 1870. For Ecol Manage 429:589–616. https://doi.org/10.1016/j.foreco.2018.07.045
Quinn Thomas R, Canham CD, Weathers KC, Goodale CL (2010) Increased tree carbon storage in response to nitrogen deposition in the US. Nature Geosci 3(1):13–17. https://doi.org/10.1038/ngeo721
R Core Team (2023) R: A language and environment for statistical computing.
Raymond CA, Muneri A (2000) Effect of fertilizer on wood properties of Eucalyptus globulus. Can J for Res 30(1):136–144. https://doi.org/10.1139/x99-186
Ross D, Buckner E, Core H, Woods F (1979) Nitrogen fertilization decreases yellow-poplar wood density. South J Appl for 3(3):119–122. https://doi.org/10.1093/sjaf/3.3.119
Scharnweber T, Manthey M, Criegee C, Bauwe A, Schröder C, Wilmking M (2011) Drought matters–Declining precipitation influences growth of Fagus sylvatica L. and Quercus robur L. in north-eastern Germany. For Ecol Manage 262(6):947–961. https://doi.org/10.1016/j.foreco.2011.05.026
Schinker MG, Hansen N, Spiecker H (2003) High-frequency densitometry-a new method for the rapid evaluation of wood density variations. IAWA J 24(3):231–239. https://doi.org/10.1163/22941932-90001592
Schmitz A, Sanders TGM, Bolte A, Bussotti F, Dirnböck T, Johnson J, Peñuelas J, Pollastrini M, Prescher A-K, Sardans J, Verstraeten A, de Vries W (2019) Responses of forest ecosystems in Europe to decreasing nitrogen deposition. Environ Pollut 244:980–994. https://doi.org/10.1016/j.envpol.2018.09.101
Shchupakivskyy R, Clauder L, Linke N, Pfriem A (2014) Anwendung der Hochfrequenz-Dichtemessung zur Erfassung der Dichteänderung im Eichenfrüh-und-spätholz (Quercus robur L.) infolge einer thermischen Modifikation. European J Wood Wood Prod 72:5–10. https://doi.org/10.1007/s00107-013-0744-x
Sheppard L, Crossley A, Parrington J, Harvey F, Cape JN (2001) Effects of simulated acid mist on a Sitka spruce forest approaching canopy closure: significance of acidified versus non-acidified nitrogen inputs. Water Air Soil Pollut 130:953–958. https://doi.org/10.1023/A:1013934417083
Sheppard LJ, Crossley A, Harvey FJ, Skiba U, Coward P, Ingleby K (2004) Effects of five years of frequent N additions, with or without acidity, on the growth and below-ground dynamics of a young Sitka spruce stand growing on an acid peat: Implications for sustainability. Hydrol Earth Syst Sci 8(3):377–391
Sievering H, Tomaszewski T, Torizzo J (2007) Canopy uptake of atmospheric N deposition at a conifer forest: Part I -canopy N budget, photosynthetic efficiency and net ecosystem exchange. Tellus B 59:483–492. https://doi.org/10.1111/j.1600-0889.2007.00264.x
Spannl S, Homeier J, Bräuning A (2016) Nutrient-induced modifications of wood anatomical traits of Alchornea lojaensis (Euphorbiaceae). Front Earth Sci 4:50. https://doi.org/10.3389/feart.2016.00050
Sparks JP (2009) Ecological ramifications of the direct foliar uptake of nitrogen. Oecologia 159(1):1–13. https://doi.org/10.1007/s00442-008-1188-6
Teglia A, Di Baccio D, Matteucci G, Scartazza A, De Cinti B, Mazzenga F, Ravaioli D, Muzzi E, Marcolini G, Magnani F (2022) Effects of simulated nitrogen deposition on the nutritional and physiological status of beech forests at two climatic contrasting sites in Italy. Sci Total Environ 834:155362. https://doi.org/10.1016/j.scitotenv.2022.155362
Tomaszewski T, Sievering H (2007) Canopy uptake of atmospheric N deposition at a conifer forest: Part II- response of chlorophyll fluorescence and gas exchange parameters. Tellus B: Chemical and Physical Meteorology 59(3):493–501. https://doi.org/10.1111/j.1600-0889.2007.00265.x
Trouvé R, Bontemps J-D, Collet C, Seynave I, Lebourgeois F (2017) Radial growth resilience of sessile oak after drought is affected by site water status, stand density, and social status. Trees 31(2):517–529. https://doi.org/10.1007/s00468-016-1479-1
Umebayashi T, Utsumi Y, Koga S, Inoue S, Fujikawa S, Arakawa K, Matsumura J, Oda K (2008) Conducting pathways in north temperate deciduous broadleaved trees. IAWA J 29(3):247–263. https://doi.org/10.1163/22941932-90000184
Van Den Berge S, Vangansbeke P, Baeten L, Vanhellemont M, Vanneste T, De Mil T, Van den Bulcke J, Verheyen K (2021) Biomass increment and carbon sequestration in hedgerow-grown trees. Dendrochronologia 70:125894. https://doi.org/10.1016/j.dendro.2021.125894
Vannoppen A, Boeckx P, De Mil T, Kint V, Ponette Q, Van den Bulcke J, Verheyen K, Muys B (2018) Climate driven trends in tree biomass increment show asynchronous dependence on tree-ring width and wood density variation. Dendrochronologia 48:40–51. https://doi.org/10.1016/j.dendro.2018.02.001
Vavrcik H, Gryc V (2012) Analysis of the annual ring structure and wood density relations in english oakand sessile oak. Wood Res 57:573–580
Ward EJ, Oren R, Bell DM, Clark JS, McCarthy HR, Kim H-S, Domec J-C (2013) The effects of elevated CO2 and nitrogen fertilization on stomatal conductance estimated from 11 years of scaled sap flux measurements at Duke FACE. Tree Physiol 33(2):135–151. https://doi.org/10.1093/treephys/tps118
Wortman E, Tomaszewski T, Waldner P, Schleppi P, Thimonier A, Eugster W, Buchmann N, Sievering H (2012) Atmospheric nitrogen deposition and canopy retention influences on photosynthetic performance at two high nitrogen deposition Swiss forests. Tellus B: Chem Phys Meteorol 64(1):17216. https://doi.org/10.3402/tellusb.v64i0.17216
Wright RF, Roelofs JGM, Bredemeier M, Blanck K, Boxman AW, Emmett BA, Gundersen P, Hultberg H, Kjønaas OJ, Moldan F, Tietema A, van Breemen N, van Dijk HFG (1995) NITREX: responses of coniferous forest ecosystems to experimentally changed deposition of nitrogen. For Ecol Manage 71(1):163–169. https://doi.org/10.1016/0378-1127(94)06093-X
Yu B, Huang J-G, Ma Q, Guo X, Liang H, Zhang S, Fu S, Wan S, Yan J, Zhang W (2019) Comparison of the effects of canopy and understory nitrogen addition on xylem growth of two dominant species in a warm temperate forest. China Dendrochronologia 56:125604. https://doi.org/10.1016/j.dendro.2019.125604
Zanne AE, Westoby M, Falster DS, Ackerly DD, Loarie SR, Arnold SEJ, Coomes DA (2010) Angiosperm wood structure: global patterns in vessel anatomy and their relation to wood density and potential conductivity. Am J Bot 97(2):207–215. https://doi.org/10.3732/ajb.0900178
Zhang S-Y, Owoundi RE, Nepveu G, Mothe F, Dhôte J-F (1993) Modelling wood density in European oak (Quercuspetraea and Quercusrobur) and simulating the silvicultural influence. Can J for Res 23(12):2587–2593. https://doi.org/10.1139/x93-320
Zhang W, Shen W, Zhu S, Wan S, Luo Y, Yan J, Wang K, Liu L, Dai H, Li P, Dai K, Zhang W, Liu Z, Wang F, Kuang Y, Li Z, Lin Y, Rao X, Li J, Zou B, Cai X, Mo J, Zhao P, Ye Q, Huang J, Fu S (2015) CAN Canopy addition of nitrogen better illustrate the effect of atmospheric nitrogen deposition on forest ecosystem? Sci Rep 5(1):11245. https://doi.org/10.1038/srep11245
Zhang S, Yu B, Zhou P, Huang J, Fu S, Zhang W (2022) Different-sized vessels of quercus variabilis blume respond diversely to six-year canopy and understory n addition in a warm-temperate transitional zone. Forests 13(7):1075. https://doi.org/10.3390/f13071075
Acknowledgements
We thank the Bolzano Province Forest Service for facilitating the study site in the Monticolo Forest. We thank Prof. Federico Magnani and Dr. Rossella Guerrieri from the University of Bologna for facilitating the study site in the Cansiglio Forest. A special gratitude to Ms. Monika Bradatsch for her invaluable assistance in wood core analysis. We thank the ETN Skill-For.Action network and all its’ partners for providing a unique professional support system. Finally, we thank all members of the research team at the Analysis and Management of Mountain Ecosystems department, Faculty of Agricultural, Environmental and Food Sciences of the Free University of Bozen-Bolzano, who have been assisting in maintaining the experimental plots in the Monticolo site.
Funding
Open access funding provided by Libera Università di Bolzano within the CRUI-CARE Agreement. The study is supported by the ETN Skill-For.Action project that has received funding from the European Union’s HORIZON 2020 research and innovation programme under the Marie Skłodowska Curie grant agreement No. 936355.
Author information
Authors and Affiliations
Contributions
Conceptualization: G.T., M.V. and C.W.; Methodology: G.T., M.V. and C.W.; Investigation: D.M; Data curation and formal analysis: DM; Sample collection and analysis D.M. and D.R.; Supplementary data processing: D.M. and P.J.; Writing – original draft: D.M.; Review, and editing: D.M., C.W. and E.U.; Supervision: C.W. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The Authors declare no conflicts of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Miren del Río.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Minikaev, D., Ventura, M., Tonon, G. et al. Canopy nitrogen application effects on Quercus petraea L. and Fagus sylvatica L. ring width and wood density. Eur J Forest Res (2024). https://doi.org/10.1007/s10342-024-01693-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10342-024-01693-x