Abstract
Water strider (Gerridae) morphology and behavior have become the focus of interdisciplinary research in biological diversification and bio-inspired technology. However, the diversity of behaviors and morphology of the large-sized Gerridae have not been intensely studied. Here, we provide locomotory behaviors and legs’ micro-morphology of the large South-East Asian water strider, Ptilomera tigrina. Using high-speed videography and experiments in natural habitats, as well as scanning electron microscopy of midlegs, we have determined that (1) P. tigrina individuals prefer relatively high flow speeds of 0.15–0.30 m/s, compared to other water striders previously studied, and they are also observed in very high flow speeds of up to 0.6 m/s; (2) they avoid stagnant water, but when on still and very slow flowing water they perform constant back-and-forth rowing using their midlegs; (3) their antipredatory reaction involves repetitive and very fast “protean” movements propelled by the midlegs; (4) their midleg tarsi and tibiae are equipped with brushes of ribbon-like hairs, which are used as paddles for rowing. As the locomotory behaviors and flow-speed preferences by P. tigrina require constant use of midlegs for rowing, the presence of special paddle structures on midlegs illustrates a hypothetical adaptive match between midlegs’ locomotory function and their micro-morphology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Semiaquatic bugs (Gerromorpha), including water striders (Gerridae), inhabit different semiaquatic niches (Carpenter 1928; Andersen 1979, 1982, 1995; Spence and Andersen 1994; Armisen et al. 2015; Crumiere et al. 2016) such as stagnant or flowing waters of streams and creeks, to which water striders have adapted over 60 million years of evolution (Andersen 1998; Andersen et al. 1993; Damgaard 2008; Ye et al 2017). A basic general view of the evolution that produces a match between various macro and micro morphological adaptations and the requirements of diverse aquatic habitats of Gerridae has been presented ~ 40 years ago (Andersen 1976, 1979, 1982, 1995), and was recently revived with a focus on the evolutionary developmental genetics of macro-morphological (Crumiere et al 2016) and micro-morphological (Finet et al. 2018, Santos et al. 2017) traits. However, apart from general categorizations of habitat types (e.g., Andersen 1976, 1979, 1989, 1995), the relevant quantitative statistical and experimental approaches to habitat selection concerned mostly stagnant and slow-flowing water habitats (e.g., Riley 1919; Vepsalainen 1973; Vepsalainen and Jarvinen 1974; Spence 1979, 1981; Cooper 1984; Haskins et al. 1997), and did not consider comparative evaluations of a hypothetical match between the locomotion required in a given aquatic habitat and detailed descriptions of micro-morphological adaptations to locomotion of a variety of water strider taxa in the diversity of aquatic habitats.
Some morphological studies concerned morphology of the “typical” Palearctic and Nearctic water striders from genera Gerris and Aquarius that live in still or relatively slow-flowing waters. For example, already 40 years ago it was known (e.g., Andersen 1976) that the midleg tarsi and tibiae (parts that interact with water surface) of these “typical” water striders are covered with diverse hairs (formally called setae but as the less formal term “hairs” has also been used we use it too). At least five different types of hairs varying in length, diameter, and cross-section shape are present in densities from hundreds (larger hair) to several thousand (smaller hairs) per mm2 with asymmetrical distribution consisting of especially diversified hair layers on the ventral side of tibiae and tarsi (surfaces that interact with water surface). Andersen (1976) also noted that the hair’s surface consists of small grooves running along a hair. It has also been known for a long time that all these morphological characteristics contribute to the hydrophobicity/superhydrophobicity of water strider legs, and that the special hair types on legs play a role in the rowing function of midlegs (Baudoin 1955; Holdgate 1955; Nightingall 1974; Andersen 1976, 1982). All the aforementioned studies focus on the morphology of mostly the two genera, Gerris and Aquarius. However, it has also been noted that some taxonomic groups of water striders such as the small marine water striders Halobatinae (Andersen 1976; Mahadik et al. 2020) or the large water striders Ptilomerinae (Esaki 1927; Jehamalar et al 2018) have special structures on the midleg tarsi and tibiae functioning as paddles.
The recent revival of interest in water striders by scientists focusing on bio-mimicry expanded this already existing knowledge (Brinkhurst 1960; Darnhofer-Demar 1969; Nightingall 1974; Andersen 1976; Bowdan 1976, 1978; Caponigro and Eriksen 1976) and resulted in a series of novel studies determining the physics of the water striders’ hydrophobicity (e.g., Gao and Jiang 2004; Feng et al. 2007; Wei et al. 2009; Watson et al 2010; Wang et al. 2015; Uesugi et al. 2020) and locomotion behavior (Hu et al 2003; Denny 2004; Buhler 2007; Perez-Godwyn et al. 2008; Hu and Bush 2010; Koh et al. 2015; Crumiere et al. 2016; Yang et al. 2016; Lu et al. 2018; Sun et al. 2018; Steinman et al. 2018, 2021; Baek et al. 2020). However, none of these new studies have explored the hypothetical adaptive functions of each of the several types of hairs, and their uneven distribution on the leg’s cuticle, as described by Andersen (1976) for Gerris. Additionally, none of these studies even considered large water striders with specialized “paddles” on their midlegs, such as the genus Ptilomera (e.g., Esaki 1927; Cheng and Fernando 1969; Jehamalar et al. 2018).
The rarely studied (except for taxonomy-focused papers such as Polhemus and Zettel 1997; Polhemus 2001; Vitheepradit and Sites 2007; Zettel 2009; Jehamalar et al. 2018; Zheng et al. 2019) large water striders from the subfamily Ptilomerinae, such as genus Ptilomera, inhabit streams and creeks of subtropical and tropical SE Asian regions (Andersen 1982; Tseng 1999, Jehamalar et al. 2018). Considering basic information in the literature (Andersen 1982; Esaki 1927; Cheng and Fernando 1969; Jehamalar et al. 2018), it is expected that these water striders differ from the “typical” water striders in the following aspects. Unlike in the well-studied genera, such as Gerris or Aquarius, the body of Ptilomera is supported only on four legs, the hindlegs’ tibiae and tarsi and the forelegs’ tarsi, leaving midlegs free for rowing. The midlegs have a “brush” of setae along the ventral edge of the midleg tibiae and tarsi. Therefore, we predicted that Ptilomera’s natural behavior in typical habitats may involve the frequent use of such a specialized midleg’s structure for rowing.
The “typical” water striders genus Aquarius are able to optimally choose their foraging positions in a stream by finding a tradeoff between the increase in the energy intake from the larger number of food items brought by the faster-flowing water and the considerable energy needed for striding against the fast current to maintain their foraging position (Rubenstein 1984). They maintain a specific position in flowing water using midlegs for rowing against the current and by relying on visual information about the surroundings (Junger and Dahmen 1988; Junger and Varju 1990). However, except for Fairbairn and Brassard’s (1988) research on Aquarius remigis, detailed quantitative tests of naturally preferred water flow speeds by water striders have not been conducted. The basic literature on Ptilomera’s biology (e.g., Cheng and Fernando1969; Andersen 1982; Tseng 1999) suggests without any quantitative statistical assessments that within the typical creeks inhabited by Ptilomera the water striders may prefer sites with relatively fast water flow.
The aim of the study was to present observations of Ptilomera tigrina’s locomotion and to quantitatively test their preferences for water flow speed in their typical natural habitats. We also aimed to provide basic micro-imaging of micro-morphological structures on their midlegs used for rowing. Based on the results, we suggest that the main function of the row of ribbon-like setae on ventral midleg tibiae and tarsi is not the support of insect body on the surface but providing thrust through rowing during striding locomotion in their preferred relatively high flow speeds and during their typical locomotory behaviors.
Materials and methods
Study sites and species
We conducted the study at two sites: the Melinh Station for Biodiversity (21° 23′ 01.9″ N 105° 42′ 44.2″ E = Google map: 21.383870, 105.712264; Fig. 1a; Fig. S1a), Vinh Phuc Province, Vietnam, and “May waterfalls” (Thac May; 20° 21′ 51.4″ N 105° 26′ 51.6″ E = Google map: 20.364275, 105.447665), in the vicinity of the Cuc Phuong National Park, Vietnam, (Fig. S1a in Supplementary Materials Part 3). Examples of habitats studied are in Fig. S1b. At both locations, we studied Ptilomera tigrina (Fig. 2a), one of the common large water striders in Vietnam (Polhemus 2001). All research was conducted in accordance with the laws of Vietnam. The Melinh Station for Biodiversity, IEBR, allowed us to collect specimens, and the Vietnam National Museum of Nature, VAST, provided the specimens loan permits.
Observational and experimental methods. a An example of flow speed sampling. The “Ptilomera present” sites (red) were chosen based on the presence of insects upon arrival of the observer who evaluated the presence of insects from a distance without disturbing their normal behavior. The “Ptilomera absent” sites (light gray) were chosen semi-randomly taking into account that they should include a full range of visually estimated flow speeds from still/nearly stagnant to high-speed flow, and that together with “Ptilomera present” sites they should cover the full creek surface area without a noticeable bias. b photo of the experimental bowl. c an example of an experimental set-up in a creek with two bowls representing two treatments: “Still water” and “Flowing water”, and with cameras set on tripods to film the experiment. All photos by W. Kim & P.G. Jablonski
The study species, Ptilomera tigrina, and brushes of ribbon-like hairs on its midlegs interacting with water surface during locomotion. a a female of Ptilomera tigrina in a natural habitat with the tibia-tarsus joint marked with a white rectangle; notice that midlegs are not used to support the insect’s body on the water surface; b zoomed-in view on the tibia-tarsus joint; notice the presence of “hair brushes” on both the distal tibia and proximal tarsus; c, d, e Scanning Electron Micrographs of the brush of ribbon-like hairs on midleg tibiae and tarsi shown at different magnifications (separate scale bars shown in each panel). Notice nano-groves on the surface of the ribbons in (e). Photos in a, c by WK & PGJ; photo in b by NPD; SEM images in d, e by PGJ, SIL
Morphology and SEM imaging
We weighed and photographed each individual water strider (GEM20 High Precision Digital Milligram Jewelry Scale, Smart Weigh, 0.001 g) in the manner that allowed extraction of morphological measurements from the photographs using ImageJ. We used Scanning Electron Microscopy (SEM (JSM-6390LV, JEOL, Japan) to examine the morphology of the brush of setae on the midleg tibia and tarsi, which were dried in a vacuum chamber, attached to the aluminum mounts by a carbon tape, and coated with gold by a sputter coater (Cressington 108auto, Cressington Scientific Instruments, UK) for ~ 200 s using a current of 30 mA. We used the same procedure to obtain SEM images of midleg tarsi of Aquarius paludum for comparison with the main study species, P. tigrina.
Observations of locomotion in the natural habitat
We conducted observations of locomotion of individual water striders in October, 2014, June 2016, and June 2018 in a creek near the Melinh Station for Biodiversity. We filmed (high speed and standard movies using Casio Exilim Z-1000 and Sony RX10-III cameras) the locomotion of water striders in various situations in natural conditions to obtain a full spectrum of natural locomotory behaviors of this species in its natural habitat. We used some of the collected movie clips to compare the stroke frequency by P. tigrina between the sites with relatively slow-flowing water and those with fast-flowing water. The categorization into slow-flowing and fast-flowing water sites was made without quantitative assessment of water flow but rather based on the visual evaluation and choice of movies that clearly represent relatively slow and those that clearly represent the relatively fast water flow (the data set is presented in Supplementary Materials Part 6).
Evaluation of water flow speed preferences in the natural habitat
In March 2016, we conducted observations of flow speed preferences in a stream near the Melinh Station for Biodiversity (Fig. 1a) at locations where Ptilomera was present in the creek (n = 63), and compared them with the velocities at locations in the creek where Ptilomera was absent (n = 65). This resulted in the total 128 locations. The strategy for choosing the locations is shown in an example in Fig. 1a. At each location, we measured the velocity of water at the surface by throwing small pieces of paper (5 × 5 mm) on the surface, filming their movement using a camera located directly above the water (120 fps), digitizing the movement of the center of each piece of paper from the video during a total of 17–1974 frames per movie to extract their speed, and finally calculating water flow speed at a location as an average value from speeds of the pieces of papers at each location. (2–3 pieces of paper at 110 locations), or using the value for one piece of paper if only one was digitized at a location (18 locations). We used quadratic logistic regression for statistical analysis of the effect of the flow speed (continuous explanatory variable) on the use of the habitat (binary variable with two values indicating water strider “present” or “absent’ at a sampled location) in R version 3.6.1. The regression model assumes that the relationship follows a specific type of function chosen by the researcher, and we used the quadratic regression. We additionally run an alternative test that does not have any assumptions about the nature of the differences between the water strider “present” and “absent” distributions: the classical non-parametric Kolmogorov-Smirnoff D test for differences in flow speed distribution between water strider present and absent locations.
For graphical purposes, we presented the distribution (with the frequency expressed as % of all observations) of Ptilomera-present and Ptilomera-absent speeds (n = 63 and n = 65, respectively) in 5 cm/s bins over the range from 0 to 30 cm/s and in 10 cm/s bins for the range of speeds 30-90 cm/s. Due to the small sample size for the very high speeds, the bin width was larger for faster speeds. Next, we created a smoothed figure using the Excel’s chart type “Scatter with Smooth Lines” and x-axis data comprising the values of mid-range for each bin and y-axis comprising % of observations in a bin. We also calculated the traditional Chi-square test for statistical comparison of water speed distributions between Ptilomera-present and Ptilomera-absent categories. As the test does not provide reliable conclusions if cell counts are near zero, we calculated the Chi-square tests for two alternative contingency tables: the 4 × 2 and 5 × 2 tables (Fig. S3c), in which the range of observed speeds was divided into either four (0–10, 10–20, 20–40, > 40 cm/s) or five classes (0–10, 10–15, 15–20, 20–40, > 40 cm/s). The results from these additional Chi-square tests are presented only in Supplementary Materials Part 5. The raw data set is presented in the Supplementary Materials Part 7.
Experiments: comparison of behavior in flowing versus non-flowing water
As the observations in the natural habitat revealed that Ptilomera tigrina appears to avoid non-flowing water, we conducted experiments in 2019 to compare their behavior on flowing and non-flowing water. We used two plastic bowls (Fig. 1b, c; diameter ~ 50 and depth ~ 15 cm) placed side by side in the middle of a creek. The bowls were freshly filled with water from the creek each time before a trial. One bowl imitated still water and the other bowl imitated flowing water conditions. We put holes on the bottom of the still water bowl to keep the water inside the bowl constantly blending with the creek water without disturbance of the water surface in the bowl. We cut two openings in the flowing water bowl’s opposite walls to allow water to flow through the bowl (Fig. 1b). To prevent water striders from escaping through these openings we covered the openings with plastic mesh (mesh size ~ 5 mm) such that water can flow but water striders cannot go through. The two bowls were located next to each other in a shady location, and experiments in the two treatments were conducted simultaneously so that the time of day and lighting do not bias the comparison between the two treatments (Fig. 1c).
A trial consisted of putting a freshly captured Ptilomera into a bowl and observing (and filming; Fig. 1c) its behavior. We counted the number of jumps out of the bowl per minute during the first 5 min as an indicator of rejection of the situation. A 20-cm-high wall around the bowl perimeter (Fig. 1b) around the bowl prevented water striders from escaping during a jump, except for relatively high jumps. We conducted 15 tests in still water bowls (10 at “May waterfall” and 5 at Melinh Station for Biodiversity) and 14 tests in flowing water bowls (10 at “May waterfall” and 4 at Melinh Station for Biodiversity, where one individual escaped before data were collected). As the relative contributions of the two locations to the number of tests in still and flowing water treatments were similar (10:5 and 10:4 for waterfall:station in still and flowing treatment respectively), we believe that we avoided the potential bias due to different contributions of the two sites to each treatment. Therefore, we analyzed the pooled data (an alternative analysis of each site separately led to the same conclusions). Each individual was tested once. Based on earlier observations in the natural habitats, we expected that Ptilomera may jump more in bowls with still water, and we used the Mann–Whitney test to evaluate this prediction. The raw data are presented in the Supplementary Materials Part 8.
Results
Observations of locomotion and midleg’s micro-morphology
Ptilomera tigrina is a relatively large water strider with the average body mass of 115 ± 30 mg and 107 ± 18 mg for males and females, respectively (Fig. 2a, and Supplementary Materials Part 2: Table S1 and Fig. S2a). While only males possess the fringe of hairs on their femur (compare femur of a male in Fig. S2a with that of a female in Fig. 2a), both sexes have a “brush” of ribbon-like setae on the ventral (water-facing, i.e., interacting with water surface) side of tibiae and tarsi of midlegs (Fig. 2b–e; Fig. S2b, d). Those structures are absent from the commonly studied water striders such as A. paludum (Fig. S2c). The ribbons in the “brush” reach the length of about 250 μm and therefore the brush adds considerably to the surface area that pushes the water surface dimple during rowing (typical rowing is without surface breaking, Supplemental Movies 1,2; description of all movies’ content is in Supplementary Materials Part 1). The “brushes” are the most developed on the distal tibia and proximal tarsus: the leg’s sections (near the tibio-tarsal joint; rectangle in Fig. 2a, b) that actively push the water meniscus during locomotion (see below). Forelegs and hindlegs do not have this structure.
Observations of Ptilomera tigrina in the natural habitat show that their midlegs are not used for support on the water surface. They are used for rowing against the current (Fig. 3a, b; Supplemental Movies 1, 2), apparently adjusting the strikes’ strength and frequency to the flow speed such that an individual can remain in approximately the same location for an extended time period (Supplemental Movies 2 and 3) resulting in high stroke frequency (4.24 ± 0.79 strokes per second; mean ± SD, n = 6) in fast-flowing water and a relatively low stroke frequency (1.96 ± 0.35 strokes per second, n = 6; Fig. 3c) on slow-flowing water (Fig. 3c). The main section of midlegs that produces the most prominent meniscus during rowing is the section comprising distal tibia and proximal tarsus (marked in Supplemental Movie 1). While midlegs are normally not used to support the body (e.g., Fig. 2a), they may serve this function when the water strider uses forelegs to handle captured prey (Fig. 3d). When a potential food item is brought on the surface with the flowing current then the individual makes extra strides to examine the potential food item and returns if the item is not edible. When a food item is captured, a water strider holds it between the femur and tibia of both forelegs making it impossible for the forelegs to support the body. In this situation, water striders use midlegs to support the body or to push the body forward using an asymmetric mode of striding when one midleg pushes the insect while another is extended forward to provide support on the surface (Supplemental Movie 4).
The behavior of Ptilomera tigrina in natural situations based on screenshots from the slow-motion movie frames and photos. a a water strider is pushed back by the water current and it regularly moves forward pushed by the midlegs’ stroke (two images were combined to illustrate the two positions of the insect); b a composite image illustrating a typical sequence of midleg positions during striding against current; c comparison of stroke frequency on slow and fast water; d a photo illustrating that midlegs are used for support when forelegs are used for prey handling (see also Supplemental Movie 4); e a photo-based schematics of typical back-and-forth slow striding on still and near-stagnant water surface (see also Supplemental Movie 5). a and b are composite images created from several frames in a movie (see also Supplemental Movies 1, 2). Photos by W. Kim and P.G. Jablonski
On very slow-flowing water and still water surface, the undisturbed water striders use midlegs to slowly push their body back and forth (Fig. 3e), which results in almost constant movements of the insect body on the water surface (Supplemental Movie 5; striding frequency in this clip was about 1 back-and-forth cycles /sec). However, when disturbed by visual or tactile (through water waves) stimuli indicating potential danger, the species performs super-fast back and forth, as well as diagonal, movements powered by the midleg’s brushes in an apparent antipredatory “protean” (Humphries and Driver 1970) type of behavior with back-and-forth strides occurring in various directions at a relatively high frequency (e.g., 4.3 strides/sec in Supplemental Movie 6). This behavior occurs in both flowing and still water and it is triggered only by stimuli indicating potential danger.
Preferences for water flow speed in natural habitats
During the survey of water flow speed preferences, water striders were only observed at locations with water flow ranging from ~ 0.1 to ~ 0.6 m/s, while the full range of recorded surface water flow speeds ranged from 0 (still water in small “bays” at the edge of the stream, separated by rocks/plants from the main current) to ~ 0.8 m/s (in the middle of rapid flow between rocks). Individuals of Ptilomera tigrina were observed especially often, relative to the observed frequency of flow speeds at sites without water striders, in the flow speed range from ~ 0.15 to ~ 0.30 m/s (Fig. 4a–c; see also Figure S3a in Supplementary Materials Part 5). The frequency distribution of flow speed for “Ptilomera present” sites was statistically significantly different (Kolmogorov–Smirnov test, D = 0.29133, p-value = 0.008761; see Supplementary Material Part 5 for additional Chi-square tests), and the effect of water flow velocity on the probability of the water strider’s occurrence can be described by quadratic logistic regression (p < 0.0001; Fig. 4d; Table S2 in Supplementary Materials Part 5). In summary, the results illustrate that water striders avoid still water and prefer water flow of ~ 0.15 to ~ 0.30 m/s, albeit they are also observed maintaining their positions in faster water flows of up to ~ 0.6 m/s.
Water flow preferences of Ptilomera tigrina water striders and their behavior on still water. a An example of natural habitat with water flow speed measured at Ptilomera present and Ptilomera absent sites; b distribution (% of locations) of flow speeds at Ptilomera present (solid line, circle markers) and Ptilomera absent (broken line, square markers) locations; c degree of preferences calculated as % for Ptilomera present minus % of Ptilomera absent. The gray area in b and c indicates the range of flow speeds that are preferred by water striders; d Results of quadratic logistic regression of presence/absence of P. tigrina as a function of water flow speed; red and gray circles represent empirical data of Ptilomera present and Ptilomera absent sites; gray vertical arrows and gray box along the x-axis indicate the range for which probability of P. tigrina’s presence is larger than 0.5. Data file and a basic histogram of the frequencies of flow speeds for Ptilomera absent and Ptilomera present sites, as well as details of statistical analyses are presented in Supplementary Materials Part 5. Photo in (a) by P.G. Jablonski
Behavior on still and flowing water in experimental containers
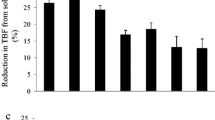
The water striders tried to escape from the container with still water significantly more often than from the container with flowing water (Fig. 5; Mann–Whitney test, two-tailed, U = 27.5, Ucritical p<0.05 = 59; z = 3.36056, p = 0.00078). Their locomotory behavior differed between the two types of containers in a manner consistent with field observations. They pushed their bodies back-and-forth in the containers with still water, and they performed forward strides against the current in the containers with flowing water (Supplemental Movie 7).
The behavior of Ptilomera tigrina water striders in artificial containers with still and flowing water located in the natural habitat. The effect of water flow on the tendency to get away from an experimental container: number of jump attempts/min in the first 5 min of test in two treatments: still (stagnant) water and flowing water. The circles indicate data points, the circle’s diameter represents the number of data points of the same value, the horizontal thick line indicates the median value. Sample sizes are n = 15 and n = 14 for stagnant and flowing treatment, respectively. The data file is provided in Supplementary Materials Part 8
Discussion
All the results are consistent with the idea that Ptilomera tigrina water striders’ water flow preferences and their typical locomotory striding behaviors involve constant rowing performed by midlegs, and that midlegs are normally not used for support on the water surface. The preferred range of water flow speeds (0.15–0.3 m/s) observed in our study is higher than the preferred range by A. remigis (~ 0.1 m/s; Fairbairn & Brassard 1988), the species that also lives in streams and represents the differentiation of hairs on the midlegs typical for water striders in the most commonly studies genera, Gerris and Aquarius (e.g., Andersen 1976). As we did not observe midlegs being used for support on the water surface (except in special situations involving prey handling with forelegs), we propose that, unlike in the “typical” water striders (e.g., genera Gerris, Aquarius), who use midlegs for support on the unbroken water surface as well as for rowing, the main function of midlegs in P. tigrina is just rowing. Therefore, we hypothesize that the unique taxon-specific brush of ribbon-like hairs on Ptilomera’s midleg tibiae and tarsi (Fig. 2b–e; Esaki 1927; Cheng and Fernando 1969; Polhemus and Zettel 1997; Jehamalar et al 2018; Kim et al. unpublished) is an adaptation to consistent rowing against relatively fast water flow (without water surface breaking), rather than to supporting insect body on the water surface.
Consistent with this interpretation is the absence of similar structures in A. paludum (Fig. S2c; Uesugi et al 2020) and other species of Aquarius or Gerris (Anderson 1976, 1982; Gao and Jiang 2004; Feng et al 2007; Su et al. 2010), which are the “typical” water striders who use midlegs for two functions: supporting the insect body on the unbroken water surface and rowing (Andersen 1976, 1982; Caponigro and Eriksen 1976; Bowdan 1978). Also, the lack of these structures on P. tigrina’s (Kim et al. unpublished) as well as other Ptilomerinae’s (e.g., Cheng and Fernando 1969; Polhemus and Zettel 1997; Jehamalar et al 2018) fore and hind legs, which do not provide thrust during rowing (but are used for support on the water surface), is consistent with this hypothesis. Finally, these structures appear to be the most developed in Ptilomera on the distal tibia and proximal tarsus near tibio-tarsal joint (our observations and information in Cheng and Fernando 1969 and Jehamalar et al 2018), i.e., along the ventral edge of the midleg’s section that actively pushes that water meniscus during locomotion (as seen in our supplemental videos). Therefore, their adaptive function clearly seems to be related to the crucial role of midlegs in constant rowing against the relatively fast and moderate water flow speeds preferred by P. tigrina in their natural habitats, and also in slow constant rowing on still water as well as super-fast “protean” locomotion (Humphries and Driver 1970) in response to danger. This fast antipredatory locomotion is different from antipredatory vertical jumps/leaps in water striders of genus Gerris or Aquarius because the antipredatory escape locomotion observed in A. nebularis and A.conformis (Krupa and Sih 1999; Haskins et al. 1997) involves a few seconds of several chaotic jumps that quickly take the water strider away from danger, while Ptilomera’s locomotory response to danger involves constantly repeated and extremely fast horizontal back-and-forth movements at approximately the same location over an extended period (see Supplemental Movie 6). This suggests the importance of persistent rowing for survival. Although an experimental laboratory-based comparison of striding performance by individuals with the brushes removed and with brushes intact is needed to completely evaluate this hypothesis, our observations in the natural habitat suggest an adaptive match between the preferences for the situations requiring frequent fast movements powered by rowing on the water surface and the presence of the specialized midleg “brushes” of ribbon-like setae that function as paddles for rowing (and are not used for support on the water surface) by P. tigrina.
Although the preferred flow speeds are larger than in the other quantitatively studied water striders (Fairbairn and Brassard 1988), they are intermediate considering all ranges of flow speeds present in the Ptilomera’s habitat. We did not observe individuals of P. tigrina in areas of still water and near-stagnant (near-zero water flow speed) even though those areas are more likely to be located near vegetation on the creek’s edge where shadows are more prevalent and might provide protection from the sun (Ptilomera tend to occur in more shady spots; T.A. Duc, personal observations). Ptilomera’s avoidance of the fastest flowing water is consistent with the optimal foraging reasoning by Rubenstein (1984) who observed that A. remigis individuals chose to spend time in the water current that is sufficiently fast to bring food items at a relatively high rate to the vicinity of an individual, but sufficiently slow to permit individuals to stay in it for an extended period using frequent rowing against the current. As we have not measured the rate of food items’ arrival in various flow speeds, we are unable to fully evaluate this hypothesis, except for direct video evidence that the rowing strike rate is obviously faster in faster currents (Supplemental Movie 3). Additionally, we cannot evaluate the likely hypothesis that a hungry individual chooses a faster flow that provides more resources and when satiated or exhausted by rowing it moves to the slower flowing parts of the creek. A bimodal shape of “Ptilomera present” flow speeds’ distribution (Fig. 4b) is consistent with, but does not fully confirm this hypothesis. It is also possible, albeit never studied, that faster flow brings more micro-debris that can clog the setae on the water striders’ legs and this may be an additional cost of staying in fast-flowing water. Naturally, we cannot exclude the opposite scenario, that stagnant and slow water at the edges of creeks accumulates debris that can clog the micro-structures on water striders’ legs, and faster water flow cleans the legs from this debris. Ptilomera tigrina seems to be a good species to test those hypotheses in the future.
Hair fringes generally similar to the “brushes” in Ptilomera are also present in another subfamily of large stream-dwelling water striders, Cylindrostethinae (T.A. Duc, personal observation), as well as in a subfamily of small water striders (Halobatinae) with a body mass of only a few milligrams (Andersen 1976; Mahadik et al. 2020; Kim W, Jablonski PG. personal observations). They apparently need powerful rowing performance as some of the species live in relatively fast freshwater creeks (e.g., genus Austrobates; Andersen and Weir 2004), and others on the surface of oceanic waters (e.g., genus Halobates; Andersen 1976, 1982; Mahadik et al. 2020) where requirements of the physical habitat may create the need for powerful rowing performance by these small-bodied insects. Similar to Ptilomerinae, the small marine water striders, Halobatinae, also do not seem to use midlegs for the main support of their body on the water surface (Andersen 1976) but only touch the surface during resting position (Kim W, Jablonski PG. personal observations on Asclepios). As the typical habitats in the subfamily of Ptilomerinae appear to be fast-flowing creeks and streams (Cheng and Fernando1969; Andersen 1982; Tseng 1999; Jehamalar et al 2018), we predict that similar local preferences for water flow speed and similar locomotory behaviors may be discovered in future quantitative studies of other species of Ptilomerinae, and maybe also in other water strider species from fast-flowing creeks. We predict that the main function of their midleg’s microstructures on tarsi/tibiae may turn out to be rowing rather than support on the water surface.
Our observations of P. tigrina in its natural habitats appear to challenge some of the recently promoted research paradigms in modern interdisciplinary science of new hydrophobic materials and water walking devices, which are inspired by truly simplified knowledge about behavior and midleg morphology of “typical” water striders, Aquarius and Gerris (e.g., Gao and Jiang 2004; Feng et al. 2007; Wei et al. 2009; Watson et al. 2010; Wang et al. 2015; Uesugi et al. 2020; Hu et al. 2003; Denny 2004; Buhler 2007; Perez-Godwyn et al. 2008; Hu and Bush 2010; Koh et al. 2015, Yang et al. 2016; Lu et al. 2018; Sun et al. 2018; Steinman et al. 2018, 2021; Baek et al. 2020). The paradigm accepts a simplified notion of a midleg as an elastic rod/tube, covered with a relatively non-differentiated layer of hairs (with nano-grooves) that simultaneously serve two functions: providing general hydrophobicity for the support of the insect’s body on the unbroken water surface and pushing the unbroken meniscus of water surface backward during rowing locomotion. Although those new studies bring a crucial new level of quantitative understanding of the physics of water strider’s hydrophobicity and locomotion, they ignore the already known diversity among water striders. Our study reminds the scientists that this simplified approach has a narrow empirical basis, and that a shift of paradigm is needed to incorporate not only the long-existing knowledge on hair differentiation on the power-providing midlegs in the two “typical” genera (e.g., Andersen 1976). More importantly, we argue that scientists and engineers need to incorporate the view that many water strider taxa have legs with diverse sophisticated and specialized micro and nano structures (as revealed in numerous taxonomic studies) that very likely serve different specific functions required for life and locomotion in their species-specific semi-aquatic habitats. The current focus of engineers on the largely simplified view of midlegs (of Gerris/Aquarius) that likely represent an adaptive trade-off between adaptations to supporting insect’s body on the unbroken water surface and adaptations to propelling the insect in striding locomotion does not allow to reveal the micro-designs of legs optimized specifically for support on water surface separately from the micro-structures optimized specifically for rowing locomotion. We propose that a collaboration between taxonomists and ethologists during intense field studies in natural habitats, and a full consideration of microstructural complexity of fore, mid and hind legs can provide new discoveries and ideas for novel morphological designs for bio-inspired technology of water walking devices.
Data availability
As the three data sets are relatively small, they are all included in the Supplementary Information file (Supplementary Materials Parts 6, 7, 8).
References
Andersen NM (1976) A comparative study of locomotion on the water surface in semiaquatic bugs (Insecta, Hemiptera, Gerromorpha). Vidensk Meddr Dansk Naturh Foren 139:337–396
Andersen NM (1979) Phylogenetic inference as applied to the study of evolutionary diversification of semiaquatic bugs (Hemiptera: Gerromorpha). Syst Zool 28:554–578
Andersen NM (1995) Cladistic inference and evolutionary scenarios: locomotory structure, function, and performance in water striders. Cladistics 11:279–295
Andersen NM, Spence JR, Wilson MVH (1993) 50 million years of structural stasis in water striders (Hemiptera: Gerridae). Am Entomol 39:174–176
Andersen NM, Weir TA (2004) Australian water bugs: Their biology and identification (Hemiptera – Heteroptera, Gerromorpha & Nepomorpha). Entomograph 4:0–344; Apollo Books, Denmark; Csiro Publishing, Australia
Andersen NM (1982) The Semiaquatic Bugs (Hemiptera: Gerromorpha) Vol. 3. Scandinavian Science Press LTD
Andersen NM (1998) Water striders from the Paleogene of Denmark with a review of the fossil record and evolution of semiaquatic bugs (Hemiptera, Gerromorpha). Biologiske Skrifter 50, The Royal Danish Academy of Sciences and Letters. Copenhagen. Det Kongelige Danske Videnskabernes Selskab, Printed in Denmark by Special-Trykkeriet Viborg a-s
Armisen D, Refki PN, Crumiere AJJ, Viala S, Toubiana W, Khila A (2015) Predator strike shapes antipredator phenotype through new genetic interactions in water striders. Nat Commun 6:8153
Baek M, Lawin KM, Codden CJ, Lim H, Yang E, Kim HY, Jablonski PG (2020) Water strider females use individual experience to adjust jumping behaviour to their weight within physical constraints of water surface tension. Sci Rep 10:18657
Baudoin R (1955) La Physico-chimie des surfaces dans la vie des Arthropodes aériens, des miroirs d’eau des rivages marins et lacustres de la zone intercotidale. Bull Biol Fr Belg 89:16–164
Bowdan E (1978) Walking and rowing in the waterstrider, Gerris remigis. I. A cinematographic study of walking. J Comp Physiol 123:43–49
Bowdan E (1976) The functional anatomy of the mesothoracic leg of the waterstrider, Gerris remigis Say (Heteroptera). Psyche 83:289
Brinkhurst RO (1960) Studies on the functional morphology of Gerris najas Degger (Hem. Het. Gerridae). Proc Zool Soc London 133:531–559
Bühler O (2007) Impulsive fluid forcing and water strider locomotion. J Fluid Mech 573:211–236
Caponigro MA, Eriksen CH (1976) Surface film locomotion by the water strider, Gerris remiges Say. Am Midland Nat 95:268–278
Carpenter GH (1928) The biology of insects. London, Sidgwick & Jakson, Ltd
Cheng L, Fernando CH (1969) A taxonomic study of the Malayan Gerridae (Hemiptera: Heteroptera) with notes on their biology and distribution. Orient Insects 3:97–160
Cooper SD (1984) The effects of trout on water striders in stream pools. Oecologia 63:376–379
Crumiere AJJ, Santos ME, Semon M, Armisen D, Moreira FF, Khila A (2016) Diversity in morphology and locomotory behavior is associated with niche expansion in the semi-aquatic bugs. Curr Biol 26:3336–3342
Damgaard J (2008) Evolution of the semi-aquatic bugs (Hemiptera: Heteroptera: Gerromorpha) with a re-interpretation of the fossil record. Acta Entomol Mus Nat Pragae 48:251–268
Darnhofer-Demar B (1969) Zur Fortbewegung des Wasserlaufers Gerris lacustris L. auf der Wasseroberflache. Verh. Deut. Zool. Ges. Innsbruck 1968. Zool Anz Suppl 32:429–439
Denny MW (2004) Paradox lost: answers and questions about walking on water. J Exp Biol 207:1601–1606
Esaki T (1927) Revision of the Ptilomera-group of the Gerridae, with descriptions of three new species (Heteroptera). Eos Rev Esp Ent 3:25J – 268
Fairbairn DJ, Brassard J (1988) Dispersion and spatial orientation of Gerris remigis in response to water current: a comparison of pre-and post-diapause adults. Physiol Entomol 13(2):153–164
Feng XQ, Gao X, Wu Z, Jiang L, Zheng QS (2007) Superior water repellency of water strider legs with hierarchical structures: experiments and analysis. Langmuir 23:4892–4896
Finet C, Decaras A, Armisen D, Khila A (2018) The achaete-scute complex contains a single gene that controls bristle development in the semi-aquatic bugs. Proc Roy Soc b 285:2852018238720182300
Gao X, Jiang L (2004) Water-repellent legs of water striders. Nature 432:36
Haskins KE, Sih A, Krupa JJ (1997) Predation risk and social interference as factors influencing habitat selection in two species of stream-dwelling waterstriders. Beh Ecol 8:351–365
Holdgate MW (1955) The wetting of insect cuticles by water. J Exp Biol 32:591–617
Hu DL, Bush JW (2010) The hydrodynamics of water-walking arthropods. J Fluid Mech 644:5–33
Hu DL, Chan B, Bush JWM (2003) The hydrodynamics of water strider locomotion. Nature 424:663–666
Humphries DA, Driver PM (1970) Protean defence by prey animals. Oecologia 5:285–302
Jehamalar EE, Chandra K, Basu S, Selvakumar C (2018) Review of Ptilomera (Ptilomera) (Hemiptera: Heteroptera: Gerridae) from India with description of a new species. Zootaxa 4370:501–518
Junger W, Dahmen HJ (1988) Waterstriders (Gerridae) use visual landmarks to compensate for drift on a moving water surface. In: Elsner N, Barth FG (eds) Sense organs: interfaces between environment and behaviour. Proceedings of the 16th Göttingen Neurobiology Conference. Thieme, Stuttgart New York p 35
Junger W, Varju D (1990) Drift compensation and its sensory basis in waterstriders (Gerris paludum F.). J Comp Physiol 167:441–446
Koh JS, Yang E, Jung GP, Jung SP, Son JH, Lee SI, Jablonski PG, Wood RJ, Kim HY, Cho KJ (2015) Jumping on water: Surface tension—Dominated jumping of water striders and robotic insects. Science 349:517–521
Krupa JJ, Sih A (1999) Comparison of antipredator responses of two related water striders to a common predator. Ethology 105:1019–1033
Lu H, Zheng Y, Ying W, Pesika TD, N, Meng Y, Tian Y, (2018) Propulsion principles of water striders in sculling forward through shadow method. J Bionic Eng 15:516–525
Mahadik GA, Hernandez-Sanchez JF, Arunachalam S, Gallo A Jr, Cheng L, Farinhaa AS, Thoroddsen ST, Mishra H, Duarte CM (2020) Superhydrophobicity and size reduction enabled Halobates (Insecta: Heteroptera, Gerridae) to colonize the open ocean. Sci Rep 10:7785. https://doi.org/10.1038/s41598-020-64563-7
Nightingall W (1974) Locomotion: Mechanics and hydrodynamics of swimming in aquatic insects. In: Rockstein M (ed): The physiology of Insecta (2nd ed) ch6: 381–432.
Perez Godwyn PJ, Want JT, Want ZJ, Ji AH, Dai ZD, Fujisaki K (2008) Water striders: the biomechanics of water locomotion and functional morphology of the hydrophobic surface (Insecta: Hemiptera-Heteroptera). J Bionic Eng 5:121–126
Polhemus JT (2001) A review of the genus Ptilomera (Heteroptera: Gerridae) in Indochina, with descriptions of two new species. J New York Entomol Soc 109:214–234
Polhemus JT, Zettel H (1997) Five new Potamometropsis species (Insecta: Heteroptera: Gerridae) from Borneo. Ann Naturhist Mus Wien 88B:21–40
Riley CFC (1919) Some habitat responses of the large water-strider, Gerris remigis Say. Am Nat 53:394–414
Rubenstein DI (1984) Resource acquisition and alternative mating strategies in water striders. Amer Zoo 24:345–353
Santos ME, Le Bouquin A, Crumière AJ, Khila A (2017) Taxon-restricted genes at the origin of a novel trait allowing access to a new environment. Science 358(6361):386–390
Spence JR (1981) Experimental analysis of microhabitat selection in water-striders (Heteroptera: Gerridae). Ecology 62:1505–1514
Spence JR, Andersen NM (1994) Biology of water striders: interactions between systematics and ecology. Annu Rev Entomol 39:101–128
Spence J (1979) Microhabitat selection and regional coexistence in water-striders (Heteroptera: Gerridae). Ph.D. Thesis, The University of British Columbia
Steinman T, Arutkin M, Cochard P, Raphael E, Casas J, Benzaquen M (2018) Unsteady wave pattern generation by water striders. J Fluid Mech 848:370–387
Steinman T, Cribellier A, Casas J (2021) Singularity of the water strider propulsion mechanisms. J Fluid Mech 915:A118. https://doi.org/10.1017/jfm.2021.156
Su Y, Ji B, Zhang K, Gao H, Huang Y, Hwang K (2010) Nano to micro structural hierarchy is crucial for stable superhydrophobic and water-repellent surfaces. Langmuir 26(7):4984–4989
Sun P, Zhao M, Jiang J, Zheng Y (2018) The study of dynamic force acted on water strider leg departing from water surface. AIP Adv. 8:015228
Tseng M (1999) Notes on mating behavior and sexual size dimorphism of two species of water striders (Hemiptera: Gerridae) in Vietnam. J Kansas Entomol Soc 72:327–329
Uesugi K, Mayama H, Morishima K (2020) Proposal of a water-repellency model of water strider and its verification by considering directly measured strider leg-rowing force. J Photopolym Sci Technol 33:185–192
Vepsalainen K (1973) The distribution and habitats of Gerris Fabr. Species (Heteroptera, Gerridae) in Finland. Ann Zool Fenn 10:419–444
Vepsalainen K, Jarvinen O (1974) Habitat utilization of Gerris argentatus (Heteroptera Gerridae). Ent Scand 5:189–195
Vitheepradit A, Sites RW (2007) A review of Ptilomera (Heteroptera: Gerridae) in Thailand, with descriptions of three new species. Ann Entomol Soc Am 100:139–151
Wang Q, Yao X, Liu H, Quere D, Jiang L (2015) Self-removal of condensed water on the legs of water striders. PNAS 112:9247–9252
Watson GS, Cribb BW, Watson JA (2010) Experimental determination of the efficiency of nanostructuring on non-wetting legs of the water strider. Acta Biomater 6:4060–4064
Wei PJ, Chen SC, Lin JF (2009) Adhesion forces and contact angles of water strider legs. Langmuir 25(3):1526–1528
Yang E, Son JH, Lee SI, Jablonski PG, Kim HY (2016) Water striders adjust leg movement speed to optimize takeoff velocity for their morphology. Nat Commun 7:13698
Ye Z, Zhen Y, Damgaard J, Chen PP, Zhu L, Bu ZC, W, (2017) Biogeography and diversification of Holarctic water striders: Cenozoic temperature variation, habitat shifting and multiple intercontinental dispersals. Syst Entomol 43:19–30
Zettel H (2009) Three new genera of Ptilomerinae (Hemiptera: Heteroptera: Gerridae) from Southeast Asia. Zootaxa 2046:26–42
Zheng C, Ye Z, Zhu X, Zhang H, Dong X, Chen P, Bu W (2019) Integrative taxonomy uncovers hidden species diversity in the rheophilic genus Potamometra (Hemiptera: Gerridae). Zool Scr 49:174–186
Acknowledgements
We would like to thank the crew of the Melinh Station for Biodiversity. We thank the following persons who helped in various matters during our research: Dang Huy Phuong (Head of Melịnh Station for Biodiversity), Nguyen Van Mon, Nguyen Van Dat, Nguyen Van Khoi, Nguyen Van Ty, Trinh Xuan Thanh. We thank E. Sterling (New York) and K. Koy (Berkeley) for providing the image of a map used in Fig. S1a. We thank Jeongwon Yoon for helping in data analysis. We also thank all individuals that helped us during the field studies. We thank three anonymous reviewers for their comments.
Funding
This study was funded by BK 21 program to the School of Biological Sciences, SNU; NRF grant 2019R1A2C1004300; DGIST Start-up Fund Program nr 20200810 of the Ministry of Science, ICT and Future Planning of Korea.
Author information
Authors and Affiliations
Contributions
Conceptualization: WK, JH, PGJ, SIL; methodology: WK, HTP, JH, PGJ, SIL; formal analysis: WK, PGJ, NPD, PGJ, SIL; investigation: WK, PGJ, NPD, JH, PGJ, SIL, TAD; writing original draft: PGJ, SIL, WK; writing-review & editing: all authors; visualization: WK, PGJ; supervision: PGJ; project administration: PGJ, SIL, HTP.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing or financial interests.
Ethical approval
No approval of research ethics committees was required for this research, which was conducted in accordance with the laws of Vietnam.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 25936 KB) Ptilomera tigrina female striding on slow current. Movie timeline: 2-7 s. - normal speed (1x); 8-30s. – slowed down 4x. Each section of a midleg (femur, tibia, and tarsus) is marked with white solid lines to show that distal tibia and proximal tarsi are the main parts of the midleg that interact with the water surface during striding
Supplementary file2 (MP4 34628 KB) Moves in slow and fast current, in the natural habitat. Movie timeline: 3-7 seconds – normal play speed (1x) in slow and fast current; 9-27 seconds – slowed down 16x in slow current; 28-40 seconds – slowed down 16x in fast current
Supplementary file3 (MP4 11022 KB) An example of striding rate differences between slow and fast current – play speed 4x
Supplementary file4 (MP4 14752 KB) The water strider holds prey in its forelegs and cannot use the forelegs for support. Therefore, it uses midlegs for support of frontal body part and for slow asymmetrical rowing (these situations happen on slow-flowing water of the P. tigrina’s habitat). Movie timeline: 5-7 seconds – normal play speed (1x); 9-17 seconds – play speed 4x. In this situation midlegs are used for support and rowing is asymmetrical
Supplementary file5 (MP4 22803 KB) An example of back-and-forth movements on the still water surface or in very slow-moving water. Movie timeline: 3-8 seconds – normal play speed (1x); 9-26 seconds – slowed down 4x.
Supplementary file6 (MP4 24873 KB) “protean” antipredatory movements– very fast, back and forth, and to the sides. Movie timeline: 4-9 seconds – normal play speed (1x); 12-29 seconds – slowed down 8x
Supplementary file7 (MP4 20621 KB) Water striders in the experimental bowls with flowing or still water. Movie timeline: 2-5 seconds – normal play speed (1x); 6-24 seconds – slowed down 8x
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kim, W., Pham, T.H., Nguyen, P.D. et al. Locomotion and flow speed preferences in natural habitats by large water striders, Ptilomera tigrina, with micro-morphological adaptations for rowing. J Ethol 40, 211–221 (2022). https://doi.org/10.1007/s10164-022-00749-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10164-022-00749-y