Abstract
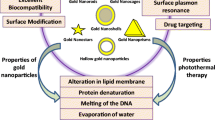
Hyperthermia is a promising alternative modality for the conventional cancer treatments. Nanoparticle-mediated photothermal therapy (PTT) has been widely applied for hyperthermia cancer therapy by a near-infrared light irradiation. Some special nanoparticles can convert light energy into heat and destroy the tumor cells. Inspired from the photothermal efficacy of the gold nanoparticles, here we synthesized, characterized, and applied novel photothermal polyethylene glycol-curcumin-gold nanoparticles (PEG-Cur-Au NPs) in cancer PTT. The effect of PEG-Cur-Au NPs upon irradiation by an 808-nm laser on C540 (B16/F10) cell line as well as implanted (bearing) melanoma tumor in inbred C57 mice was investigated. In vitro temperature increment, cell viability evaluation, and histological analyses were performed. The results showed a dose-dependent cytotoxicity of PEG-Cur-Au NPs toward C540 (B16/F10) cell line at concentrations ≥ 25 μg mL−1 with an IC50 value of 42.7 μg mL−1 in dark (and with no toxicity for 10 μg mL−1). On the other hand, 808-nm laser irradiation alone (without using PEG-Cur-Au NPs) for 10 min induced killing effect on the C540 (B16/F10) cell line in a laser power-dependent manner at power density > 0.5 W cm−2 (no toxicity for 0.5 W cm−2). However, PPT using PEG-Cur-Au NPs was tremendously observed after laser illumination. Even under laser irradiation at a power density of 0.5 W cm−2 of PEG-Cur-Au NPs of concentrations < 10 μg mL−1, PTT of the cells was substantial. Histological analyses and volume measurements of the induced tumors in the mice revealed an appropriate control of the tumors upon PTT by PEG-Cur-Au NPs. Combination of PEG-Cur-Au NP administration and 808-nm diode laser irradiation destroyed the melanoma cancer cells in the animal model.






Similar content being viewed by others
References
Zbytek B, Carlson JA, Granese J, Ross J, Mihm MC Jr, Slominski A (2008) Current concepts of metastasis in melanoma. Expert Rev Dermatol 3:569–585
Wyld L, Audisio RA, Poston GJ (2015) The evolution of cancer surgery and future perspectives. Nat Rev Clin Oncol 12:115–124
Moran MS (2015) Radiation therapy in the locoregional treatment of triple-negative breast cancer. Lancet Oncol 16:113–122
Deng GL, Zeng S, Shen H (2015) Chemotherapy and target therapy for hepatocellular carcinoma: new advances and challenges. World J Hepatol 7:787–798
Sahu A, Choi WI, Lee JH, Tae G (2013) Graphene oxide mediated delivery of methylene blue for combined photodynamic and photothermal therapy. Biomaterials 34:6239–6248
Mousavy SJ, Riazi GH, Kamarei M, Aliakbarian H, Sattarahmady N, Sharifizadeh A, Safarian S, Ahmad F, Moosavi-Movahedi AA (2009) Effects of mobile phone radiofrequency on the structure and function of the normal human hemoglobin. Int J Biol Macromol 44:278–285
Shibu ES, Hamada M, Murase N, Biju V (2013) Nanomaterials formulations for photothermal and photodynamic therapy of cancer. J Photochem Photobiol C: Photochem Rev 15:53–72
Huang XH, El-Sayed IH, Qian W, El-Sayed MA (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128:2115–2120
Kubler C, Reuther T (2007) Photodynamic therapy. Possibilities and limits for head and neck carcinomas. Onkologe 13:158–164
Choi WI, Kim JY, Kang C, Byeon CC, Kim YH, Tae G (2011) Tumor regression in vivo by photothermal therapy based on gold-nanorod-loaded, functional nanocarriers. ACS Nano 5:1995–2003
Okuno T, Kato S, Hatakeyama Y, Okajima J, Maruyama S, Sakamoto M, Mori S, Kodama T (2013) Photothermal therapy of tumors in lymph nodes using gold nanorods and near-infrared laser light. J Control Release 172:879–884
Boca SC, Potara M, Gabudean AM, Juhem A, Baldeck PL, Astilean S (2011) Chitosan-coated triangular silver nanoparticles as a novel class of biocomptible, highly effective photothermal transducers for in vitro cancer cell therapy. Cancer Lett 311:131–140
Wang TT, Zhang LY, Su ZM, Wang CG, Liao Y, Fu Q (2011) Multifunctional hollow mesoporous silica nanocages for cancer cell detection and the combined chemotherapy and photodynamic therapy. ACS Appl Mater Interfaces 3:2479–2486
Tian QW, Tang MH, Sun YG, Zou RJ, Chen ZG, Zhu MF, Yang SP, Wang JL, Wang JH (2011) Hydrophilic flower-like CuS superstructures as an efficient 980 nm laser-driven photothermal agent for ablation of cancer cells. Adv Mater 23:3542–3547
Huang XQ, Tang SH, Mu XL, Dai Y, Chen GX, Zhou ZY, Ruan FX, Yang ZL, Zheng NF (2011) Free standing palladium nanosheets with plasmonic and catalytic properties. Nat Nanotechnol 6:28–32
Sattarahmady N, Rezaie-Yazdi M, Tondro GH, Akbari N (2017) Bactericidal laser ablation of carbon dots: an in vitro study on wild-type and antibiotic-resistant Staphylococcus aureus. J Photochem Photobiol B 166:323–332
Huang XH, Jain PK, El-Sayed IH, El-Sayed MA (2008) Plasmonic photothermal therapy (PPTT) using gold nanoparticles. Lasers Med Sci 23:217–228
Sattarahmady N, Tondro GH, Golchin M, Heli H (2015) Gold nanoparticles biosensor of Brucella spp. genomic DNA: visual and spectrophotometric detections. Biochem Eng J 97:1–7
Gad SC, Sharp KL, Montgomery C, Payne JD, Goodrich GP (2012) Evaluation of the toxicity of intravenous delivery of auroshell particles (gold-silica nanoshells). Int J Toxicol 31:584–594
Sattarahmady N, Rahi A, Heli H (2017) A signal-on built in-marker electrochemical aptasensor for human prostate-specific antigen based on a hairbrush-like gold nanostructure. Sci Rep 7:11238
Hu J, Sanz-Rodrigue F, Rivero F, Rivero EM, Torres RA, Ortgies DH, Sole JG, Alfonso F, Jaque D (2018) Gold nanoshells: contrast agents for cell imaging by cardiovascular optical coherence tomography. Nano Res 11:676–685
Moradi M, Sattarahmady N, Rahi A, Hatam GR, Sorkhabadi SMR, Heli H (2016) Label-free, PCR-free and signal-on electrochemical DNA biosensor for Leishmania major based on gold nanoleaves. Talanta 161:48–53
Dehdari Vais R, Sattarahmady N, Karimian K, Heli H (2015) Green electrodeposition of gold hierarchical dendrites of pyramidalnanoparticles and determination of azathioprine. Sensors Actuators B 215:113–118
Chen J, Liang H, Lin L, Guo ZP, Sun PJ, Chen MW, Tian HY, Deng MX, Chen XS (2016) Gold nanorods-based gene carriers with the capability of photoacoustic imaging and photothermal therapy. ACS Appl Mater Interfaces 8:31558–31566
Heidari M, Sattarahmady N, Azarpira N, Heli H, Mehdizadeh A, Zare T (2016) Photothermal cancer therapy by gold-ferrite nanocomposite and near-infrared laser in animal model. Lasers Med Sci 31:221–227
Kirui DK, Rey DA, Batt CA (2010) Gold hybrid nanoparticles for targeted phototherapy and cancer imaging. Nanotechnology 21:105105
Qiu JJ, Wei WD (2014) Surface plasmon-mediated photothermal chemistry. J Phys Chem C 118:20735–20749
Sattarahmady N, Kayani Z, Heli H (2015) Highly simple and visual colorimetric detection of Brucella melitensis genomic DNA in clinical samples based on gold nanoparticles. J Iran Chem Soc 12:1569–1576
Gamal-Eldeen AM, Moustafa D, El-Daly SM, El-Hussieny EA, Saleh S, Khoobchandani M, Bacon KL, Gupta S, Katti K, Shukla R, Katti KV (2016) Photothermal therapy mediated by gum Arabic-conjugated gold nanoparticles suppresses liver preneoplastic lesions in mice. J Photochem Photobiol B 163:47–56
Nakamura T, Tamura A, Murotani H, Oishi M, Jinji Y, Matsuishi K, Nagasaki Y (2010) Large payloads of gold nanoparticles into the polyamine network core of stimuli-responsive PEGylated nanogels for selective and noninvasive cancer photothermal therapy. Nano 2:739–746
Abo-Elfadi MT, Gamal-Eldeen AM, Elshafey MM, Abdalla GM, Ali SS, Ali MRK, Zawrah ZMFM (2016) Photothermal therapeutic effect of PEGylated gold nano-semicubes in chemically-induced skin cancer in mice. J Photochem Photobiol B 164:21–29
Chen R, Zheng XC, Qian HQ, Wang X, Wang J, Jiang XQ (2013) Combined near-IR photothermal therapy and chemotherapy using gold-nanorod/chitosan hybrid nanospheres to enhance the antitumor effect. Biomater Sci 1:285–293
Martins CVB, da Silva DL, Neres ATM, Magalhaes TFF, Watanabe GA, Modolo LV, Sabino AA, de Fatima A, de Resende MA (2009) Curcumin as a promising antifungal of clinical interest. J Antimicrob Chemother 63:337–339
Kuttan R, Bhanumathy P, Nirmala K, George MC (1985) Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett 29:197–202
Ribeiro APD, Pavarina AC, Dovigo LN, Brunetti IL, Bagnato VS, Vergani CE, Costa CAD (2013) Phototoxic effect of curcumin on methicillin-resistant Staphylococcus aureus and L929 fibroblasts. Lasers Med Sci 28:391–398
Paschoa MA, Tonon CC, Spolidorio DMP, Bagnato VS, Giusti JSM, Santos-Pinto L (2013) Photodynamic potential of curcumin and blue LED against Streptococcus mutans in a planktonic culture. Photodiagn Photodyn Ther 10:313–319
Choudhuri T, Pal S, Agwarwal ML, Das T, Sa G (2002) Curcumin induces apoptosis in human breast cancer cells through p53-dependent Bax induction. FEBS Lett 512:334–340
Javvadi P, Segan AT, Tuttle SW, Koumenis C (2008) The chemopreventive agent curcumin is a potent radiosensitizer of human cervical tumor cells via increased reactive oxygen species production and overactivation of the mitogen-activated protein kinase pathway. Mol Pharmacol 73:1491–1501
Liu HL, Chen Y, Cui GH, Zhou JF (2005) Curcumin, a potent anti-tumor reagent, is a novel histone deacetylase inhibitor regulating B-NHL cell line Raji proliferation. Acta Pharmacol Sin 26:603–609
Ahn JC, Kang JW, Shin JI, Chung PS (2012) Combination treatment with photodynamic therapy and curcumin induces mitochondria-dependent apoptosis in AMC-HN3 cells. Int J Oncol 41:2184–2190
Dujic J, Kippenberger S, Ramirez-Bosca A, Diaz-Alperi J, Bereiter-Hahn J, Kaufmann R, Bernd A, Hofmann M (2009) Curcumin in combination with visible light inhibits tumor growth in a xenograft tumor model. Int J Cancer 124:1422–1428
Wu WT, Shen J, Banerjee P, Zhou SQ (2011) Water-dispersible multifunctional hybrid nanogels for combined curcumin and photothermal therapy. Biomaterials 32:598–609
Dovigo LN, Pavarina AC, Ribeiro APD, Brunetti IL, Costa CAD, Jacomassi DP, Bagnato VS, Kurachi C (2011) Investigation of the photodynamic effects of curcumin against Candida albicans. Photochem Photobiol 87:895–903
Singh DK, Jagannathan R, Khandelwal P, Abraham PM, Poddar P (2013) In situ synthesis and surface functionalization of gold nanoparticles with curcumin and their antioxidant properties: an experimental and density functional theory investigation. Nano 5:1882–1893
Manju S, Sreenivasan K (2012) Gold nanoparticles generated and stabilized by water soluble curcumin-polymer conjugate: blood compatibility evaluation and targeted drug delivery onto cancer cells. J Colloid Interface Sci 368:144–151
Palmal S, Maity AR, Singh BK, Basu S, Jana NR (2014) Inhibition of amyloid fibril growth and dissolution of amyloid fibrils by curcumin-gold nanoparticles. Chem Eur J 20:6184–6191
Gangwar RK, Dhumale VA, Kumari D, Nakate UT, Gosavi SW, Sharma RB, Kale SN, Datar S (2012) Conjugation of curcumin with PVP capped gold nanoparticles for improving bioavailability. Mater Sci Eng C 32:2659–2663
Bot A, Obrocea M, Marincola FM (eds) (2011) Cancer vaccines: from research to clinical practice. Taylor and Francis, p 271
Song CW, Park HJ, Lee CK, Griffin R (2005) Implications of increased tumor blood flow and oxygenation caused by mild temperature hyperthermia in tumor treatment. Int J Hyperth 21:761–767
Kaneti YV, Chen CY, Liu MS, Wang XC, Yang JL, Taylor RA, Jiang XC, Yu AB (2015) Carbon-coated gold nanorods: a facile route to biocompatible materials for photothermal applications. ACS Appl Mater Interfaces 7:25658–25668
Bush JA, Cheung KJJ, Li G (2001) Curcumin induces apoptosis in human melanoma cells through a Fas receptor/caspase-8 pathway independent of p53. Exp Cell Res 271:305–314
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4(2007):807–818
Zeng XB, Leung AWN, Xia XS, Yu HP, Bai DQ, Xiang JY, Jiang Y, Xu CS (2010) Effect of blue light radiation on curcumin-induced cell death of breast cancer cells. Laser Phys 20:1500–1503
Link S, El-Sayed MA (2000) Shape and size dependence of radiative, non-radiative and photothermal properties of gold nanocrystals. Int Rev Phys Chem 19:409–453
Zharov VP, Galitovsky V, Viegas M (2003) Photothermal detection of local thermal effects during selective nanophotothermolysis. Appl Phys Lett 83:4897–4899
Zharov VP, Galitovskaya EN, Johnson C, Kelly T (2005) Synergistic enhancement of selective nanophotothermolysis with gold nanoclusters: potential for cancer therapy. Lasers Surg Med 37:219–226
Liao HW, Hafner JH (2005) Gold nanorod bioconjugates. Chem Mater 17:4636–4641
Maeda H, Fang J, Inutsuka T, Kitamoto Y (2003) Vascular permeability enhancement in solid tumor: various factors, mechanisms involved and its implications. Int Immunopharmacol 3:319–328
Acknowledgements
This paper was extracted from F. Rahimi-Moghaddam M.Sc. thesis supported by the Research Council of Shiraz University of Medical Sciences (11648).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was conducted according to the Committee on the Ethics of Animal Experiments of Shiraz University of Medical Sciences.
Rights and permissions
About this article
Cite this article
Rahimi-Moghaddam, F., Azarpira, N. & Sattarahmady, N. Evaluation of a nanocomposite of PEG-curcumin-gold nanoparticles as a near-infrared photothermal agent: an in vitro and animal model investigation. Lasers Med Sci 33, 1769–1779 (2018). https://doi.org/10.1007/s10103-018-2538-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-018-2538-1