Abstract
The relative stabilities of the alkali [M ⊂ 222]+ cryptates (M = Na, K, Rb and Cs) in the gas phase and in solution (80:20 v/v methanol:water mixture) at 298 K, are computed using a combination of ab initio quantum-chemical calculations (HF/6-31G and MP2/6-31+G*//HF/6-31+G*) and explicit-solvent Monte Carlo free-energy simulations. The results suggest that the relative stabilities of the cryptates in solution are due to a combination of steric effects (compression of large ions within the cryptand cavity), electronic effects (delocalization of the ionic charge onto the cryptand atoms) and solvent effects (dominantly the ionic dessolvation penalty). Thus, the relative stabilities in solution cannot be rationalized solely on the basis of a simple match or mismatch between the ionic radius and the cryptand cavity size as has been suggested previously. For example, although the [K ⊂ 222]+ cryptate is found to be the most stable in solution, in agreement with experimental data, it is the [Na ⊂ 222]+ cryptate that is the most stable in the gas phase. The present results provide further support to the notion that the solvent in which supramolecules are dissolved plays a key role in modulating molecular recognition processes.

Alkali cryptates [M ⊂ 222]+ (M = Na, K, Rb and Cs) relative stabilities in gas and methanol:water solution: solvent effects and molecular recognition


Similar content being viewed by others
References
Lehn JM (1995) Supramolecular chemistry: Concepts and perspectives. VCH, New York
Dobler M (1981) Ionophores and their structures. Wiley, New York
Lehn JM (1993) Science 260:1762–1763
Cram DJ (1988) Angew Chem - Int Ed Eng 27:1009–1020
Lehn JM (1978) Pure Appl Chem 50:871–892
Cram DJ, Cram JM (1974) Science 183:803–809
Kyba EP (1977) J Am Chem Soc 99:2564–2571
Rauwolf C, Straßner T (1997) J Mol Mod 3:1–16
Bell SEJ, McKervey MA, Fayne D, Kane P, Diamond D (1998) J Mol Mod 4:44–52
Puchta R, Seitz V, Hommes NvE, Saalfrank RW (2000) J Mol Mod 6:126–132
Dickert FL, Landgraf S, Sikorski R (2000) J Mol Mod 6:491–497
Puchta R, Clark T, Bauer W (2006) J Mol Mod 12:739–747
Pumera M, Rulíšek L (2006) J Mol Mod 12:799–803
Stryer L (1988) Biochemistry, 3rd edn. W. H. Freeman and Company, New York, p 967
Marrone TJ, Merz Jr KM (1995) J Am Chem Soc 117:779–791
Auffinger P, Wipff G (1991) J Am Chem Soc 113:5976–5988
Boudon S, Wipff G (1991) J Chim Phys 88:2443–2449
Troxler L, Wipff G (1994) J Am Chem Soc 116:1468–1480
Feller D (1997) J Phys Chem A 101:2723–2731
More MB, Ray D, Armentrout PB (1999) J Am Chem Soc 121:417–423
Glendening ED, Feller D, Thompson MA (1994) J Am Chem Soc 116:10657–10669
Auffinger P, Wipff G (1991) J Chim Phys 88:2525–2534
Wipff G (1992) J Coord Chem 27:7–37 Part B
Zhang XX, Izatt RM, Bradshaw JS, Krakowiak KE (1998) Coord Chem Rev 174:179–189
Martell AE, Hancock RD, Motekaitis RJ (1994) Coord Chem Rev 133:39–65
Solomons TWG (1992) Organic chemistry, 5th edn. Willey, New York, p 443
Galle M, Puchta R, Hommes NvE, van Eldik R (2006) Z Phys Chem 220:511–523
[K ⊂ 222]+: Moras D, Metz B, Weiss R (1973) Acta Cryst B 29:383–388; [Rb ⊂ 222]+ and [Cs ⊂ 222]+: Moras D, Metz B, Weiss R (1973) Acta Cryst B 29:388–395; [Na ⊂ 222]+: Moras D, Weiss R (1973) Acta Cryst B 29:396–399
Krakowiak KE, Zhang XX, Bradshaw JS, Zhu CY, Izatt RM (1995) J Incl Phenom 23:223–231
Chen Q, Cannell K, Nicoll J, Deaden DV (1996) J Am Chem Soc 118:6335–6344
McQuarrie DA (1976) Statistical Mechanics, 1st edn. HarperCollins Publishers, New York
Frisch MJ, Trucks GW, Schlegel HB, Gill PMW, Johnson BG, Robb MA, Cheeseman JR, Keith TA, Petersson GA, Montgomery JA, Raghavachari K, Al-Laham MA, Zakrzewski VG, Ortiz JV, Foresman JB, Peng CY, Ayala PY, Wong MW, Andres JL, Replogle ES, Gomperts R, Martin RL, Fox DJ, Binkley JS, Defrees DJ, Baker J, Stewart JP, Head-Gordon M, Gonzalez C, Pople JA (1995) Gaussian 94 (Revision D.1) Gaussian Inc, Pittsburgh PA
Hehre WJ, Radom L, Schleyer PvR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Kollman P (1993) Chem Rev 93:2395–2417
Allen MP, Tildesley DJ (1997) Computer simulation of liquids. Clarendon Press, Oxford
Barker JA, Watts RO (1973) Mol Phys 26:789–792
Jorgensen WL (1998) OPLS force fields. In: Schleyer PvR (ed) The encyclopedia of computational chemistry, vol 3. Wiley, New York, p 1986
Breneman CM, Wiberg KB (1990) J Comp Chem 11:361–373
Huheey JE, Keiter EA, Keiter PL (1993) Inorganic chemistry: Principles and reactivity, 4th edn. Harper Collins, New York, p 114
Fuentealba P, Preuss H, Stoll H, Szentpály LV (1982) Chem Phys Lett 89:418–422
Jorgensen WL, Blake JF, Buckner JK (1989) Chem Phys 129:193–200
Freitas LCG (1991) Diadorim Program. Theoretical Chemistry Laboratory, Departamento de Química, UFSCar, São Carlos, SP, Brazil; Freitas LCG (1993) J Mol Struct: Theochem 101:151–158
Kastenholz MA, Hünenberger PH (2006) J Chem Phys 124:224501-1-20
Helgaker T, Jorgensen P, Olsen J (2000) Molecular electronic-structure theory, 1st edn. Wiley, New York, chapter 15
Soong L-L, Leroi GE, Popov AI (1992) J Inclusion Phenom 12:253–262
Marcus Y (1997) Ion properties, 1st edn. Marcel Dekker, New York, p 121
Kalidas C, Hefter G, Marcus Y (2000) Chem Rev 100:819–852
Lehn JM, Sauvage JP (1975) J Am Chem Soc 97:6700–6707
Born M (1920) Z Phys 1:45–48
Bem-Naim A (1987) Solvation thermodynamics. Plenum Press, New York
Freitas LCG, Botelho LF (1994) Quim Nova 17:489–495
Acknowledgements
This work was partially supported by FAPESP, CAPES, CNPq, PADCT and CENAPAD-SP. ESL thanks FAPESP for the award of a fellowship.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
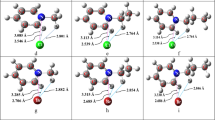
The Supplementary Material contains the Cartesian coordinates of the optimized structures as well as their corresponding pictures with the atom labels.
Figure S2
Structure of the [K ⊂ 222]+ endo cryptate optimized with the HF/6-31+G* method, together with the corresponding atom-labeling scheme (see coordinates in Table S3). (DOC 99.5 kb)
Figure S3
Structure of the [K ⊂ 222]+ endo cryptate optimized with the HF/6-31+G*/ LanL2DZ method, together with the corresponding atom-labeling scheme (see coordinates in Table S4). (DOC 92.5 kb)
Figure S4
Structure of the [Rb ⊂ 222]+ endo cryptate optimized with the HF/6-31+G*/LanL2DZ method, together with the corresponding atom-labeling scheme (see coordinates in Table S5). (DOC 94 KB)
Figure S5
Structure of the [Cs ⊂ 222]+ endo cryptate optimized with the HF/6-31+G*/LanL2DZ method, together with the corresponding atom-labeling scheme (see coordinates in Table S6). (DOC 95.5 kb)
Figure S6
Structure of the [Cs ⊂ 222]+ exo cryptate optimized with the HF/6-31+G*/LanL2DZ method, together with the corresponding atom-labeling scheme (see coordinates in Table S7). (DOC 93 kb)
Table S1
Partial atomic charges (in |e|) used in the explicit-solvent MC simulations of the [M ⊂ 222]+ cryptates. The charges were derived from a CHELPG analysis based on the optimized structures (at the same level of theory as the optimization for [Na ⊂ 222]+ and [K ⊂ 222]+, or at the HF/6-31G level with an SDD pseudopotential [27] for the Rb and Cs atoms in [Rb ⊂ 222]+ and [Cs ⊂ 222]+). See Figures S1, S2, S3 and S4 for the atom-labeling scheme. (DOC 42.5 kb)
Table S2
Cartesian coordinates of the [Na ⊂ 222]+ endo cryptate optimized with the HF/6-31+G* method (energy -1422.3664973 E h; MP2 energy -1426.152491 E h). See Fig. S1 for the structure and atom-labeling scheme. (DOC 42.5 kb)
Table S3
Cartesian coordinates of the [K ⊂ 222]+ endo cryptate optimized with the HF/6-31+G* method (energy -1859.6493126 E h; MP2 energy -1863.4713692 E h). See Fig. S2 for the structure and atom-labeling scheme. (DOC 42 kb)
Table S4
Cartesian coordinates of the [K ⊂ 222]+ endo cryptate optimized with the HF/6-31+G*/LanL2DZ method (energy -1288.3847267 E h; MP2 energy -1292.2569353 E h). See Fig. S3 for the structure atom labeling scheme. (DOC 42 kb)
Table S5
Cartesian coordinates of the [Rb ⊂ 222]+ endo cryptate optimized with the HF/6-31+G*/LanL2DZ method (energy -1284.0926095 E h; MP2 energy -1287.9527188 E h). See Fig. S4 for the structure and atom-labeling scheme. (DOC 42 kb)
Table S6
Cartesian coordinates of the [Cs ⊂ 222]+ endo cryptate (based on the crystallographic structure) optimized with the HF/6-31+G*/LanL2DZ method (energy -1280.1065032 E h; MP2 energy -1283.9468867 E h). See Fig. S5 for the structure and atom-labeling scheme. (DOC 41.5 kb)
Rights and permissions
About this article
Cite this article
Leite, E.S., Santana, S.R., Hünenberger, P.H. et al. On the relative stabilities of the alkali cations 222 cryptates in the gas phase and in water-methanol solution. J Mol Model 13, 1017–1025 (2007). https://doi.org/10.1007/s00894-007-0213-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-007-0213-8