Abstract
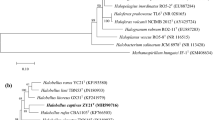
A single extremely halophilic strain was isolated from salt brine produced when a fresh water lake flooded a large salt mine located beneath the lake. The water that entered this mine contained less than 0.34 M NaCl, but over time, this sealed brine became saturated by Cenozoic age salt (121–125 million-year BCE). The isolated strain requires at least 1.7 M NaCl for survival and grows optimally in 3.1 M NaCl. Therefore, it could not have survived or been present in the waters that flooded this salt mine. The strain grows at a pH range from 6.5 to 9.0 and has a wide tolerance to temperatures from 25 ℃ to at least 60 ℃. The comparison of 16S rRNA and rpoB′ genes revealed that strain 1–13-28T is related to Halorubrum tebenquichense DSM 14210T showing 98.6% and 98.1% similarities, respectively. Phylogenetic analyses based on 16S rRNA, rpoB′ genes and 122 concatenated archaeal genes show that the strain 1–13-28T consistently forms a cluster with Halorubrum tebenquichense of the genus Halorubrum. Strain 1–13-28T contained sulfated mannosyl glucosyl diether, and the polar lipid profile was identical to those of most Halorubrum species. Based on the overall combination of physiological, phylogenetic, polar lipids and phylogenomic characteristics, strain 1–13-28T (= ATCC 700083T = CGMCC 1.62627T) represents a newly identified species within the genus Halorubrum for which the name Halorubrum hochsteinianum is proposed.



Similar content being viewed by others
Data availability
The sequences determined in this study have been deposited in the NCBI Genbank database.
References
Amoozegar MA, Siroosi M, Atashgahi S, Smidt H, Ventosa A (2017) Systematics of haloarchaea and biotechnological potential of their hydrolytic enzymes. Microbiology 163(5):623–645. https://doi.org/10.1099/mic.0.000463
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA et al (2008) The RAST Server: rapid annotations using subsystems technology. BMC Genomics 9:75. https://doi.org/10.1186/1471-2164-9-75
Chen S, Liu HC, Zhou J, Xiang H (2016) Halorubrum pallidum sp. nov., an extremely halophilic archaeon isolated from a subterranean rock salt. Int J Syst Evol Microbiol. 66(8):2980–2986. https://doi.org/10.1099/ijsem.0.001129
Cui HL, Zhou PJ, Oren A, Liu SJ (2009) Intraspecific polymorphism of 16S rRNA genes in two halophilic archaeal genera, Haloarcula and Halomicrobium. Extremophiles 13:31–37. https://doi.org/10.1007/s00792-008-0194-2
Cui HL, Gao X, Yang X, Xu XW (2010) Halorussus rarus gen. nov., sp. nov., a new member of the family Halobacteriaceae isolated from a marine solar saltern. Extremophiles 14:493–499. https://doi.org/10.1007/s00792-010-0329-0
Cui HL, Yang X, Mou YZ (2011) Salinarchaeum laminariae gen. nov., sp. nov.: a new member of the family Halobacteriaceae isolated from salted brown alga Laminaria. Extremophiles 15(6):625–631. https://doi.org/10.1007/s00792-011-0393-0
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Biol 20:406–416. https://doi.org/10.2307/2412116
Gold M (1981) Who pulled the plug on Lake Peigneur? Sci 81:56–63
Gonzalez C, Gutierrez C, Ramirez C (1978) Halobacterium vallismortis sp. nov. an amylolytic and carbohydrate-metabolizing, extremely halophilic bacterium. Can J Microbiol. 24(6):710–715. https://doi.org/10.1139/m78-119
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, Tiedje JM (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Han D, Cui HL (2015) Halorubrum laminariae sp. Nov., isolated from the brine of salted brown alga Laminaria. Antonie van Leeuwenhoek. 107(1):217–223. https://doi.org/10.1007/s10482-014-0319-9
Kanehisa M, Goto S, Hattori M, Aoki-Kinoshita KF, Itoh M, Kawashima S et al (2006) From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res 34:D354–D357. https://doi.org/10.1093/nar/gkj102
Kim M, Oh HS, Park SC, Chun J (2014) Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. Int J Syst Evol Microbiol 64:346–351. https://doi.org/10.1099/ijs.0.059774-0
Konstantinidis KT, Rosselló-Móra R, Amann R (2017) Uncultivated microbes in need of their own taxonomy. ISME J 11(11):2399–2406. https://doi.org/10.1038/ismej.2017.113
Lizama C, Monteoliva-Sanchez M, Suarez-Garcıa A, Rosello-Mora R, Aguilera M et al (2002) Halorubrum tebenquichense sp. nov., a novel halophilic archaeon isolated from the Atacama Saltern. Chile Int J Syst Evol Microbiol 52:149–155. https://doi.org/10.1099/00207713-52-1-149
Luo C, Rodriguez-R LM, Konstantinidis KT (2014) MyTaxa: an advanced taxonomic classifier for genomic and metagenomic sequences. Nucleic Acids Res 42(8):e73. https://doi.org/10.1093/nar/gku169
McGenity TJ, Gemmell RT, Grant WD, Stan-Lotter H (2000) Origins of halophilic microorganisms in ancient salt deposits. Environ Microbiol 2(3):243–250. https://doi.org/10.1046/j.1462-2920.2000.00105.x
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:60. https://doi.org/10.1186/1471-2105-14-60
Minegishi H, Kamekura M, Itoh T, Echigo A, Usami R, Hashimoto T (2010) Further refinement of the phylogeny of the Halobacteriaceae based on the full-length RNA polymerase subunit B’ (rpoB’) gene. Int J Syst Evol Microbiol 60(10):2398–2408. https://doi.org/10.1099/ijs.0.017160-0
Mormile MR, Biesen MA, Gutierrez MC, Ventosa A, Pavlovich JB, Onstott TC, Fredrickson JK (2003) Isolation of Halobacterium salinarum retrieved directly from halite brine inclusions. Environ Microbiol 5(11):1094–1102. https://doi.org/10.1046/j.1462-2920.2003.00509.x
Nguyen LT, Schmidt HA, Haeseler AV, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Oren A (2018) Halorubrum. In: Trujillo ME, Dedysh S, DeVos P, Hedlund B, Kämpfer P, Rainey FA, Whitman Bergey;s WB (eds) Manual of systematics of archaea and bacteria. Wiley, New York. https://doi.org/10.1002/9781118960608.gbm00487.pub2
Oren A, Ventosa A, Grant WD (1997) Proposed minimal standards for description of new taxa in the order Halobacteriales. Int J Syst Evol Microbiol 47:233–238. https://doi.org/10.1099/00207713-47-1-233
Park JS, Vreeland RH, Cho BC, Lowenstein TK, Timofeeff MN et al (2009) Haloarchaeal diversity in 23, 121 and 419 MYA salts. Geobiology 7(5):515–523. https://doi.org/10.1111/j.1472-4669.2009.00218.x
Parks DH, Chuvochina M, Waite DW, Rinke C, Skarshewski A et al (2018) A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat Biotechnol 36(10):996–1004. https://doi.org/10.1038/nbt.4229
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci U S A 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Richter M, Rosselló-Móra R, Olive GF (2016) JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics 32(6):929–931. https://doi.org/10.1093/bioinformatics/btv681
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Siakotos AN, Rouser G (1965) Analytical separation of nonlipid water soluble substances and gangliosides from other lipids by dextran gel column chromatography. J Am Oil Chem Soc 42:913–919. https://doi.org/10.1007/BF02632444
Stan-Lotter H, Pfaffenhuemer M, Legat A, Busse HJ, Radax C, Gruber C (2002) Halococcus dombrowskii sp. nov. an archaeal isolate from a Permo-Triassic alpine salt deposit. Int J Syst Evol Microbiol 52:1807–1814. https://doi.org/10.1099/00207713-52-5-1807
Sun YP, Wang BB, Zheng XW, Wu ZP, Hou J, Cui HL (2022) Description of Halosolutus amylolyticus gen. nov., sp. nov., Halosolutus halophilus sp nov and Halosolutus gelatinilyticus sp nov, and genome-based taxonomy of genera Natribaculum and Halovarius. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.005598
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 30(12):2725–2729. https://doi.org/10.1093/molbev/mst197
Vaskovsky VE, Kostetsky EY (1968) Modified spray for the detection of phospholipids on thin-layer chromatograms. J Lipid Res 9(3):396
Vreeland RH, Litchfield CD, Martin EL, Elliot E (1980) Halomonas elongata, a new genus and species of extremely salt-tolerant bacteria. Int J Syst Bacteriol 30:485–495. https://doi.org/10.1099/00207713-30-2-485
Vreeland RH, Rosenzweig WD, Powers DW (2000) Isolation of a 250 million year old bacterium from a primary salt crystal. Nature 407(6806):897–900. https://doi.org/10.1038/35038060
Vreeland RH, Jones J, Monson A, Rosenzweig WD, Lowenstein TK, Timofeeff M et al (2007) Isolation of live cretaceous (121–112 million years old) halophilic archaea from primary salt crystals. Geomicrobiol J 24:275–282. https://doi.org/10.1080/01490450701456917
Wainø M, Tindall BJ, Ingvorsen K (2000) Halorhabdus utahensis gen. nov., sp. nov., an aerobic, extremely halophilic member of the archaea from great salt lake. Utah Int J Syst Evol Microbiol 50:183–190. https://doi.org/10.1099/00207713-50-1-183
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by Oren.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vreeland, R.H., Sun, YP., Wang, BB. et al. Halorubrum hochsteinianum sp. nov., an ancient haloarchaeon from a natural experiment. Extremophiles 28, 1 (2024). https://doi.org/10.1007/s00792-023-01320-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00792-023-01320-4