Abstract
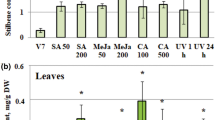
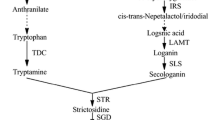
Vindoline is an important alkaloid produced in Catharanthus roseus leaves. It is the more important monomer of the scarce and costly anticancer bisindole alkaloids, vincristine, and vinblastine, as unlike catharanthine (the other monomer), its biosynthesis is restricted to the leaves. Here, biotic (bacterial endophyte, phytoplasma, virus) and abiotic (temperature, salinity, SA, MeJa) factors were studied for their effect on vindoline accumulation in C. roseus. Variations in vindoline pathway-related gene expression were reflected in changes in vindoline content. Since allene oxide cyclase (CrAOC) is involved in jasmonate biosynthesis and MeJa modulates many vindoline pathway genes, the correlation between CrAOC expression and vindoline content was studied. It was taken up for full-length cloning, tissue-specific expression profiling, in silico analyses, and upstream genomic region analysis for cis-regulatory elements. Co-expression analysis of CrAOC with vindoline metabolism-related genes under the influence of aforementioned abiotic/biotic factors indicated its stronger direct correlation with the tabersonine-to-vindoline genes (t16h, omt, t3o, t3r, nmt, d4h, dat) as compared to the pre-tabersonine genes (tdc, str, sgd). Its expression was inversely related to that of downstream-acting peroxidase (prx) (except under temperature stress). Direct/positive relationship of CrAOC expression with vindoline content established it as a key gene modulating vindoline accumulation in C. roseus.







Similar content being viewed by others
Abbreviations
- CMV:
-
Cucumber mosaic virus
- dat :
-
Acetyl coenzyme A: 4-O-deacetylvindoline 4-O-acetyltransferase
- d4h :
-
Desacetoxyvindoline 4-hydroxylase
- JA:
-
Jasmonate
- MeJa:
-
Methyl jasmonate
- MSA:
-
Multiple sequence alignment
- nmt :
-
S-adenosyl-L methionine: 16-methoxy-2,3-dihydro-3-hydroxytabersonine-N-methyltransferase
- omt :
-
16-Hydroxytabersonine-16-O-methyltransferase
- OPDA:
-
12-Oxo-phytodienoic acid
- ORCA:
-
Octadecanoid-responsive Catharanthus AP2-domain protein
- prx :
-
Peroxidase
- SA:
-
Salicylic acid
- sgd :
-
Strictosidine β-D-glucosidase
- str :
-
Strictosidine synthase
- tdc :
-
Tryptophan decarboxylase
- t16h :
-
Tabersonine 16-hydroxylase
- t3o :
-
Tabersonine 3-oxygenase
- t3r :
-
Tabersonine 3-reductase
References
Aerts RJ, Gisi D, De Carolis E, De Luca V, Baumann TW (1994) Methyl jasmonate vapor increases the developmentally controlled synthesis of alkaloids in Catharanthus and Cinchona seedlings. Plant J 5:635–643
Aerts RJ, Schafer A, Hesse M, Baumann TW, Slusarenko A (1996) Signalling molecules and the synthesis of alkaloids in Catharanthus roseus seedlings. Phytochemistry 42:417–422
Besseau S, Kellner F, Lanoue A, Thamm AMK, Salim V, Schneider B, Geu-Flores F, Höfer R, Guirimand G, Guihur A, Oudin A, Glevarec G, Foureau E, Papon N, Clastre M, Giglioli-Guivarc’h N, St-Pierre B, Werck-Reichhart D, Burlat V, De Luca V, O’Connor SE, Courdavault V (2013) A pair of tabersonine 16-hydroxylases initiates the synthesis of vindoline in an organ-dependent manner in Catharanthus roseus. Plant Physiology 163:1792–1803
Cartharius K, Frech K, Grote K, Klocke B, Haltmeier M, Klingenhoff A, Frisch M, Bayerlein M, Werner T (2005) MatInspector and beyond: promoter analysis based on transcription factor binding sites. Bioinformatics 21:2933–2942
Chatel G, Montiel G, Pre M, Memelink J, Thiersault M, Saint-Pierre B, Doireau P, Gantet P (2003) CrMYC1, a Catharanthus roseus elicitor-and jasmonate-responsive bHLH transcription factor that binds the G-box element of the strictosidine synthase gene promoter. J Exp Bot 54:2587–2588
Chaturvedi Y, Tewari AK, Upadhyaya PP, Prabhuji SK, Rao GP (2009) Association of Candidatus phytoplasma asteris with little leaf and phyllody disease of Catharanthus roseus in Eastern Uttar Pradesh, India. Med Plants 1:103–108
Choi YH, Tapias EC, Kim HK, Lefeber AWM, Erkelens C, Verhoeven JTJ, Brzin J, Zel J, Verpoorte R (2004) Metabolic discrimination of Catharanthus roseus leaves infected by phytoplasma using 1H-NMR spectroscopy and multivariate data analysis. Plant Physiol 135:2398–2410
Deng S, Hiruki C (1991) Amplification of 16S rRNA genes from culturable and nonculturable mollicutes. J Microbiol Methods 14:53–61
Dutta A, Sen J, Deswal R (2007) Downregulation of terpenoid indole alkaloid biosynthetic pathway by low temperature and cloning of a AP2 type C-repeat binding factor (CBF) from Catharanthus roseus (L). G. Don. Plant Cell Reports 26:1869–1878
Dutta A, Sen J, Deswal R (2013) New evidences about strictosidine synthase (Str) regulation by salinity, cold stress and nitric oxide in Catharanthus roseus. J Plant Biochem Biotechnol 22:124–131
El-Sayed M, Verpoorte R (2007) Catharanthus terpenoid indole alkaloids: biosynthesis and regulation. Phytochem Rev 6:277–305
El-Sayed M, Verpoorte R (2004) Growth, metabolic profiling and enzymes activities of Catharanthus roseus seedlings treated with plant growth regulators. Plant Growth Regul 44:53–58
Favali MA, Musetti R, Benvenuti S, Bianchi A, Pressacco L (2004) Catharanthus roseus L. plants and explants infected with phytoplasmas: alkaloid production and structural observations. Protoplasma 223:45–51
Fragoso V, Rothe E, Baldwin IT, Kim SG (2014) Root jasmonates signaling regulates folivore-induced shoot metabolites and increases Nicotiana attenuata resistance. New Phytol 202:1335–1345
Fraser VN, Philmus B, Megraw M (2020) Metabolomics analysis reveals both plant variety and choice of hormone treatment modulate vinca alkaloid production in Catharanthus roseus. Plant Direct 4(9):e00267
Gasperini D, Chauvin A, Acosta IF, Kurenda A, Stolz S, Chetelat A, Wolfender JL, Farmer EE (2015) Axial and radial oxylipin transport. Plant Physiol 169:2244–2254
Geerlings A, Ibanez MML, Memelink J, van der Heijden R, Verpoorte R (2000) Molecular cloning and analysis of strictosidine β-D-glucosidase, an enzyme in terpenoid indole alkaloid biosynthesis in Catharanthus roseus. J Biol Chem 275:3051–3056
Gu XC, Chen JF, Xiao Y, Di P, Xuan HJ, Zhou X, Zhang L, Chen WS (2012) Overexpression of allene oxide cyclase promoted tanshinone/phenolic acid production in Salvia miltiorrhiza. Plant Cell Rep 31:2247–2259
Gundersen DE, Lee IM (1996) Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathol Mediterr 35:144–151
Guo X-R, Yang L, Yu J-H, Tang Z-H, Zu Y-G (2007) Alkaloid variations in Catharanthus roseus seedlings treated by different temperatures in short term and long term. J Forest Res 18:313–315
Gupta AK, Shukla AK, Singh P, Mall M, Yadav S, Shasany AK, Shanker K, Baskaran K, Sundaresan V, Talha M, Srivastava A, Gupta S, Khare P, Mani DN, Samad A (2020) CIM-Sushil: A high vindoline yielding variety of Catharanthus roseus. J Med Aromatic Plant Sci 42:51–63
Hause B, Stenzel I, Miersch O, Maucher H, Kramell R, Ziegler J, Wasternack C (2000) Tissue-specific oxylipin signature of tomato flowers: allene oxide cyclase is highly expressed in distinct flower organs and vascular bundles. Plant J 24:113–126
Hofmann E, Zerbe P, Schaller F (2006) The crystal structure of Arabidopsis thaliana allene oxide cyclase: insights into the oxylipin cyclization reaction. Plant Cell 18:3201–3217
Idrees M, Naeem M, Aftab T, Khan MAM, Moinuddin. (2011) Salicylic acid mitigates salinity stress by improving antioxidant defence system and enhances vincristine and vinblastine alkaloids production in periwinkle [Catharanthus roseus (L.) G. Don]. Acta Physiologia Plantarum 33:987–999
Jiang K, Pi Y, Hou R, Jiang L, Sun X, Tang K (2009) Promotion of nicotine biosynthesis in transgenic tobacco by overexpressing allene oxide cyclase from Hyoscyamus niger. Planta 229:1057–1063
Jaleel CA, Gopi R, Manivannan P, Panneerselvam R (2008) Soil salinity alters the morphology in Catharanthus roseus and its effects on endogenous mineral constituents. EurAsian J Biosci 2:18–25
Kamińska M, Sliwa H (2005) The reaction of Catharanthus roseus test plant to infection with two polish strains of aster yellows phytoplasma. Phytopathol Pol 37:23–32
Khanuja SPS, Shasany AK, Darokar MP, Kumar S (1999) Rapid isolation of DNA from dry and fresh samples of plants producing large amounts of secondary metabolites and essential oils. Plant Mol Biol Report 17:74
Kuboyama T, Yokoshima S, Tokuyama H, Fukuyama T (2004) Stereo controlled total synthesis of (+)-vincristine. Proc Natl Acad Sci USA 101:11966–11970
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327
Liu B, Wang W, Gao J, Chen F, Wang S, Xu Y, Tang L, Jia Y (2010) Molecular cloning and characterization of a jasmonate biosynthetic pathway gene for allene oxide cyclase from Jatropha curcas. Acta Physiol Plant 32:531–539
Lu X, Zhang F, Shen Q, Jiang W, Pan Q, Lv Z, Yan T, Fu X, Wang Y, Qian H, Tang K (2014) Overexpression of allene oxide cyclase improves the biosynthesis of artemisinin in Artemisia annua L. PLoS One 9:e91741
Mall M, Singh P, Kumar R, Shanker K, Gupta AK, Khare P, Shasany AK, Khatoon S, Sundaresan V, Baskaran K, Yadav S, Shukla AK (2021) Phenotypic, genetic and expression profiling of a vindoline-rich genotype of Catharanthus roseus. S Afr J Bot 139:50–57
Mall M, Verma RK, Gupta MM, Shasany AK, Khanuja SPS, Shukla AK (2019) Influence of seasonal and ontogenic parameters on the pattern of key terpenoid indole alkaloids biosynthesized in the leaves of Catharanthus roseus. S Afr J Bot 123:98–104
Menke FLH, Champion A, Kijne JW, Memelink J (1999a) A novel jasmonate- and elicitor-responsive element in the periwinkle secondary metabolite biosynthetic gene Str interacts with a jasmonate- and elicitor-inducible AP2-domain transcription factor, ORCA2. EMBO J 18:4455–4463
Menke FLH, Parchmann S, Mueller MJ, Kijne JW, Memelink J (1999b) Involvement of the octadecanoid pathway and protein phosphorylation in fungal elicitor-induced expression of terpenoid indole alkaloid biosynthesis genes in Catharanthus roseus. Plant Physiol 119:1289–1296
Pan Q, Chen Y, Wang Q, Yuan F, Xing S, Tian Y, Zhao J, Sun X, Tang K (2010) Effect of plant growth regulators on the biosynthesis of vinblastine, vindoline and catharanthine in Catharanthus roseus. Plant Growth Regul 60:133–141
Pandey SS, Singh S, Vivek Babu CS, Shanker K, Srivastava NK, Kalra A (2016a) Endophytes of opium poppy differentially modulate host plant productivity and genes for the biosynthetic pathway of benzylisoquinoline alkaloids. Planta 243:1097–1114
Pandey SS, Singh S, Vivek Babu CS, Shanker K, Srivastava NK, Shukla AK, Kalra A (2016b) Fungal endophytes of Catharanthus roseus enhance vindoline content by modulating structural and regulatory genes related to terpenoid indole alkaloid biosynthesis. Sci Rep 6:26583
Pi Y, Jiang K, Cao Y, Wang Q, Huang Z, Li L, Hu L, Li W, Sun X, Tang K (2009) Allene oxide cyclase from Camptotheca acuminata improves tolerance against low temperature and salt stress in tobacco and bacteria. Mol Biotechnol 41:115–122
Pi Y, Liao Z, Jiang K, Huang B, Deng Z, Zhao D, Zeng H, Sun X, Tang K (2008) Molecular cloning, characterization and expression of a jasmonate biosynthetic pathway gene encoding allene oxide cyclase from Camptotheca acuminata. Biosci Rep 28:349–355
Qu Y, Easson MLAE, Froese J, Simionescu R, Hudlicky T, De Luca V (2015) Completion of the seven-step pathway from tabersonine to the anticancer drug precursor vindoline and its assembly in yeast. Proc Natl Acad Sci USA 112:6224–6229
Raina SK, Wankhede DP, Jaggi M, Singh P, Jalmi SK, Raghuram B, Sheikh AH, Sinha AK (2012) CrMPK3, a mitogen activated protein kinase from Catharanthus roseus and its possible role in stress induced biosynthesis of monoterpenoid indole alkaloids. BMC Plant Biol 12:134
Raucher S, Bray BL (1985) Total synthesis of (±)-catharanthine. J Org Chem 50:3236–3237
Roepke J, Salim V, Wu M, Thamm AMK, Murata J, Ploss K, Boland W, de Luca V (2010) Vinca drug components accumulate exclusively in leaf exudates of Madagascar periwinkle. Proceedings of the National Academy of Sciences of the USA 107:15287–15292
Samad A, Ajayakumar PV, Gupta MK, Shukla AK, Darokar MP, Somkuwar B, Alam M (2008) Natural infection of periwinkle (Catharanthus roseus) with Cucumber mosaic virus, subgroup IB. Aust Plant Dis Notes 3:30–34
Samad A, Dharni S, Singh M, Yadav S, Khan A, Shukla AK (2011a) First report of a natural infection of Stevia rebaudiana by a group 16SrXXIV phytoplasma in India. Plant Dis 95:1582
Samad A, Panda S, Gupta MK, Ajayakumar PV, Shukla AK (2011b) Molecular identification of a phytoplasma associated with Ajwain (Trachyspermum ammi) in India. Eur J Plant Pathol 130:1–4
Schluttenhofer C, Pattanaik S, Patra B, Yuan L (2014) Analyses of Catharanthus roseus and Arabidopsis thaliana WRKY transcription factors reveal involvement in jasmonate signaling. BMC Genomics 15:502
Sears JE, Barker TJ, Boger DL (2015) Total synthesis of (-)-vindoline and (+)-4-epi-vindoline based on a 1,3,4-oxadiazole tandem intramolecular [4+2]/[3+2] cycloaddition cascade initiated by an allene dienophile. Org Lett 17:5460–5463
Shukla, A.K., & Khanuja, S.P.S. (2013). Catharanthus roseus: the metabolome that represents a unique reservoir of medicinally important alkaloids under precise genomic regulation. In: OMICS Applications in Crop Science (ed D. Barh), pp. 325–384. CRC Press (Taylor & Francis Group), Boca Raton.
Shukla AK, Shasany AK, Gupta MM, Khanuja SPS (2006) Transcriptome analysis in Catharanthus roseus leaves and roots for comparative terpenoid indole alkaloid profiles. J Exp Bot 57:3921–3932
Shukla AK, Shasany AK, Khanuja SPS (2005) Isolation of poly (A)+ mRNA for downstream reactions from some medicinal and aromatic plants. Indian J Exp Biol 43:197–201
Shukla AK, Shasany AK, Verma RK, Gupta MM, Mathur AK, Khanuja SPS (2010) Influence of cellular differentiation and elicitation on intermediate and late steps of terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Protoplasma 242:35–47
Shukla YN, Rani A, Kumar S (1997) Effect of temperature and pH on the extraction of total alkaloids from Catharanthus roseus leaves. J Medi Aromatic Plant Sci 19:430–431
Soltani N, Nazarian-Firouzabadi F, Shafeinia A, Sadr AS, Shirali M (2020) The expression of Terpenoid Indole Alkaloid (TIAs) pathway genes in Catharanthus roseus in response to salicylic acid treatment. Mol Biol Rep 47:7009–7016
Srivastava A, Chandra G, Raj SK (2004) Molecular characterization of a strain of cucumber mosaic virus based on coat protein and movement protein genes. Acta Virol 48:229–239
Srivastava S, Pandey R, Kumar S, Nautiyal CS (2014) Correspondence between flowers and leaves in terpenoid indole alkaloid metabolism of the phytoplasma-infected Catharanthus roseus plants. Protoplasma 251:1307–1320
Stenzel I, Hause B, Miersch O, Kurz T, Maucher H, Weichert H, Ziegler J, Feussner I, Wasternack C (2003) Jasmonate biosynthesis and the allene oxide cyclase family of Arabidopsis thaliana. Plant Mol Biol 51:895–911
Stenzel I, Otto M, Delker C, Kirmse N, Schmidt D, Miersch O, Hause B, Wasternack C (2012) ALLENE OXIDE CYCLASE (AOC) gene family members of Arabidopsis thaliana: tissue- and organ-specific promoter activities and in vivo heteromerization. J Exp Bot 63:6125–6138
Tiwari R, Awasthi A, Mall M, Shukla AK, Satya Srinivas KVN, Syamasundar KV, Kalra A (2013) Bacterial endophyte-mediated enhancement of in planta content of key terpenoid indole alkaloids and growth parameters of Catharanthus roseus. Ind Crops Prod 43:306–310
van der Fits L, Memelink J (2000) ORCA3, a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 289:295–297
van der Fits L, Memelink J (2001) The jasmonate-inducible AP2/ERF-domain transcription factor ORCA3 activates gene expression via interaction with a jasmonate-responsive promoter element. Plant J 25:43–53
Vazquez-Flota FA, De Luca V (1998) Jasmonate modulates development- and light-regulated alkaloid biosynthesis in Catharanthus roseus. Phytochemistry 49:395–402
Verma, M., Ghangal, R., Sharma, R., Sinha, A.K., & Jain, M. (2014). Transcriptome analysis of Catharanthus roseus for gene discovery and expression profiling. PLoS ONE, 9, e103583.
Wang J-Y, Liu Z-P (2010) Alkaloid accumulation in Catharanthus roseus increases with addition of seawater salts to the nutrient solution. Pedosphere 20:718–724
Wang J-Y, Liu Z-P, Liu L, Liu C (2008) Effects of NaCl on the growth and alkaloid content of Catharanthus roseus seedlings]. [Article in Chinese. Ying Yong Sheng Tai Xue Bao 19:2143–2148
Wei S (2010) Methyl jasmonic acid induced expression pattern of terpenoid indole alkaloid pathway genes in Catharanthus roseus seedlings. Plant Growth Regul 61:243–251
Wu Q, Wu J, Sun H, Zhang D, Yu D (2011) Sequence and expression divergence of the AOC gene family in soybean: insights into functional diversity for stress responses. Biotech Lett 33:1351–1359
Yokoshima S, Ueda T, Kobayashi S, Sato A, Kuboyama T, Tokuyama H, Fukuyama T (2002) Stereocontrolled total synthesis of (+)-vinblastine. J Am Chem Soc 124:2137–2139
Acknowledgements
The authors are grateful to Director, CSIR-CIMAP, for encouragement and providing facilities for conducting the experiments. They are also thankful to Dr. Anil K. Gupta, Senior Principal Scientist, CSIR-CIMAP and Curator, National Gene Bank for Medicinal and Aromatic Plants at CSIR-CIMAP, Lucknow, for his help and cooperation.
Funding
This study was financially supported by a grant (EMR/2016/002026) from the Science and Engineering Research Board (SERB), India. MM was supported by a UGC Research Fellowship. This is CIMAP Communication No.: CIMAP/PUB/2020/OCT/108.
Author information
Authors and Affiliations
Contributions
MM carried out the experiments, recorded the observations and analyzed the data. KS carried out the chemical analysis for vindoline. AS identified the phytoplasma and CMV infections on the plant as well as helped in their characterization. AK contributed the endophytes and helped in the endophyte-related experiments. VS helped in data analysis. AKS conceived and planned the study and interpreted the data. MM and AKS prepared the manuscript that was read and approved by all the authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Handling Editor: Peter Nick
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mall, M., Shanker, K., Samad, A. et al. Stress responsiveness of vindoline accumulation in Catharanthus roseus leaves is mediated through co-expression of allene oxide cyclase with pathway genes. Protoplasma 259, 755–773 (2022). https://doi.org/10.1007/s00709-021-01701-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-021-01701-6