Abstract
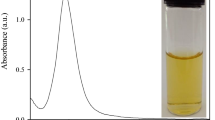
We report on a colorimetric probe for the determination of Pb(II). It is based on the use of silver nanoparticles that have been functionalizd with iminodiacetic acid (IDA-Ag NPs). The absorption spectrum and solution color of IDA-Ag NPs undergo dramatic changes on exposure to Pb(II) with a new absorption peak appearing at 650 nm and a concomitant color change from yellow to green. This is assumed to result from the aggregation of IDA-Ag NPs induced by Pb(II). Under optimum conditions, there is a linear relationship between the ratio of the absorbances at 650 and 396 nm, respectively, and the concentration of Pb(II) in the 0.4 to 8.0 μM concentration range, with a detection limit of 13 nM. The method was applied to the determination of Pb(II) in tap water and urea samples, and recoveries ranged from 93.7 % to 98.6 %.

A colorimetric probe based on iminodiacetic acid-functionalized silver nanoparticles (IDA-Ag NPs) was obtained and used for determination of Pb2+. The color change from yellow to green was assumed to result from the aggregation of the NPs induced by Pb(II) ions. The assay was possessed highly selectivity to lead(II) over the other ions.






Similar content being viewed by others
References
Lanphear BP (1998) The paradox of lead poisoning prevention. Science 281:1617
Lanphear BP, Roghmann KJ (1997) Pathways of lead exposure in urban children. Environ Res 74:67
Strużyńska L, Walski M, Gadamski R, Dabrowska-Bouta B, Rafałowska U (1997) Lead-induced abnormalities in blood–brain barrier permeability in experimental chronic toxicity. Mol Chem Neuropathol 31:207
Rifai N, Cohen G, Wolf M, Cohen L, Faser C, Savory J, DePalma L (1993) Incidence of lead poisoning in young children from inner-city, suburban, and rural communities. Ther Drug Monit 15:71
Needleman H (2004) Lead poisoning. Annu Rev Med 55:209
Soylak M, Cay RS (2007) Separation/preconcentration of silver(I) and lead(II) in environmental samples on cellulose nitrate membrane filter prior to their flame atomic absorption spectrometric determinations. J Hazard Mater 146:142
MdGA K, de Andrade JB, de Jesus DS, Lemos VA, Bandeira MLSF, dos Santos WNL, Bezerra MA, Amorim FAC, Souza AS, Ferreira SLC (2006) Separation and preconcentration procedures for the determination of lead using spectrometric techniques: a review. Talanta 69:16
Liu J, Lu Y (2006) Preparation of aptamer-linked gold nanoparticle purple aggregates for colorimetric sensing of analytes. Nat Protoc 1:246
Chai F, Wang C, Wang T, Li L, Su Z (2010) Colorimetric detection of Pb2+ using glutathione functionalized gold nanoparticles. ACS Appl Mater Inter 2:1466
Xue Y, Zhao H, Wu Z, Li X, He Y, Yuan Z (2011) Colorimetric detection of Cd2+ using gold nanoparticles cofunctionalized with 6-mercaptonicotinic acid and l-cysteine. Analyst 136:3725
Kim S, Kim J, Lee NH, Jang HH, Han MS (2011) A colorimetric selective sensing probe for calcium ions with tunable dynamic ranges using cytidine triphosphate stabilized gold nanoparticles. Chem Commun 47:10299
Zhang Y, Li B, Chen X (2010) Simple and sensitive detection of dopamine in the presence of high concentration of ascorbic acid using gold nanoparticles as colorimetric probes. Microchim Acta 168:107
Lin YW, Huang CC, Chang HT (2011) Gold nanoparticle probes for the detection of mercury, lead and copper ions. Analyst 136:863
Ai K, Liu Y, Lu L (2009) Hydrogen-bonding recognition-induced color change of gold nanoparticles for visual detection of melamine in raw milk and infant formula. J Am Chem Soc 131:9496
Tan F, Liu X, Quan X, Chen J, Li X, Zhao H (2011) Selective detection of nanomolar Cr(VI) in aqueous solution based on 1,4-dithiothreitol functionalized gold nanoparticles. Anal Method 3:343
Sun J, Ge J, Liu W, Fan Z, Zhang H, Wang P (2011) Highly sensitive and selective colorimetric visualization of streptomycin in raw milk using Au nanoparticles supramolecular assembly. Chem Commun 47:9888
Han C, Zhang L, Li H (2009) Highly selective and sensitive colorimetric probes for Yb3+ ions based on supramolecular aggregates assembled from β-cyclodextrin-4,4′-dipyridine inclusion complex modified silver nanoparticles. Chem Commun 45:3545
Zhang F, Zeng L, Zhang Y, Wang H, Wu A (2011) A colorimetric assay method for Co2+ based on thioglycolic acid functionalized hexadecyl trimethyl ammonium bromide modified Au nanoparticles (NPs). Nanoscale 3:2150
Xu Y, Deng L, Wang H, Ouyang X, Zheng J, Li J, Yang R (2011) Metal-induced aggregation of mononucleotides-stabilized gold nanoparticles: an efficient approach for simple and rapid colorimetric detection of Hg(II). Chem Commun 47:6039
Chansuvarn W, Imyim A (2012) Visual and colorimetric detection of mercury(II) ion using gold nanoparticles stabilized with a dithia-diaza ligand. Microchim Acta 1:57
Li HB, Cui ZM, Han CP (2009) Glutathione-stabilized silver nanoparticles as colorimetric sensor for Ni2+ ion. Sensor Actuat B-Chem 143:87
Li HB, Zhang LA, Yao Y, Han CP, Jin S (2010) Synthesis of aza-crown ether-modified silver nanoparticles as colorimetric sensors for Ba2+. Supramol Chem 22:544
Cao Q, Zhao H, He Y, Li X, Zeng L, Ding N, Wang J, Yang J, Wang G (2010) Hydrogen-bonding-induced colorimetric detection of melamine by nonaggregation-based Au-NPs as a probe. Biosens Bioelectron 25:2680
Kanayama N, Takarada T, Maeda M (2011) Rapid naked-eye detection of mercury ions based on non-crosslinking aggregation of double-stranded DNA-carrying gold nanoparticles. Chem Commun 47:2077
Beqa L, Singh AK, Khan SA, Senapati D, Arumugam SR, Ray PC (2011) Gold nanoparticle-based simple colorimetric and ultrasensitive dynamic light scattering assay for the selective detection of Pb(II) from paints, plastics, and water samples. ACS Appl Mater Inter 3:668
Zhang BH, Qi L, Wu FY (2010) Functionalized manganese-doped zinc sulfide core/shell quantum dots as selective fluorescent chemodosimeters for silver ion. Microchim Acta 170:147
Li HB, Li FY, Han CP, Cui ZM, Xie GY, Zhang AQ (2010) Highly sensitive and selective tryptophan colorimetric sensor based on 4,4-bipyridine-functionalized silver nanoparticles. Sensor Actuat B-Chem 145:194
Su YH, Chang SH, Teoh LG, Chu WH, Tu SL (2009) Plasmons: chemical bonding coupling induced surface plasmon resonance splitting in self-assembled gold nanoparticles. J Phys Chem C 113:3923
Yusa S, Fukuda K, Yamamoto T, Iwasaki Y, Watanabe A, Akiyoshi K, Morishima Y (2007) Salt effect on the heat-induced association behavior of gold nanoparticles coated with poly(N-isopropylacrylamide) prepared via reversible addition--fragmentation chain transfer (RAFT) radical polymerization. Langmuir 23:12842
Bjerrum J, Schwarzenbach G (1958) Stability constants, Chem Soc, London
Hung YL, Hsiung TM, Chen YY, Huang YF, Huang CC (2010) Colorimetric detection of heavy metal ions using label-free gold nanoparticles and alkanethiols. J Phys Chem C 114:16329
Zhu D, Li X, Liu X, Wang J, Wang Z (2012) Designing bifunctionalized gold nanoparticle for colorimetric detection of Pb2+ under physiological condition. Biosens Bioelectron 31:505
Liu J, Lu Y (2003) A colorimetric lead biosensor using DNAzyme-directed assembly of gold nanoparticles. J Am Chem Soc 125:6642
Kim IB, Dunkhorst A, Gilbert J, Bunz UHF (2005) Sensing of lead ions by a carboxylate-substituted PPE: multivalency effects. Macromolecules 38:4560
Rabenstein DL, Fairhurst MT (1975) Nuclear magnetic resonance studies of the solution chemistry of metal complexes. XI. Binding of methylmercury by sulfhydryl-containing amino acids and by glutathione. J Am Chem Soc 97:2086
Wu SP, Chen YP, Sung YM (2011) Colorimetric detection of Fe3+ ions using pyrophosphate functionalized gold nanoparticles. Analyst 136:1887
Zhang J, Wang Y, Xu X, Yang X (2011) Specifically colorimetric recognition of calcium, strontium, and barium ions using 2-mercaptosuccinic acid-functionalized gold nanoparticles and its use in reliable detection of calcium ion in water. Analyst 136:3865
Acknowledgements
Financial supports from Natural Science Foundation of China (no. 20965006) are highly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 321 kb)
Rights and permissions
About this article
Cite this article
Qi, L., Shang, Y. & Wu, F. Colorimetric detection of lead (II) based on silver nanoparticles capped with iminodiacetic acid. Microchim Acta 178, 221–227 (2012). https://doi.org/10.1007/s00604-012-0832-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-012-0832-3