Abstract
Purpose
To identify the risk factors for apical node metastasis in colorectal cancer (CRC) patients who underwent laparoscopic radical resection with D3 lymphadenectomy.
Methods
The subjects were 578 consecutive patients who underwent laparoscopic radical resection with D3 lymphadenectomy for CRC between February, 2003 and December, 2009. The Cox proportional regression model was used to evaluate the association between apical node metastasis and survival and the logistic regression model was used to identify the risk factors for apical node metastasis.
Results
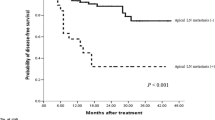
Thirty patients (5.2 %) had apical node metastasis, which was an independent risk factor for poor disease-free survival [hazard ratio (HR) = 3.02, P = 0.019]. Multivariate logistic regression analysis revealed that poor differentiation [odds ratio (OR) = 4.58, P < 0.001] and ulcerative/infiltrative morphology (OR = 5.57, P = 0.023) were independent risk factors for apical node metastasis. In the subgroup analysis based on pT3–4 tumors, multivariate logistic regression analysis also suggested that these factors were significantly associated with apical node metastasis.
Conclusions
Apical node metastasis is an independent risk factor for poor oncologic outcome. T3–4 stage, poor differentiation, and ulcerative/infiltrative morphology are significantly associated with apical node metastasis. Notably, in this study, T1–2 stage tumors had no apical node metastasis, suggesting that D3 lymphadenectomy may be over-performed without considering the T stage of tumors.
Similar content being viewed by others
References
Gervasoni JE Jr, Taneja C, Chung MA, Cady B. Biologic and clinical significance of lymphadenectomy. Surg Clin N Am. 2000;80:1631–73.
Kawamura YJ, Umetani N, Sunami E, Watanabe T, Masaki T, Muto T. Effect of high ligation on the long-term result of patients with operable colon cancer, particularly those with limited nodal involvement. Eur J Surg. 2000;166:803–7.
Titu LV, Tweedle E, Rooney PS. High tie of the inferior mesenteric artery in curative surgery for left colonic and rectal cancers: a systematic review. Dig Surg. 2008;25:148–57.
Hida J, Okuno K. High ligation of the inferior mesenteric artery in rectal cancer surgery. Surg Today. 2013;43:8–19.
Nelson H, Petrelli N, Carlin A, Couture J, Fleshman J, Guillem J, et al. Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst. 2001;93:583–96.
Mammen JM, James LE, Molloy M, Williams A, Wray CJ, Sussman JJ. The relationship of lymph node dissection and colon cancer survival in the veterans affairs central cancer registry. Am J Surg. 2007;194:349–54.
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation—technical notes and outcome. Colorectal Dis. 2009;11:354–64 (discussion 364–355).
Kanemitsu Y, Hirai T, Komori K, Kato T. Survival benefit of high ligation of the inferior mesenteric artery in sigmoid colon or rectal cancer surgery. Br J Surg. 2006;93:609–15.
Slanetz CA Jr, Grimson R. Effect of high and intermediate ligation on survival and recurrence rates following curative resection of colorectal cancer. Dis Colon Rectum. 1997;40:1205–18 (discussion 1218–1209).
Malassagne B, Valleur P, Serra J, Sarnacki S, Galian A, Hoang C, et al. Relationship of apical lymph node involvement to survival in resected colon carcinoma. Dis Colon Rectum. 1993;36:645–53.
Kim JC, Lee KH, Yu CS, Kim HC, Kim JR, Chang HM, et al. The clinicopathological significance of inferior mesenteric lymph node metastasis in colorectal cancer. Eur J Surg Oncol. 2004;30:271–9.
Ang CW, Tweedle EM, Campbell F, Rooney PS. Apical node metastasis independently predicts poor survival in Dukes C colorectal cancer. Colorectal Dis. 2011;13:526–31.
Huh JW, Kim YJ, Kim HR. Distribution of lymph node metastases is an independent predictor of survival for sigmoid colon and rectal cancer. Ann Surg. 2012;255:70–8.
Yi JW, Lee TG, Lee HS, Heo SC, Jeong SY, Park KJ, et al. Apical-node metastasis in sigmoid colon or rectal cancer: is it a factor that indicates a poor prognosis after high ligation? Int J Colorectal Dis. 2012;27:81–7.
Liang YZ, Yu J, Zhang C, Wang YN, Cheng X, Huang F, et al. Construction and application of evaluation system of laparoscopic colorectal surgery based on clinical data mining. Zhonghua Wei Chang Wai Ke Za Zhi. 2010;13:741–4.
Koyama Y, Kotake K. Overview of colorectal cancer in Japan: report from the registry of the Japanese Society for Cancer of the Colon and Rectum. Dis Colon Rectum. 1997;40:S2–9.
Washington MK, Berlin J, Branton PA, Burgart LJ, Carter DK, Fitzgibbons PL, et al. Protocol for the examination of specimens from patients with primary carcinomas of the colon and rectum. Arch Pathol Lab Med. 2008;132:1182–93.
Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471–4.
Hida J, Okuno K, Yasutomi M, Yoshifuji T, Uchida T, Tokoro T, et al. Optimal ligation level of the primary feeding artery and bowel resection margin in colon cancer surgery: the influence of the site of the primary feeding artery. Dis Colon Rectum. 2005;48:2232–7.
Chin CC, Yeh CY, Tang R, Changchien CR, Huang WS, Wang JY. The oncologic benefit of high ligation of the inferior mesenteric artery in the surgical treatment of rectal or sigmoid colon cancer. Int J Colorectal Dis. 2008;23:783–8.
Liang JT, Huang KC, Lai HS, Lee PH, Sun CT. Oncologic results of laparoscopic D3 lymphadenectomy for male sigmoid and upper rectal cancer with clinically positive lymph nodes. Ann Surg Oncol. 2007;14:1980–90.
Alici A, Kement M, Gezen C, Akin T, Vural S, Okkabaz N, et al. Apical lymph nodes at the root of the inferior mesenteric artery in distal colorectal cancer: an analysis of the risk of tumor involvement and the impact of high ligation on anastomotic integrity. Tech Coloproctol. 2010;14:1–8.
Kang J, Hur H, Min BS, Kim NK, Lee KY. Prognostic impact of inferior mesenteric artery lymph node metastasis in colorectal cancer. Ann Surg Oncol. 2011;18:704–10.
Yamauchi H, Togashi K, Kawamura YJ, Horie H, Sasaki J, Tsujinaka S, et al. Pathological predictors for lymph node metastasis in T1 colorectal cancer. Surg Today. 2008;38:905–10.
Glasgow SC, Bleier JI, Burgart LJ, Finne CO, Lowry AC. Meta-analysis of histopathological features of primary colorectal cancers that predict lymph node metastases. J Gastrointest Surg. 2012;16:1019–28.
Chang HC, Huang SC, Chen JS, Tang R, Changchien CR, Chiang JM, et al. Risk factors for lymph node metastasis in pT1 and pT2 rectal cancer: a single-institute experience in 943 patients and literature review. Ann Surg Oncol. 2012;19:2477–84.
Choi PW, Yu CS, Jang SJ, Jung SH, Kim HC, Kim JC. Risk factors for lymph node metastasis in submucosal invasive colorectal cancer. World J Surg. 2008;32:2089–94.
Seike K, Koda K, Saito N, Oda K, Kosugi C, Shimizu K, et al. Laser Doppler assessment of the influence of division at the root of the inferior mesenteric artery on anastomotic blood flow in rectosigmoid cancer surgery. Int J Colorectal Dis. 2007;22:689–97.
Watanabe T, Itabashi M, Shimada Y, Tanaka S, Ito Y, Ajioka Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17:1–29.
Huh JW, Kim HR, Kim YJ. Lymphovascular or perineural invasion may predict lymph node metastasis in patients with T1 and T2 colorectal cancer. J Gastrointest Surg. 2010;14:1074–80.
Chok KS, Law WL. Prognostic factors affecting survival and recurrence of patients with pT1 and pT2 colorectal cancer. World J Surg. 2007;31:1485–90.
Gleeson FC, Clain JE, Papachristou GI, Rajan E, Topazian MD, Wang KK, et al. Prospective assessment of EUS criteria for lymphadenopathy associated with rectal cancer. Gastrointest Endosc. 2009;69:896–903.
Schwarz RE. Factors influencing change of preoperative treatment intent in a gastrointestinal cancer practice. World J Surg Oncol. 2007;5:32.
Sample CB, Watson M, Okrainec A, Gupta R, Birch D, Anvari M. Long-term outcomes of laparoscopic surgery for colorectal cancer. Surg Endosc. 2006;20:30–4.
Scala A, Huang A, Dowson HM, Rockall TA. Laparoscopic colorectal surgery—results from 200 patients. Colorectal Dis. 2007;9:701–5.
Neudecker J, Klein F, Bittner R, Carus T, Stroux A, Schwenk W, et al. Short-term outcomes from a prospective randomized trial comparing laparoscopic and open surgery for colorectal cancer. Br J Surg. 2009;96:1458–67.
Acknowledgments
We gratefully acknowledge the financial support for this project form the National Key Technology R&D Program (Grant Number: 2013BAI05B00), the Program of the National key Clinical Medical Specialty, the Major Program of Science and Technology Program of Guangzhou (Grant Number: 201300000087), and Research Fund of Public welfare in Health Industry (Grant Number: 201402015), 2014, Health Ministry of China.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding authors
Additional information
H. Chen and Y. Wang contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Chen, H., Wang, Y., Liu, H. et al. Factors influencing apical node metastasis in colorectal cancer patients treated with laparoscopic radical resection with D3 lymphadenectomy: results from two centers in China. Surg Today 45, 569–575 (2015). https://doi.org/10.1007/s00595-014-1013-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-014-1013-2