Abstract
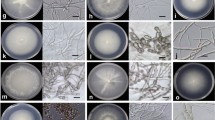
Orchid conservation efforts, using seeds and species-specific fungi that support seed germination, require the isolation, identification, and germination enhancement testing of symbiotic fungi. However, few studies have focused on developing such techniques for the epiphytes that constitute the majority of orchids. In this study, conducted in Xishuangbanna Tropical Botanical Garden, Yunnan, China, we used seeds of Dendrobium aphyllum, a locally endangered and medicinally valuable epiphytic orchid, to attract germination promoting fungi. Of the two fungi isolated from seed baiting, Tulasnella spp. and Trichoderma spp., Tulasnella, enhanced seed germination by 13.6 %, protocorm formation by 85.7 %, and seedling development by 45.2 % (all P < 0.0001). Epulorhiza, another seed germination promoting fungi isolated from Cymbidium mannii, also enhanced seed germination (6.5 %; P < 0.05) and protocorm formation (20.3 %; P < 0.0001), but Trichoderma suppressed seed germination by 26.4 % (P < 0.0001). Tulasnella was the only treatment that produced seedlings. Light increased seed imbibition, protocorm formation, and two-leaved seed development of Tulasnella inoculated seeds (P < 0.0001). Because the germination stage success was not dependent on fungi, we recommend that Tulasnella be introduced for facilitating D. aphyllum seed germination at the protocorm formation stage and that light be provided for increasing germination as well as further seedling development. Our findings suggest that in situ seed baiting can be used to isolate seed germination-enhancing fungi for the development of seedling production for conservation and reintroduction efforts of epiphytic orchids such as D. aphyllum.




Similar content being viewed by others
References
Arditti J (1967) Factors affecting the germination of orchid seeds. Bot Rev 33:1–97
Arditti J (1992) Fundamentals of orchid biology. Wiley, New York
Arditti J, Ernst R (1984) Physiology of germinating orchid seeds. In: Arditti J (ed) Orchid biology, reviews and perspectives, 3rd edn. Cornell University Press, New York, pp 170–222
Arditti J, Ghani AKA (2000) Numerical and physical properties of orchid seeds and their biological implications. New Phytol 145:367–421
Atwood JT (1986) The size of the Orchidaceae and the systematic distribution of epiphytic orchids. Selbyana 9:171–186
Bao XS, Shun QS, Chen LZ (2001) The medicinal plants of Dendrobium (Shi-Hu) in China, a coloured atlas. Fudan University Press, Shanghai (in Chinese)
Batty AL, Dixon KW, Brundrett M, Sivasithamparam K (2001) Constraints to symbiotic germination of terrestrial orchid seed in a Mediterranean bushland. New Phytol 152:511–520
Batty AL, Brundrett MC, Dixon KW, Sivasithamparam K (2006) In situ symbiotic seed germination and propagation of terrestrial orchid seedlings for establishment at field sites. Aust J Bot 54:375–381
Bidartondo MI, Read DJ (2008) Fungal specificity bottlenecks during orchid germination and development. Mol Ecol 17:3707–3716
Bretz F, Hothorn T, Westfall P (2010) Multiple comparisons using R. CRC, Boca Raton
Brundrett MC, Scade A, Batty AL, Dixon KW, Sivasithamparam K (2003) Development of in situ and ex situ seed baiting techniques to detect mycorrhizal fungi from terrestrial orchid habitats. Mycol Res 107:1210–1220
Catalano V, Vergara M, Hauzenberger JR, Seiboth B, Sarrocco S, Vannacci G, Kubicek CP, Seidl-Seiboth V (2011) Use of a non-homologous end-joining-deficient strain (delta-ku70) of the biocontrol fungus Trichoderma virens to investigate the function of the laccase gene lcc1 in sclerotia degradation. Curr Genet 57:13–23
Chen XQ (1999) Orchidaceae. In: Flora of China 19. Science Press, Beijing, pp 106–108, in Chinese
Chen XM, Guo SX (2005) Effects of four species of endophytic fungi on the growth and polysaccharide and alkaloid contents of Dendrobium nobile. Zhongguo Zhong Yao Za Zhi 30:253 (in Chinese)
Chen J, Wang H, Guo SX (2012) Isolation and identification of endophytic and mycorrhizal fungi from seeds and roots of Dendrobium (Orchidaceae). Mycorrhiza 22:297–307
Chutima R, Dell B, Lumyong S (2011) Effects of mycorrhizal fungi on symbiotic seed germination of Pecteilis susannae (L.) Rafin (Orchidaceae), a terrestrial orchid in Thailand. Symbiosis 53:149–156
Cribb PJ, Kell SP, Dixon KW, Barrett RL (2003) Orchid conservation: a global perspective. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ (eds) Orchid conservation. Natural History Publications, Kota Kinabalu, pp 1–24
Dearnaley JDW (2007) Further advances in orchid mycorrhizal research. Mycorrhiza 17:475–486
Dearnaley JDW, Martos F, Selosse MA (2012) Orchid Mycorrhizas: molecular ecology, physiology, evolution and conservation aspects. In: Hock B (ed) Fungal associations. Springer, Berlin, pp 207–230
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15 obtained from http://irc.igd.cornell.edu/Protocols/DoyleProtocol.pdf. Accessed 23 January 2011
Dressler RL (1993) Phylogeny and classification of the orchid family. Cambridge University Press, New York
Duan CF, Li ZL, Fang F, Yang GH, Chen SS, Hong QY (2010) Isolation and identification research of mycorrhizal isolated from several orchids in Yunnan Province. Xi Nan Nong Ye Xue Bao 23:756–759, in Chinese
Eriksson O, Kainulainen K (2011) The evolutionary ecology of dust seeds. Perspect Plant Ecol Evol Syst 13:73–87
Fu LK (1992) China plant red data book: rare and endangered plants. I. Science Press, Beijing (in Chinese)
Gao JY, Liu Q, Yu DL (2014) Orchids in Xishuangbanna: diversity and conservation. China Forestry Publishing House, Beijing (in Chinese)
Godo T, Komori M, Nakaoki E, Yukawa T, Miyoshi K (2010) Germination of mature seeds of Calanthe tricarinata Lindl., an endangered terrestrial orchid, by asymbiotic culture in vitro. In Vitro Cell Dev Biol Plant 46:323–328
Hágsater E, Dumont V (1996) Conservation threats. In: Prindgeon AM (ed) Orchids: status, survey, and conservation action plan. IUCN, Gland, pp 6–9
Harley JL (1950) Recent progress in the study of endotrophic mycorrhiza. New Phytol 49:213–247
Harvais G (1973) Growth requirements and development of Cypripedium reginae in axenic culture. Can J Bot 51(2):327–332
Herre EA, Mejía LC, Kyllo DA, Rojas E, Maynard Z, Butler A, Van Bael SA (2007) Ecological implications of anti-pathogen effects of tropical fungal endophytes and mycorrhizae. Ecology 88:550–558
Hollick PS (2004) Mycorrhizal specificity in endemic Western Australian terrestrial orchids (tribe Diurideae): implications for conservation. Doctoral dissertation, Murdoch University
Hou XQ, Guo SX (2009) Interaction between a dark septate endophytic isolate from Dendrobium sp. and roots of D. nobile seedlings. J Integr Plant Biol 51:374–381
Huang L, He XR, Zheng LM, Cai J (2004) Preliminary studies on mycorrhizal fungi in promoting the growth of orchid seedlings from tissue culture. Re Dai Zuo Wu Xue Bao 25:36–38 (in Chinese)
Johnson TR, Stewart SL, Dutra D, Kane ME, Richardson L (2007) Asymbiotic and symbiotic seed germination of Eulophia alta (Orchidaceae)—preliminary evidence for the symbiotic culture advantage. Plant Cell Tissue Organ Cult 90:313–323
Kartzinel TR, Trapnell DW, Shefferson RP (2013) Highly diverse and spatially heterogeneous mycorrhizal symbiosis in a rare epiphyte is unrelated to broad biogeographic or environmental features. Mol Ecol. doi:10.1111/mec.12536
Kauth PJ, Vendrame WA, Kane ME (2006) In vitro seed culture and seedling development of Calopogon tuberosus. Plant Cell Tissue Organ Cult 85:91–102
Keel BG, Zettler LW, Kaplin BA (2011) Seed germination of Habenaria repens (Orchidaceae) in situ beyond its range, and its potential for assisted migration imposed by climate change. Castanea 76:43–54
Knudson L (1922) Nonsymbiotic germination of orchid seeds. Bot Gaz 73:1–25
Koopowitz H, Hawkins BA (2012) Global climate change is confounding species conservation strategies. Integr Zool 7:158–164
Li H, Ma Y, Liu W, Liu W (2009) Clearance and fragmentation of tropical rain forest in Xishuangbanna, SW, China. Biodivers Conserv 18:3421–3440
Liu H, Luo Y, Liu H (2010) Studies of mycorrhizal fungi of Chinese orchids and their role in orchid conservation in China-a review. Bot Rev 76:241–262
Massey EE, Zettler LW (2007) An expanded role for in vitro symbiotic seed germination as a conservation tool: two case studies in North America (Platanthera leucophaea and Epidendrum nocturnum). Lankesteriana 7:303–308
Masuhara G, Katsuya K (1994) In situ and in vitro specificity between Rhizoctonia spp. and Spiranthes sinensis (Persoon) Ames, var. amoena (M. Bieberstein) Hara (Orchidaceae). New Phytol 127:711–718
McCormick MK, Whigham DF, Sloan D, O'Malley K, Hodkinson B (2006) Orchid-fungus fidelity: a marriage meant to last? Ecology 87:903–911
McCormick MK, Lee Taylor D, Juhaszova K, Brunett RK, Whigham DF, O'Neill JP (2012) Limitations on orchid recruitment: not a simple picture. Mol Ecol 21:1511–1523
Moore RT (1987) The genera of Rhizoctonia-like fungi: Ascorhizoctonia, Ceratorhiza gen. nov., Epulorhiza gen. nov., Moniliopsis, and Rhizoctonia. Mycotaxon 29:91–99
Nontachaiyapoom S, Sasirat S, Manoch L (2010) Isolation and identification of Rhizoctonia-like fungi from roots of three orchid genera, Paphiopedilum, Dendrobium, and Cymbidium, collected in Chiang Rai and Chiang Mai provinces of Thailand. Mycorrhiza 20:459–471
Otero JT, Ackerman JD, Bayman P (2002) Diversity and host specificity of endophytic Rhizoctonia-like fungi from tropical orchids. Am J Bot 89:1852–1858
Park EJ, Lee WY (2013) In vitro symbiotic germination of myco-heterotrophic Gastrodia elata by Mycena species. Plant Biotechnol Rep 7:185–191
Perkins AJ, McGee PA (1995) Distribution of the orchid mycorrhizal fungus, Rhizoctonia solani, in relation to its host, Pterostylis acuminata, in the field. Aust J Bot 43:565–575
Phillips RD, Barrett MD, Dixon KW, Hopper SD (2011) Do mycorrhizal symbioses cause rarity in orchids? J Ecol 99:585–869
R Development Core Team (2011) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/. Accessed 30 June 2013
Rasmussen HN (2002) Recent developments in the study of orchid mycorrhiza. Plant Soil 244:149–163
Rasmussen HN, Rasmussen FPN (1991) Climatic and seasonal regulation of seed plant establishment in Dactylorhiza majalis inferred from symbiotic experiments in vitro. Lindleyana 6:221–227
Rasmussen HN, Rasmussen FN (2009) Orchid mycorrhiza: implications of a mycophagous life style. Oikos 118:334–345
Rasmussen HN, Whigham DF (1993) Seed ecology of dust seeds in situ: a new study technique and its application in terrestrial orchids. Am J Bot 80:1374–1378
Rasmussen HN, Whigham DF (1998a) Importance of woody debris in seed germination of Tipularia discolor (Orchidaceae). Am J Bot 85:829–834
Rasmussen HN, Whigham DF (1998b) The underground phase: a special challenge in studies of terrestrial orchid populations. Bot J Linn Soc 126:49–64
Romand-Monnier F (2013) Dendrobium aphyllum. IUCN 2013. IUCN red list of threatened species. Version 2013.1. http://www.iucnredlist.org/details/15053780/0. Accessed 19 November 2013.
Sanchez MS, Bills GF, Zabalgogeazcoa I (2008) Diversity and structure of the fungal endophytic assemblages from two sympatric coastal grasses. Fungal Divers 33:87–100
Seaton P, Kendon JP, Pritchard HW, Puspitaningtyas DM, Marks TR (2013) Orchid conservation: the next ten years. Lankesteriana 13:93–101
Selosse MA, Boullard B, Richardson D (2011) Noël Bernard (1874–1911): orchids to symbiosis in a dozen years, one century ago. Symbiosis 54:61–68
Sheng CL, Lee YI, Gao JY (2012) Ex situ symbiotic seed germination, isolation and identification of effective symbiotic fungus in Cymbidium mannii (Orchidaceae). Zhi Wu Sheng Tai Xue Bao 36:859–869 (in Chinese)
Shimura H, Sadamoto M, Matsuura M, Kawahara T, Naito S, Koda Y (2009) Characterization of mycorrhizal fungi isolated from the threatened Cypripedium macranthos in a northern island of Japan: two phylogenetically distinct fungi associated with the orchid. Mycorrhiza 19:525–534
Sileshi GW (2012) A critique of current trends in the statistical analysis of seed germination and viability data. Seed Sci Res 22:145–159
Stewart SL, Kane ME (2006) Symbiotic seed germination of Habenaria macroceratitis (Orchidaceae), a rare Florida terrestrial orchid. Plant Cell Tissue Organ Cult 86:159–167
Stewart SL, Zettler LW (2002) Symbiotic germination of three semi-aquatic rein orchids Habenaria repens, H. quinquiseta, H. macroceratitis from Florida. Aquat Bot 72:25–35
Subedi A, Kunwar B, Gravendeel B et al (2013) Collection and trade of wild-harvested orchids in Nepal. J Ethnobiol Ethnomed 9:64
Swarts ND, Dixon KW (2009a) Terrestrial orchid conservation in the age of extinction. Ann Bot 104:543–556
Swarts ND, Dixon KW (2009b) Perspectives on orchid conservation in botanic gardens. Trends Plant Sci 14:590–598
Swarts ND, Sinclair EA, Francis A, Dixon KW (2010) Ecological specialization in mycorrhizal symbiosis leads to rarity in an endangered orchid. Mol Ecol 19:3226–3242
Takahashi K, Ogiwara I, Hakoda N (2000) Seed germination of Habenaria (pecteilis) radiata (Orchidaceae: Orchideae) in vitro. Lindleyana 15:59–63
Wang H, Zhu H, Li BG (1997) Vegetation on limestone in Xishuangbanna Southwest China. Guangxi Zhi Wu 17:101–117 (in Chinese)
Wang D, Jia SH, Zhang ZX, Cai YP, Lin Y (2007) Isolation and culture of an endophytic fungus associated with Dendrobium huoshanense and its effects on the growth of plantlets. J Fungal Res 5:84–88 (in Chinese)
Wang H, Fang H, Wang Y, Duan L, Guo S (2011) In situ seed baiting techniques in Dendrobium officinale Kimuraet Migo and Dendrobium nobile Lindl.: the endangered Chinese endemic Dendrobium (Orchidaceae). World J Microbiol Biotechnol 27:2051–2059
Warcup JH, Talbot PHB (1967) Perfect states of Rhizoctonias associated with orchids. New Phytol 66:631–641
Warcup JH, Talbot PHB (1971) Perfect states of rhizoctonias associated with orchids. II. New Phytol 70:35–40
Warcup JH, Talbot PHB (1980) Perfect states of rhizoctonias associated with orchids. III. New Phytol 86:267–272
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to methods and applications. Academic, New York, pp 315–322
Xing X, Ma X, Deng Z, Chen J, Wu F, Guo S (2012) Specificity and preference of mycorrhizal associations in two species of the genus Dendrobium (Orchidaceae). Mycorrhiza 23:317–324. doi:10.1007/s00572-012-0473-8
Yoder JA, Zettler LW, Stewart SL (2000) Water requirements of terrestrial and epiphytic orchid seeds and seedlings, and evidence for water uptake by means of mycotrophy. Plant Sci 156:145–150
Yonzone R, Lama D, Bhujel RB, Rai S, Kendra DKV, Viswavidyalaya UBK, Kalimpong PO (2011) Epiphytic orchid species diversity of Darjeeling Himalaya of West Bengal, India. Asian J Pharm Life Sci ISSN 2231:4423
Yuan ZL, Chen YC, Yang Y (2009) Diverse non-mycorrhizal fungal endophytes inhabiting an epiphytic, medicinal orchid (Dendrobium nobile): estimation and characterization. World J Microbiol Biotechnol 25:295–303
Zettler LW, Hofer CJ (1998) Propagation of the little club-spur orchid Platanthera clavellata by symbiotic seed germination and its ecological implications. Environ Exp Bot 39:189–195
Zettler LW, McInnis JTM (1994) Light enhancement of symbiotic seed germination and development of an endangered terrestrial orchid, Platanthera integrilabia. Plant Sci 102:133–138
Zettler LW, Piskin KA (2011) Mycorrhizal fungi from protocorms, seedlings, and mature plants of the eastern prairie fringed orchid, Platanthera leucophaea: a comprehensive list to augment conservation. Am Midl Nat 166:29–39
Zhang J, Cao M (1995) Tropical forest vegetation of Xishuangbanna, SW China and its secondary changes, with special reference to some problems in local nature conservation. Biol Conserv 73:229–238
Zhu JM, Zhao YM, Bai J, Li YT, Zhang KM, Chen JW, Wu JB, Wang HZ (2011) Preliminary studies on the antagonism of Dendrobium mycorrhizal fungi (Trichoderma persoon). J Hangzhou Normal Univ (Nat Sci Ed) 10:340–344 (in Chinese)
Ziegler AD, Fox JM, Xu J (2009) The rubber juggernaut. Science 324(5930):1024–1025
Acknowledgments
This research was supported by the National Natural Science Foundation of China (grant no. 31170358) to JYG and manuscript writing was supported by China National Post Doctoral Fund 2013 to UMG and Chinese Academy of Science CAS 135 program (XTBG-T01). We thank Richard T. Corlett and two anonymous reviewers for constructive comments on a previous version of the manuscript. We are also grateful to Wenliu Zhang, Xuli Fan, Hua Lin, Qiang Liu for field and laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiao-Meng Zi and Chun-Ling Sheng contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zi, XM., Sheng, CL., Goodale, U.M. et al. In situ seed baiting to isolate germination-enhancing fungi for an epiphytic orchid, Dendrobium aphyllum (Orchidaceae). Mycorrhiza 24, 487–499 (2014). https://doi.org/10.1007/s00572-014-0565-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-014-0565-8