Abstract
Background
Perforated peptic ulcer (PPU) remain a surgical emergency accounting for 37% of all peptic ulcer-related deaths. Surgery remains the standard of care. The benefits of laparoscopic approach have been well-established even in the elderly. However, because of inconsistent results with specific regard to some technical aspects of such technique surgeons questioned the adoption of laparoscopic approach. This leads to choose the type of approach based on personal experience. The aim of our study was to critically appraise the use of the laparoscopic approach in PPU treatment comparing it with open procedure.
Methods
A retrospective study with propensity score matching analysis of patients underwent surgical procedure for PPU was performed. Patients undergoing PPU repair were divided into: Laparoscopic approach (LapA) and Open approach (OpenA) groups and clinical-pathological features of patients in the both groups were compared.
Results
A total of 453 patients underwent PPU simple repair. Among these, a LapA was adopted in 49% (222/453 patients). After propensity score matching, 172 patients were included in each group (the LapA and the OpenA). Analysis demonstrated increased operative times in the OpenA [OpenA: 96.4 ± 37.2 vs LapA 88.47 ± 33 min, p = 0.035], with shorter overall length of stay in the LapA group [OpenA 13 ± 12 vs LapA 10.3 ± 11.4 days p = 0.038]. There was no statistically significant difference in mortality [OpenA 26 (15.1%) vs LapA 18 (10.5%), p = 0.258]. Focusing on morbidity, the overall rate of 30-day postoperative morbidity was significantly lower in the LapA group [OpenA 67 patients (39.0%) vs LapA 37 patients (21.5%) p = 0.002]. When stratified using the Clavien–Dindo classification, the severity of postoperative complications was statistically different only for C–D 1–2.
Conclusions
Based on the present study, we can support that laparoscopic suturing of perforated peptic ulcers, apart from being a safe technique, could provide significant advantages in terms of postoperative complications and hospital stay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The incidence of peptic ulcer disease has decreased, particularly in Western countries after the introduction of proton-pump inhibitors (PPI) along with antibiotic therapy for Helicobacter pylori eradication. Nowadays, peptic ulcer disease has a reported annual incidence between 0.03 and 0.19% worldwide [1,2,3,4,5,6]. However, although the number of patients with ulcer is decreased to less than one third, there was no marked decrease in the number of deaths from ulcer, and perforated peptic ulcer (PPU) remain a surgical emergency associated with increased mortality, accounting for 37% of all peptic ulcer-related deaths [7]. This indicates that the clinical picture of ulcer became more severe, in particular, combined use of LDA (low-dose aspirin) and non-steroidal anti-inflammatory drugs (NSAIDs) and advanced age serve as risk factors for the occurrence of LDA-induced ulcer and also increase the risk of haemorrhage and aggravation [8,9,10,11,12]. Surgery remains the standard of care for patients with PPU. The benefits of laparoscopic approach (LA) for patients with perforated peptic ulcer have been well-established even in the elderly [13,14,15,16]. The first laparoscopic repair of PPU was reported by Mouret et al. [17]. Laparoscopy is associated with less intraoperative blood loss, better pulmonary function, reduced postoperative pain, quicker return of bowel function, a shorter hospital stay, and a lower incisional hernia incidence when compared with standard open surgery [18]. Thereafter, retrospective studies found acceptable outcomes of LA for PPU. However, because of inconsistent results with specific regard to some technical aspects of such technique, i.e., peritoneal lavage, surgeons questioned the adoption of laparoscopic approach to perform ulcer repair [19, 20]. This leads the surgeons to choose the type of approach based on their personal experience. The aim of our study was to critically appraise the use of the laparoscopic approach in PPU treatment comparing it with open procedure.
Material and methods
Study settings and protocol
This research originates from a previous well-consolidated experience [21] thus following a similar methodology, a new collaborative research group was founded. The IGo-GIPS (Italian Group for Gastro-Intestinal Postoperative Surveillance) is a large, nationwide network created with the aim to undertake both prospective and/or retrospective studies investigating the perioperative outcomes of specific topics mainly concerning gastrointestinal surgery [22]. Centres were included on a volunteer basis, and neither investigators nor participating hospitals were paid for their collaboration. Clinical decisions, including operative technique, were always based on the criteria of individual centres and staff surgeons. Although procedures were not standardised per a study protocol, it is important to note that they were likely similar among participating hospitals, with some slight technical differences across institutions seldom taken into account because they were judged to not influence the outcome. The investigators were informed about the objectives of the project and asked for complete details about the surgical management of patients following standard methods and collection protocols as already described. Data regarding patients were prospectively collected from the study participating centres from January 2017 to June 2018, while data regarding other patients from July 2018 to December 2021 were retrospectively retrieved from hospital electronic databases. The former prospective study protocol was approved by the Ethics Committee of Sapienza University of Rome and of all the centres while no formal approval was requested for any other retrospective non-interventional study except in case of specific indication deemed by a single centre. However, a signed consent for the treatment and the analysis of data for scientific purposes was obtained from all patients before surgery. This study was conducted in accordance with the Declaration of Helsinki and its later amendments. All parts of the studies and the present manuscript have been checked and presented according to the checklist for Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) [23].
Inclusion and exclusion criteria and collected data confirmation
For the aim of the present study, we initially retrieved records of all patients having > 18 years with ICD-9-CM code ranging from 531.× to 534.× requiring emergency surgery from January 2017 to December 2021. Furthermore, bleeding ulcer, neoplastic perforation, sole endoscopic procedures, and emergency operations during the course of any other elective surgery were discarded. Other exclusion criteria were the following: age < 18 years; location other than stomach or duodenal bulb; previous upper GI surgery; lack of informed consent for the study participation, if requested; patients participating in other randomised or interventional clinical trials. Submissions made by unconfirmed participants, duplicate submissions, records with more than 5% of missing data were also excluded. Although the patient's demographic information was collected, all data were anonymized before analysis even for centre identification.
Patients’ characteristics, preoperative variables and objectives of this study
Data collected included patient demographic characteristics and clinical variables, procedure details, and outcomes. Demographics variables and clinical data included: age, gender, weight, height, body mass index (BMI), Glasgow Coma Scale (GCS), heart rate, systolic blood pressure, medical and surgical history (comorbidities), common preoperative biochemical blood examination (including C-Reactive Protein [CPR], and arterial blood gas analysis). Procedure details included: site and size of the ulcer, type of surgical approach and procedure performed, timing, conversion rate, peritoneal contamination). Comorbidity was recorded if the condition was being medically treated at the time of admission, or if previous treatment for the condition was described in the admission report. The Age-adjusted Charlson Comorbidity Index (age-CACI) was calculated and a score ≥ 6 was used to categorise patients having a severe comorbid condition. Preoperative risk was assessed with anaesthesiologist-assigned American Society of Anaesthesiologists (ASA). Furthermore, Systemic inflammatory response syndrome (SIRS) was evaluated according to the original consensus study (Sepsis-1) [24]. SIRS criteria ≥ 2 met the definition of SIRS. When appropriated, the Frailty profile was investigated by the 5-modified Frailty Index (5-mFI) and the Emergency Surgery Frailty Index (EmSFI). As previously described [21], the assessment of Activities of Daily Living and the Fried Frailty criteria was considered and in order to simplifying the concerning statements, the item “Altered autonomy” should be referred to the assessment of activities of daily living, while the item “Altered mobility” has been used to outline some of the Fried’s frailty criteria. When statistical analysis was performed, 5-mFI ≥ 0.4 score was used for categorising frailty as a binary variable according to current literature [25]. Sepsis was evaluated according to the qSOFA score. The Shock index, the Age-Shock Index, the Boey score, the Mannheim Peritonitis Index (MPI), the PULP Score, and the Jabalpur score were also calculated. Postoperative complications have been reported and categorised according to the Clavien–Dindo classification system by the study leader in each of the participating centres and the Comprehensive Complication Index was also calculated [26, 27]. Furthermore, morbidity was divided into three groups named as follows: C–D 1–2, C–D 3, and C–D 4. Although morbidity and mortality have been considered as the 30-day standard period definition, adverse outcomes have been reported regardless of the time elapsed from the surgical procedure if reasonably related to it and occurred during the hospitalisation following the main emergency procedure. First, the entire study cohort was investigated by analysing the initial approach selected regardless of the procedure performed. Furthermore, only the suture repair was taken into account because it was the most common technique adopted. Patients undergoing PPU repair were divided into two groups named Laparoscopic approach (LapA) and Open approach (OpenA) and clinical-pathological features of patients in the both groups were compared. The laparoscopic group included all patients who underwent an attempted pure laparoscopic procedure. An open conversion was defined as when a procedure was attempted via the minimally invasive approach but required an open incision to be completed. Patients who required conversion to laparotomy were included and analysed on an intention-to-treat basis. Leakage was defined as when bile or gastric content was detected in the drain output or by means of CT scan with oral water-soluble contrast. No routine use of the latter or of the methylene blue test was adopted. As well as stated above, due to the multicentre observational design of the study, there was not a uniform standardised protocol neither for the suture technique performed nor for the suture material used. However, because the simple suture, the suture plus omental flap, and suture with omental patch were equally distributed in both groups, the lack of uniformity in the technique and material used was not considered as a bias.
Statistical analysis
Statistical analysis was carried out using StataCorp 2019 STATA Statistical Software: release 16 (College Station,TX: StataCorp LLC). Dichotomous data and counts were presented in frequencies, whereas continuous data were presented as mean values ± standard deviations (SD) and/or median with 25–75 Interquartile Range (IQR) and minimum–maximum range. Differences between means were compared using the independent sample Student’s T-test or the Mann–Whitney U test when indicated. Fisher’s exact test or χ2 test, with or without Yates correction, were used to compare differences in frequencies. After an initial entire cohort summary description, the patients managed with gastric resection and those with lavage and drainage were excluded and a deeper statistical analysis was performed only on the patients who underwent the simple suture repair. Firstly, the accuracy of the morbidity and mortality prediction of MPI, Boey score, PULP score, and Jabalpur score was evaluated by receiver-operating characteristic (ROC) curve analysis, and the corresponding area under the curve (AUC) values was compared. Univariate analysis was performed in order to assess the Odds Ratio of any factor for suture leak and conversion to open. Odds Ratio was calculated by means of a 2 × 2 Contingency table for binary variables while by means of logistic regression for those continuous. All variables with a p value < 0.10 at univariate analysis were entered into a backward stepwise logistic regression model. If needed, receiver-operating characteristic (ROC) curve analysis was performed to calculate the cut-off value corresponding to the maximum Youden’s index of continuous variables in order to clarify the potential clinical relevance of such Odds Ratio values.
Thereafter, a propensity score matching was carried out. The Italian Version of IBM Corp. Released 2012. IBM SPSS Statistics for Macintosh, Version 21.0. IBM Analytics (Italy, Segrate, Milan) integrated with SPSS R Essentials for R Statistical Software version 2.14.2 (Foundation for Statistical Computing, Vienna, Austria) was used. The model was constructed to eliminate selection bias between groups and was reported according to the recommendations of Lonjon et al. [28]. Variables influencing decision regarding surgical approach and variables with potential influence on outcomes were assigned propensity scores using a bivariate logistic regression model. The final model included the following variables: sex as exact, age, ASA score, Age-Shock Index, and Age-CACI. We matched propensity scores 1:1 with the use of the nearest neighbour methods without replacement using the closest callipers width to achieve the maximum number of cases without statistical differences in confounders. In this instance, the calliper width was set at 0.2. All tests were two-tailed and a p value ≤ 0.05 was considered statistically significant.
Result
Entire cohort
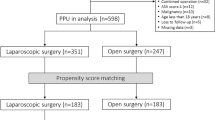
A total of 509 patients fulfilling the inclusion criteria were evaluated (Fig. 1). The overall mean age was 62.9 ± 17.4 (range 18 to 100 years), 273 (53.6%) were male. A laparoscopy approach (LA) was performed in 212 patients (41.6%) while an open approach (OA) was performed in 276 patients (54.2%). The overall conversion rate was 4.1% (21 patients). Four hundred and fifty-three patients (89.0%) underwent simple repair with or without an omental patch, 50 patients (9.8%) underwent gastric resection, while a peritoneal lavage and drainage was performed in 6 (1.2%) patients. Table 1 reports demographics, clinical and operative data stratified by the type of approach.
Simple repair group data
A total of 453 (89%) patients underwent PPU simple repair. Among these, a LA was adopted in 49% (222/453 patients) defined as “LapA group” with a conversion rate of 7.2% (16/222 patients), while an open approach was adopted in 51% (231/453 patients) defined as “OpenA group”.
Table 2 summarises demographics features and clinical data of simple repair patients before and after propensity score matching. The rate of male patients was similar between the groups as well as the BMI. The ASA score was significantly higher in the OpenA group [OpenA 123 patient (53.2%) vs LapA 95 patients (43.0%), p = 0.030]. No differences were found between the groups in terms of preoperative laboratory value (Hb, lactate, glycemia, WBC, PLT CRP) except for creatinine which was significantly higher in the OpenA patients (OpenA 1.3 ± 1.2 vs LapA 1.0 ± 0.7 p = 0.001). Regarding the scores that reflect patient general status such as shock index, CACI, EmSFI, and qSOFA only the last one was higher in the OpenA group than in the LapA group (qSofa: OpenA 64 ± 3.2 vs LapA 0.41 ± 2.60. p = 0.001). SIRS was anyway more frequent in the OpenA group [OpenA 108 patients (47.0%) vs LapA 71 patients (32.0%) p = 0.001]. Moreover, patients in the OpenA group were more fragile as reflected by a lower independency in daily activity and a higher EmSFI index. Operative details and postoperative outcomes before and after propensity score matching are summarized in Table 3.
Boeys score, MPI and Jabalpur score were not different between-groups while Pulp score was slightly lower in the LapA group (OpenA 5 ± 3.4 vs LapA 4 ± 3.0). The mean operative time was 86.1 ± 31.3 min in the LapA group and 98.3 ± 40.1 in the OpenA group. The difference was statistically significant (p = 0.001). Mean diameter of the perforation and site were similar between the groups and no difference in terms of leak rate was retrieved. The overall morbidity rates was higher in the OpenA group [OpenA 93 patients (40.3%) vs LapA 49 patients (22.1%) p < 0.001] as well as the postoperative 30-day mortality rate [OpenA 45 patients (19.5%) vs LapA 19 patients (8.6%) (p = 0.001)]. As concern morbidity, MPI, Boey score, PULP score, and Jabalpur score showed no discrimination ability [AUC 0.491 (95% CI 0.433–0.548), 0.523 (95% CI 0.468–0.578), 0.562 (95% CI 0.507–0.617), and 0.501 (95% CI 0.442–0.560), respectively; p = 0.174] (Fig. 2). As regard to mortality Boey score e Jabalpur score exhibited poor discrimination ability [AUC 0.663 (95% CI 0.592–0.733), 0.604 (95% CI 0.528–0.680), respectively] while the accuracy of PULP score and MPI was acceptable [AUC 0.860 (95% CI 0.817–0.904), 0.740 (95% CI 0.674–0.807), respectively; p < 0.001] (Fig. 3). After propensity score matching, 172 patients were included in each group (the LapA and the OpenA). The analysis revealed that the pre-operative variables found to be significantly different before matching (i.e., age, ASA, CACI, EmSFI) were then well-balanced between both groups (Table 2) except for qSofa and altered autonomy, which have remained higher in the OpenA group. Outcomes of the propensity score matching demonstrated increased operative times in the OpenA (OpenA: 96.4 ± 37.2 vs LapA 88.47 ± 33 min, p = 0.035), with shorter overall length of stay in the LapA group (OpenA 13 ± 12 vs LapA 10.3 ± 11.4 days p = 0.038), and more frequent discharge to home (OpenA 81.3% vs LapA 72.6%, p = 0.017) (Table 3). Moreover, there was no statistically significant difference in mortality [OpenA 26 (15.1%) vs LapA 18 (10.5%), p = 0.258]. While focusing on morbidity, the overall rate of 30-day postoperative morbidities was significantly lower in the LapA group than in the OpenA group [OpenA 67 patients (39.0%) vs LapA 37 patients (21.5%) p = 0.002]. When stratified using the Clavien–Dindo classification, the severity of postoperative complications was statistically different only for C–D 1–2. However, it is important to note that when C–D 4 was taken into account the statistical analysis showed an Odds Ratio for OpenA almost three times [OR 2.870; (95% Conf. Interval) 0.888–9.265]. The results of regression analysis concerning suture leak and conversion to open surgery are shown in Table 4. Multivariate analysis revealed that pyloric/duodenal site, Boey score, and Age-Shock Index were the variables statistically related to suture leak while SIRS, platelet, serum lactate, MPI, and dimension were significantly associated with conversion to open surgery.
Discussion
The surgeons’ greater experience in laparoscopic surgery and the remarkable development of various laparoscopic surgical equipment and new anaesthesiology techniques had led to the improvement of the safety and feasibility of laparoscopic procedures. As a consequence, a large diffusion of minimally invasive approaches for the execution of more complex and demanding operations has been seen also in the emergency setting [18, 29, 30]. The surgical approach to perforated peptic ulcer has changed dramatically in recent decades. In the 1970s, vagotomy and pyloroplasty, with the whole risks associated with this type of surgery, was the procedure more frequently performed. In the following decade, a less aggressive approach was recommended: simple suture with or without omentoplasty [31]. In the last decades, many studies have reported the safety and the efficacy of LA for the treatment of PPU [16, 32]. Nevertheless, clinical reality differs from the results of randomised studies by a complex series of non-objectionable real-world data influencing treatment plans. In light of this, it is pivotal to define which patients are suitable for a laparoscopic approach. Laparoscopic repair of a PPU was described as early as 1990, but rates of adoption of this approach have been un-clear. Some international studies have reported a laparoscopic repair rate ranging from 41 to 76% and our series resulted in line with this findings [33, 34]. Moreover, in the 2020 WSES guidelines, the laparoscopic approach is suggested to be the first-line treatment for stable patients with small ulcers as long as surgeons are familiar with the skill and appropriate equipment is available [35]. In clinical practice, the patients who undergo a laparoscopic approach compared to an open approach have different characteristics, in terms of clinical presentation and comorbidities [36,37,38,39]. Our study shows that LapA patients are less fragile (lower CACI and EmSFI) and have a better overall general and inflammatory status (i.e., lower qSofa, lower rate of SIRS, and lower MPI). However, after propensity model only altered autonomy and qSofa were found to be statistically higher in the OpenA group. The main clinical scoring systems used to predict morbidity and mortality in PPU patients are Boey score, PULP Score and Jabalpur score [37, 38]. They are often used in conjunction with the Mannheim Peritonitis Index and ASA for improved comparison of the severity of physiologic derangement [38, 40, 41]. The Boey score seems to be the most disease-specific and is simple to calculate taking into consideration major medical illness, preoperative shock, and duration of perforation longer than 24 h before surgery. It is reported that a Boey score of 2 or higher indicates mortality rate greater than 30% [42,43,44]. Similarly to what performed by other authors, having many collected data, we retrospective calculated several common scores. The overall mean of the scores was similar between the OpenA and LapA groups both before and after propensity score matching. A large English population-based cohort study of Leusink et al. confirmed that laparoscopic repair of PPU is associated with a significant reduction in 30-day and 90-day mortality, postoperative pneumonia, and length of hospital stay [15]. A recent meta-analysis of randomized clinical trials comparing laparotomy to laparoscopic repair found no difference in morbidity or mortality between the two procedures [45]. Although previous cohort studies have compared outcomes between LA and OA, the matched nature of this study makes the findings more informative for the surgeon's daily activities. We found that the benefits of a laparoscopic approach include a shorter operative time and length of stay and a slightly lower mortality rate. According to the literature, perforation carries an associated mortality rate of 10–40%; our overall mortality rate is approaching the lower limit and we evidenced that the rate of death after laparoscopic surgery was similar to the rate of the open surgery. These findings are in line with the literature: a recent Danish propensity analysis with a large study population failed to demonstrate a lower mortality in the laparoscopic group [46]. Only one English retrospective population-based study, and a meta-analysis with non-randomized studies included in the analysis, were able to demonstrate the benefits of laparoscopy in terms of mortality [15]. In a meta-analysis, Zhou et al. found significant differences in hospital mortality between the laparoscopic repair and open repair groups in the high quality non-randomized studies, but not in the clinical trials [47]. Like Mirabella et al., we believe that mortality depends more on the risk factors of the patient and the aggressiveness of the ulcer than on the surgical approach [48]. Focusing on morbidity, literature reports that postoperative complications usually occur in 30% of cases. Our morbidity rate is in line with the literature with an overall morbidity rate of 30.2% that drops to 21% in case of laparoscopic approach with simple repair. Moreover, the severity of postoperative complications was statistically different only for C–D 1–2, but it is important to note that when C–D 4 was taken into account the Odds Ratio for OpenA group was almost three times. The predictive value of morbidity of all the analysed scores was very low, equivalent to chance. On the contrary, when the accuracy of the scores was evaluated in predicting mortality, our series showed that PULP score and MPI had acceptable accuracy with the former having the highest AUC value. The main cause for reoperation following surgical repair is suture leak. Proposed explanations, based on current literature data, include the difficulty in laparoscopic knot tying, ulcer diameter (> 2 cm) and abdominal contamination [49, 50]. Based on our multivariate analysis the factors associated with leak were ulcer site (pyloric/duodenal), higher Boey Score and higher Age-shock index while PULP score revealed his statistically significant association with leakage only at the univariate analysis. A number of studies have reported the utility of Boey score and PULP score in predicting conversion of laparoscopic PPU repair while Muller et al. stated that the conversion to an open approach could only be assessed intraoperatively [51, 52]. In our series, as regards conversion to open surgery, PULP score and Jabalpur score were identified as risk factors only at univariate analysis while Boey score was not associated with conversion. Furthermore, our multivariate analysis showed that SIRS, platelet, serum lactate, MPI, and ulcer dimension were statistically risk factors and this is unanimous with the literature where is reported that the main reasons for conversion are a posterior location that did not allow a proper inspection of the ulcer, a large perforation size (> 2 cm), severe inflammatory involvement of the surrounding tissue, adhesions, and suspected tumours [51, 53]. In light of this, investigating our series, we were not able to determine the actual causes which required conversion to laparotomy because the data collected did not include enough information about this item. However, a brief survey allowed us to detect that the main cause of conversion was the ulcer size followed by anaesthesiologist decision for medical reasons and by technical difficulty due to adhesions. Consequently, we agree that a conversion to an open procedure should only be assessed intraoperatively.
Limitations
As it clearly derives from above, this study has several limitations. First, it is a mixed prospective and retrospective multi-centre study with not previously established common treatment protocol. Although data were collected following a homogeneous method, decisions regarding the timing and the choice of approach might differ among institutions or even in the same hospital due to attending surgeon preference and expertise or intra- and inter-hospital settings and instrumental availability in not a referral centre or after-hours. In addition, the suture technique could be slightly different by the surgeon’s preference. Therefore, the analysis was not stratified by participating surgeons or institutions because the protocol did not enable a separate comparison of data for obvious professional ethical reasons. Another issue to take into account is that a part of the study included patients treated during the COVID-19 pandemic, which has dramatically altered the clinical practice also in emergency surgery. This is why in this study there is a high risk of patient selection bias. However, the number of cases recruited along with the large amount of collected data provided a good sample size for multiple logistic analyses mitigating these limitations. Moreover, a multicentre study allows better generalisation of results than a single centre, while the propensity score model let us to compare two similar restricted groups excluding confounders and correcting for undetectable selection bias. Furthermore, in order to evaluate the potential benefits of the laparoscopic approach, it is remarkable that postoperative pain was not assessed because medical records did not always provide this data. Lastly, no details about long-term outcomes such as incisional hernia or recurrence of PPU or bleeding were evaluated.
Conclusion
Despite the promising results of laparoscopy and the suggestion of literature guidelines, the open approach is still widely adopted as our research also witnesses. Based on the results of the present study, we can support that laparoscopic suturing of perforated peptic ulcers, apart from being a safe technique, could provide significant advantages in terms of postoperative complications and hospital stay showing its non-inferiority when compared with open approach. Therefore, we advocate that laparoscopy should be always used as first choice in stable patients in order to confirm the diagnosis, localise the site and size of the perforation, determine the extent of peritonitis, and try to perform suture even with the use of barbed knotless suture.
References
Dadfar A, Edna TH (2020) Epidemiology of perforating peptic ulcer: a population-based retrospective study over 40 years. World J Gastroenterol 26(35):5302–5313. https://doi.org/10.3748/wjg.v26.i35.5302
Johnson CH, McLean RC, McCallum I, Perren D, Phillips AW (2019) An evaluation of the epidemiology, management and outcomes for perforated peptic ulcers across the North of England over 15 years: a retrospective cohort study. Int J Surg 64:24–32. https://doi.org/10.1016/j.ijsu.2019.03.005
Azhari H, King JA, Coward S et al (2022) The global incidence of peptic ulcer disease is decreasing since the turn of the 21st century: a study of the organisation for economic co-operation and development (OECD). Am J Gastroenterol 117(9):1419–1427. https://doi.org/10.14309/ajg.0000000000001843
Lanas A, Chan FKL (2017) Peptic ulcer disease. Lancet 390(10094):613–624
Sivaram P, Sreekumar A (2018) Preoperative factors influencing mortality and morbidity in peptic ulcer perforation. Eur J Trauma Emerg Surg 44(2):251–257. https://doi.org/10.1007/s00068-017-0777-7
Ahmed M, Mansoor T, Rab AZ, Rizvi SAA (2022) Risk factors influencing postoperative outcome in patients with perforated peptic ulcer: a prospective cohort study. Eur J Trauma Emerg Surg 48(1):81–86. https://doi.org/10.1007/s00068-020-01597-6
Cirocchi R, Soreide K, Di Saverio S et al (2018) Meta-analysis of perioperative outcomes of acute laparoscopic versus open repair of perforated gastroduodenal ulcers. J Trauma Acute Care Surg 85(2):417–425. https://doi.org/10.1097/TA.0000000000001925
Lanas A, Garcia-Rodriguez LA, Polo-Tomas M, Ponce M, Quintero E, Perez-Aisa MA, Gisbert JP, Bujanda L, Castro M, Munoz M, Del-Pino MD, Garcia S, Calvet X (2011) The changing face of hospitalisation due to gastrointestinal bleeding and perforation. Aliment Pharmacol Ther 33:585–591. https://doi.org/10.1111/j.1365-2036.2010.04563.x
Kubosawa Y, Mori H, Kinoshita S et al (2019) Changes of gastric ulcer bleeding in the metropolitan area of Japan. World J Gastroenterol 25(42):6342–6353. https://doi.org/10.3748/wjg.v25.i42.6342
Nagasue T, Nakamura S, Kochi S et al (2015) Time trends of the impact of Helicobacter pylori infection and nonsteroidal anti-inflammatory drugs on peptic ulcer bleeding in Japanese patients. Digestion 91(1):37–41. https://doi.org/10.1159/000368810
Leow AH, Lim YY, Liew WC, Goh KL (2016) Time trends in upper gastrointestinal diseases and Helicobacter pylori infection in a multiracial Asian population–a 20-year experience over three time periods. Aliment Pharmacol Ther 43:831–837. https://doi.org/10.1111/apt.13550
Nagata N, Niikura R, Sekine K et al (2015) Risk of peptic ulcer bleeding associated with Helicobacter pylori infection, nonsteroidal anti-inflammatory drugs, low-dose aspirin, and antihypertensive drugs: a case-control study. J Gastroenterol Hepatol 30(2):292–298. https://doi.org/10.1111/jgh.12805
Katkhouda N, Mavor E, Mason RJ, Campos GMR, Soroushyari A, Berne TV (1999) Laparoscopic repair of perforated duodenal ulcers: outcome and efficacy in 30 consecutive patients. Arch Surg 134(8):845–850. https://doi.org/10.1001/archsurg.134.8.845
Coe PO, Lee MJ, Boyd-Carson H, Lockwood S, Saha A (2022) Open versus laparoscopic repair of perforated peptic ulcer disease: a propensity-matched study of the National Emergency Laparotomy Audit. Ann Surg 275(5):928–932. https://doi.org/10.1097/SLA.0000000000004332
Leusink A, Markar SR, Wiggins T, Mackenzie H, Faiz O, Hanna GB (2018) Laparoscopic surgery for perforated peptic ulcer: an English national population-based cohort study. Surg Endosc 32(9):3783–3788. https://doi.org/10.1007/s00464-018-6058-7
Fransvea P, Costa G, Lepre L et al (2020) Laparoscopic repair of perforated peptic ulcer in the elderly: an interim analysis of the FRAILESEL Italian Multicenter Prospective Cohort Study. Surg Laparosc Endosc Percutan Tech 31(1):2–7. https://doi.org/10.1097/SLE.0000000000000826
Mouret P, François Y, Vignal J, Barth X, Lombard-Platet R (1990) Laparoscopic treatment of perforated peptic ulcer. Br J Surg 77(9):1006. https://doi.org/10.1002/bjs.1800770916
Costa G, Fransvea P, Podda M, Pisanu A, Carrano FM, Iossa A, Balducci G, Agresta F, ERASO (Elderly Risk Assessment and Surgical Outcome) Collaborative Study Group (2020) The use of emergency laparoscopy for acute abdomen in the elderly: the FRAILESEL Italian Multicenter Prospective Cohort Study. Updates Surg 72:513–525
Pereira A, Santos Sousa H, Gonçalves D et al (2021) Surgery for perforated peptic ulcer: is laparoscopy a new paradigm? Minim Invasive Surg 2021:8828091. https://doi.org/10.1155/2021/8828091
Quah GS, Eslick GD, Cox MR (2019) Laparoscopic repair for perforated peptic ulcer disease has better outcomes than open repair. J Gastrointest Surg 23(3):618–625. https://doi.org/10.1007/s11605-018-4047-8
Costa G, Massa G, ERASO (Elderly Risk Assessment and Surgical Outcome) Collaborative Study Group (2018) Frailty and emergency surgery in the elderly: protocol of a prospective, multicenter study in Italy for evaluating perioperative outcome (The FRAILESEL Study). Updates Surg 70:97–104
Costa G, Fransvea P, Puccioni C et al (2022) Gastro-intestinal emergency surgery: evaluation of morbidity and mortality. Protocol of a prospective, multicenter study in Italy for evaluating the burden of abdominal emergency surgery in different age groups. (The GESEMM study). Front Surg 9:927044. https://doi.org/10.3389/fsurg.2022.927044
von Elm E, Altman DG, Egger M et al (2014) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg 12(12):1495–1499. https://doi.org/10.1016/j.ijsu.2014.07.013
Bone RC, Balk RA, Cerra FB (1992) American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 20(6):864–874. https://doi.org/10.1097/00003246-199206000-00025
Chimukangara M, Helm MC, Frelich MJ, Bosler ME, Rein LE, Szabo A, Gould JC (2017) A 5-item frailty index based on NSQIP data correlates with outcomes following paraesophageal hernia repair. Surg Endosc 31(6):2509–2519. https://doi.org/10.1007/s00464-016-5253-7
Dindo D, Demartines N, Clavien PA (2004) Classification of Surgical Complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Slankamenac K, Graf R, Barkun J, Puhan MA, Clavien PA (2013) The comprehensive complication index: a novel continuous scale to measure surgical morbidity. Ann Surg 258(1):1–7. https://doi.org/10.1097/SLA.0b013e318296c732
Lonjon G, Boutron I, Trinquart L et al (2014) Comparison of treatment effect estimates from prospective nonrandomized studies with propensity score analysis and randomized controlled trials of surgical procedures. Ann Surg 259(1):18–25. https://doi.org/10.1097/SLA.0000000000000256
Heywood N, Parmar KL, Stott M et al (2021) The laparoscopy in emergency general surgery (LEGS) study: a questionnaire survey of UK practice. Ann R Coll Surg Engl 103(2):120–129. https://doi.org/10.1308/rcsann.2020.7005
Agresta F, Campanile FC, Podda M et al (2017) Current status of laparoscopy for acute abdomen in Italy: a critical appraisal of 2012 clinical guidelines from two consecutive nationwide surveys with analysis of 271,323 cases over 5 years. Surg Endosc 31(4):1785–1795. https://doi.org/10.1007/s00464-016-5175-4
Chung KT, Shelat VG (2017) Perforated peptic ulcer - an update. World J Gastrointest Surg 9(1):1–12. https://doi.org/10.4240/wjgs.v9.i1.1
Olufajo OA, Wilson A, Yehayes B, Zeineddin A, Cornwell EE, Williams M (2020) Trends in the surgical management and outcomes of complicated peptic ulcer disease. Am Surg 86(7):856–864. https://doi.org/10.1177/0003134820939929
Mohamedahmed AYY, Albendary M, Patel K et al (2022) Comparison of omental patch closure versus simple closure for laparoscopic repair of perforated peptic ulcer: a systematic review and meta-analysis. Am Surg 2022:31348211067991. https://doi.org/10.1177/00031348211067991
Sanabria AE, Morales CH, Villegas MI (2005) Laparoscopic repair for perforated peptic ulcer disease. Cochrane Database Syst Rev 4:CD004778. https://doi.org/10.1002/14651858.CD004778.pub2
Tarasconi A, Coccolini F, Biffl WL et al (2020) Perforated and bleeding peptic ulcer: WSES guidelines. World J Emerg Surg 15:3. https://doi.org/10.1186/s13017-019-0283-9
Lee MJ, Coe PO, O’Donoghue R, Peirson M, Saha A (2020) Variation in descriptors of patient characteristics in randomized clinical trials of peptic ulcer repair: a systematic review. Br J Surg 107(12):1570–1579. https://doi.org/10.1002/bjs.11771
Rivai MI, Suchitra A, Janer A (2021) Evaluation of clinical factors and three scoring systems for predicting mortality in perforated peptic ulcer patients, a retrospective study. Ann Med Surg (Lond) 69:102735. https://doi.org/10.1016/j.amsu.2021.102735
Patel S, Kalra D, Kacheriwala S, Shah M, Duttaroy D (2019) Validation of prognostic scoring systems for predicting 30-day mortality in perforated peptic ulcer disease. Turk J Surg 35(4):252–258. https://doi.org/10.5578/turkjsurg.4211
Tan S, Wu G, Zhuang Q et al (2016) Laparoscopic versus open repair for perforated peptic ulcer: a meta analysis of randomized controlled trials. Int J Surg 33(Pt A):124–132. https://doi.org/10.1016/j.ijsu.2016.07.077
Koranne A, Byakodi KG, Teggimani V, Kamat VV, Hiregoudar A (2022) A comparative study between peptic ulcer perforation score, mannheim peritonitis index, ASA score, and Jabalpur score in predicting the mortality in perforated peptic ulcers. Surg J (N Y) 8(3):e162–e168. https://doi.org/10.1055/s-0042-1743526
Saafan T, El Ansari W, Al-Yahri O et al (2019) Assessment of PULP score in predicting 30-day perforated duodenal ulcer morbidity, and comparison of its performance with Boey and ASA, a retrospective study. Ann Med Surg (Lond) 42:23–28. https://doi.org/10.1016/j.amsu.2019.05.001
Lohsiriwat V, Prapasrivorakul S, Lohsiriwat D (2009) Perforated peptic ulcer: clinical presentation, surgical outcomes, and the accuracy of the Boey scoring system in predicting postoperative morbidity and mortality. World J Surg 33(1):80–85. https://doi.org/10.1007/s00268-008-9796-1
Anbalakan K, Chua D, Pandya GJ, Shelat VG (2015) Five year experience in management of perforated peptic ulcer and validation of common mortality risk prediction models - are existing models sufficient? A retrospective cohort study. Int J Surg 14:38–44. https://doi.org/10.1016/j.ijsu.2014.12.022
Akin E, Altintoprak F, Akdeniz Y et al (2022) Open versus laparoscopic technique in peptic ulcus perforation, how effective are score systems? Single-center experience and literature review. Peptik ulkus perforasyonunda laparoskopik ve açık tekniğin karşılaştırılması, skor sistemleri ne kadar etkin? Tek merkez deneyimi ve literatür derlemesi. Ulus Travma Acil Cerrahi Derg 28(10):1437–1441. https://doi.org/10.14744/tjtes.2022.78938
Bertleff MJ, Halm JA, Bemelman WA, van der Ham AC, van der Harst E, Oei HI, Smulders JF, Steyerberg EW, Lange JF (2009) Randomized clinical trial of laparoscopic versus open repair of the perforated peptic ulcer: the LAMA Trial. World J Surg 33(7):1368–1373
Zogovic S, Bojesen AB, Andos S, Mortensen FV (2019) Laparoscopic repair of perforated peptic ulcer is not prognostic factor for 30-day mortality (a nationwide prospective cohort study) [published correction appears in Int J Surg. 2020;75:11-12]. Int J Surg 72:47–54. https://doi.org/10.1016/j.ijsu.2019.10.017
Zhou C, Wang W, Wang J et al (2015) An updated meta-analysis of laparoscopic versus open repair for perforated peptic ulcer. Sci Rep 5:13976. https://doi.org/10.1038/srep13976
Mirabella A, Fiorentini T, Tutino R, Falco N, Fontana T, De Marco P, Gulotta E, Gulotta L, Licari L, Salamone G, Melfa I, Scerrino G, Lupo M, Speciale A, Cocorullo G (2018) Laparoscopy is an available alternative to open surgery in the treatment of perforated peptic ulcers: a retrospective multicenter study. BMC Surg 18(1):78. https://doi.org/10.1186/s12893-018-0413-4
Lund S, Chauhan KK, Zietlow J et al (2021) Risk factors for gastrointestinal leak after perforated peptic ulcer disease operative repair. Am Surg 87(12):1879–1885. https://doi.org/10.1177/00031348211056263
Dogra P, Kaushik R, Singh S, Bhardwaj S (2022) Risk factors for leak after omentopexy for duodenal ulcer perforations. Eur J Trauma Emerg Surg. https://doi.org/10.1007/s00068-022-02058-y
Muller MK, Wrann S, Widmer J, Klasen J, Weber M, Hahnloser D (2016) Perforated peptic ulcer repair: factors predicting conversion in laparoscopy and postoperative septic complications. World J Surg 40(9):2186–2193. https://doi.org/10.1007/s00268-016-3516-z
Wang YH, Wu YT, Fu CY, Liao CH, Cheng CT, Hsieh CH (2022) Potential use of peptic ulcer perforation (PULP) score as a conversion index of laparoscopic-perforated peptic ulcer (PPU) repair. Eur J Trauma Emerg Surg 48(1):61–69. https://doi.org/10.1007/s00068-020-01552-5
Kim JH, Chin HM, Bae YJ, Jun KH (2015) Risk factors associated with conversion of laparoscopic simple closure in perforated duodenal ulcer. Int J Surg 15:40–44. https://doi.org/10.1016/j.ijsu.2015.01.028
Acknowledgements
Ferdinando Agresta MD U.O.C. Chirurgia Generale, Ospedale di Vittorio Veneto, Treviso, Italy. Giovanni Alemanno MD Emergency Surgery Department, Careggi University Hospital, Florence, Italy. Massimo Antropoli MD Emergency Surgery Department "A. Cardarelli" Hospital, Naples Italy. Nicola Apice MD UOC Chirurgia Generale—ASL Frosinone. Giulio Argenio MD Emergency Surgery Unit, AOU San Giovanni di Dio e Ruggi d'Aragona, University of Salerno, Salerno, Italy. Nicola Avenia MD General and Endocrine Surgery, School of Medical, University of Perugia, S. Maria University Hospital, 05100 Terni, Italy. Antonio Azzinnaro MD Department of Abdominal Surgery—General and Hepatopancreatobiliary Surgery Unit, Ente Ospedaliero (E.O.) Galliera Hospital, Genova, Italy. Andrea Barberis Department of Abdominal Surgery—General and Hepatopancreatobiliary Surgery Unit, Ente Ospedaliero (E.O.) Galliera Hospital, Genova, Italy. Giorgio Badessi MD Department of General and Emergency Surgery, Policlinico G. Martino, University of Messina, Italy. Gianadnrea Baldazzi MD General Surgery Unit, PO Sesto San Giovanni, ASST Nord Milano, Sesto San Giovanni, MI, Italy. Carlo Bergamini MD Emergency Surgery Department, Careggi University Hospital, Florence, Italy. G. Bianco Fondazione Policlinico Universitario Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Alan Biloslavo MD Department of General Surgery, ASUITS, Cattinara Hospital, Trieste, Italy. Cristina Bombardini MD UO Chirurgia 1, Dipartimento Chirurgico, Arcispedale Sant'Anna, Azienda Ospedaliero-Universitaria di Ferrara, Ferrara, Italy. Giuseppe Borzellino MD Department of General Surgery, University Hospital of Verona, Piazzale A. Stefani 1, 37128, Verona, Italy. Gioia Brachini MD Emergency Department, Policlinico Umberto I, Sapienza University, Rome, Italy. Buonanno Gennaro Maurizio MD UOC Chirurgia Generale e di Urgenza AORN San Pio Benevento Italy. Tiberio Canini Department of General Surgery and Emergency Surgery, Fondazione IRCCS Cà Granda, Ospedale Maggiore Policlinico, Milan-Italy. Gabriella Teresa Capolupo MD Surgery Centre, Colorectal Surgery Unit, Fondazione Policlinico Universitario Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Filippo Carannante MD Surgery Centre, Colorectal Surgery Unit, Fondazione Policlinico Universitario Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Marco Caricato MD Surgery Centre, Colorectal Surgery Unit, Fondazione Policlinico Universitario Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Diletta Cassini MD General Surgery Unit, PO Sesto San Giovanni, ASST Nord Milano, Sesto San Giovanni, MI, Italy. Maurizio Castriconi MD Emergency Surgery Department "A. Cardarelli" Hospital, Naples Italy. Alberto Catamerò MD Fondazione Policlinico Universitario Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Marco Catarci MD General Surgery Unit, Sandro Pertini Hospital, ASL Roma 2, 00157 Rome, Italy. Ceccarelli Graziano General and Robotic Surgery Department, San Giovanni Battista Hospital, Foligno, Italy. Ceresoli Marco, General and Emergency Surgery, School of Medicine and Surgery, Milano-Bicocca University, Monza, Italy. Massimo Chiarugi MD General and Emergency Surgery Unit, Trauma Center, New Santa Chiara Hospital, University of Pisa, Via Paradisa, Pisa, Italy. Nicola Cillara MD S.O.C. Chirurgia Generale, P.O. Santissima Trinità, Cagliari, Italy. Roberto Cirocchi MD Department of General Surgery, University of Perugia, 06100, Perugia, Italy. Luigi Cobuccio MD Emergency Surgery Unit, Azienda Ospedaliero Universitaria Pisana Cisanello Hospital, Via Piero Trivella, 56124 Pisa, Italy. Federico Coccolini MD Emergency Surgery Unit, Azienda Ospedaliero Universitaria Pisana Cisanello Hospital, Via Piero Trivella, 56124 Pisa, Italy. Gianfranco Cocorullo MD Unit of General and Emergency Surgery, Department of Surgical Oncological and Oral Sciences, Policlinico "P. Giaccone"-University of Palermo, 90127 Palermo, Italy. Alessandro Costa, Unicamillus University. Gianluca Costa MD Colorectal Surgery Unit—Fondazione Policlinico Campus Bio-Medico, University Hospital of University Campus Bio-Medico of Rome, Rome, Italy. Valerio Cozza MD Emergency Surgery and Trauma—Fondazione Policlinico Universitario "A. Gemelli" IRCCS, Rome, Italy. Antonio Crucitti MD Cristo Re Hospital, Rome, Italy. Eugenio Cucinotta MD Department of Human Pathology of the Adult and Evolutive Age "Gaetano Barresi", Section of General Surgery Unit, University of Messina, Messina, Italy. Rosella D’Alessio MD Department of General Surgery 2, ASST Opedali Civili di Brescia, Brescia, Italy. Andrea de Manzoni Garberini U.O.C. di Chirurgia Generale 1 dell'Ospedale Civile Spirito Santo di Pescara. Nicolò De Manzini MD Department of General Surgery, ASUITS, Cattinara Hospital, Trieste, Italy. Carlo De Nisco MD General Surgery Unit, San Francesco Hospital, ASSL Nuoro, Nuoro, Italy. Marco De Prizio MD General Surgery Unit, San Donato Hospital, 52100 Arezzo, Italy. Elena Finotti MD General Surgery Unit, "SS. Giovanni e Paolo" Hospital, Venezia, Italy. Pietro Fransvea MD Emergency Surgery and Trauma—Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy. Barbara Frezza MD General Surgery Unit, San Donato Hospital, 52100 Arezzo, Italy. Giovanni Maria Garbarino MD Department of Medical-Surgical Science and Translational Medicine, Sapienza University of Rome, Sant'Andrea Hospital, Rome, Italy. Gianluca Garulli MD General Surgery Unit, Infermi Hospital, 47900 Rimini, Italy. Michele Genna MD Department of General Surgery, Unit of General and Obesity Surgery, University and Hospital Trust of Verona, Verona, Italy. Stefano Giannessi MD Operative Unit of General Surgery, San Jacopo Hospital, Pistoia, Italy. Alessio Giordano MD Emergency Surgery Unit Careggi Hospital Firenze, Italy. Mario Guerrieri MD Department of General Surgery, Università Politecnica Delle Marche, Piazza Roma 22, 60121, Ancona, Italy. Veronica Iacopini Unit of Emergency Surgery, Emergency Department, Careggi University Hospital, Florence, Italy. Hayato Kurihara MD Emergency Surgery Unit, IRCCS Fondazione Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy. Antonio La Greca MD Emergency Surgery and Trauma—Fondazione Policlinico Universitario "A. Gemelli" IRCCS, Rome, Italy. Giovanni Laracca Guglielmo Surgical and Medical Department of Translational Medicine, Sant'Andrea Teaching Hospital, Sapienza University of Rome, Via di Grottarossa 1035, 00189 Rome, Italy. Ernesto Laterza MD Department of General Surgery, ULSS9 Scaligera, Mater Salutis Hospital, Legnago, Verona, Italy. Luca Lepre MD General Surgery Unit, Santo Spirito in Sassia Hospital, Rome, Italy. Gianluca Liotta MD Department of Surgery, Azienda Ospedaliera S. Camillo Forlanini, Rome, Italy. Diego Mariani MD Department of Emergency General Surgery, Legnano Hospital, ASST Ovest Milanese, Legnano, MI, Italy. Pierluigi Marini MD Department of Surgery, Azienda Ospedaliera S. Camillo Forlanini, Rome, Italy. Rinaldo Marzaioli MD Department of Emergency and Organ Transplantation (DETO), University Medical School "A. Moro" Bari, Bari, Italy. Gianluca Mascianà MD Surgery Centre, Colorectal Surgery Unit, Fondazione Policlinico Universitariio Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Gianluca Mazzoni MD General Surgery Unit, G.B. Grassi Hospital, ASL Roma 3, Rome, Italy. Paolo Mercantini MD Surgical and Medical Department of Translational Medicine, Sant'Andrea Hospital, Sapienza University of Rome, Italy. Valentina Miacci MD Fondazione Policlinico Universitariio Campus Bio-Medico, Università Campus Bio-Medico, Rome, Italy. Andrea Mingoli MD Department of Surgical Sciences, Sapienza University of Rome, Piazzale Aldo Moro 5, 00185 Rome, Italy. Giuseppe Miranda MD Department of General Surgery, Università Politecnica Delle Marche, Ancona, Italy. Savini Occhionorelli MD Department of Translational Medicine, University of Ferrara, Ferrara, Italy. Nadiane Paderno First General Surgery Unit, Bergamo, Italy. Gian M Palini Unit of General and Minimally Invasive Surgery, Infermi Hospital of Rimini, AUSL Della Romagna, Rimini, Italy. Daniele Paradies MD Department of Emergency and Organ Transplant—Minimally invasive and Endocrine Surgery Unit, University Medical School of Bari, Bari, Italy. Luca Petruzzelli MD Department of Emergency Surgery, City of Health and Science, Turin, Italy. Angela Pezzolla MD Department of Emergency and Organ Transplantation, School of Medicine, University of Bari Aldo Moro, Bari, Italy. Diego Piazza MD Department of General Surgery, Santissimo Salvatore Hospital, Paternò, Catania, Italy. Vincenzo Piazza Department of General Surgery, Santissimo Salvatore Hospital, Paternò, Catania, Italy. Giusto Pignata MD General Surgery 2, ASST Spedali Civili of Brescia, Brescia, Italy. E. Pinotti Department of Surgery, Ponte San Pietro Hospital, Bergamo, Italy. Adolfo Pisanu Emergency Surgery Unit, Department of Surgical Science, Policlinico Universitario "D. Casula", Azienda Ospedaliero-Universitaria di Cagliari, University of Cagliari. Caterina Puccioni MD Emergency Surgery and Trauma—Fondazione Policlinico Universitario "A. Gemelli" IRCCS, Rome, Italy. Aldo Rocca MD Department of General Surgery, Pineta Grande Hospital, Castel Volturno, Caserta, Italy. Fabio Rondelli Department of Surgery, School of Medicine, University of Perugia, Perugia, Italy. Paolo Ruscelli MD Department of Emergency Surgery, Azienda Ospedaliero Universitaria-Ospedali Riuniti Ancona, 60126 Ancona, Italy. Paolo Sapienza MD Emergency Department, Policlinico Umberto I, Sapienza University, Rome, Italy. Marco Scattizzi General Surgery Unit, Santa Maria Annunziata & Serristori Hospital, 50012 Firenze, Italy. Angelo Serao Department of General and Emergency Surgery, Ospedale dei Castelli (NOC), ASL Roma 6, 00072 Rome, Italy. Gabriele Sganga MD Emergency Surgery and Trauma—Fondazione Policlinico Universitario "A. Gemelli" IRCCS, Rome, Italy. Dario Tartaglia General and Emergency Surgery Unit, Trauma Center, New Santa Chiara Hospital, University of Pisa. Giovanni Tebala MD Digestive and Emergency Surgery Unit, S.Maria Hospital Trust, Terni, Italy. Cristian Tranà MD Department of Surgery, Macerata Hospital, ASUR Marche, Macerata, Italy. Mauro Zago MD General and Emergency Surgery Department, A. Manzoni Hospital, Lecco, Italy.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, not for-profit sectors.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Disclosures
Drs. Gianluca Costa, Pietro Fransvea, Luca Lepre, Gianluca Liotta, Gianluca Mazzoni, Alan Biloslavo, Valentina Bianchi, Savino Occhionorelli, Alessandro Costa, Gabriele Sganga and the IGo-GIPS study group have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Italian Group for Gastro-Intestinal Surgery Postoperative Surveillance (IGo-Gips) Study Group Collaborators in Acknowledgement section.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Costa, G., Fransvea, P., Lepre, L. et al. Perforated peptic ulcer (PPU) treatment: an Italian nationwide propensity score-matched cohort study investigating laparoscopic vs open approach. Surg Endosc 37, 5137–5149 (2023). https://doi.org/10.1007/s00464-023-09998-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-023-09998-5