Abstract
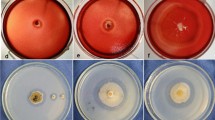
Aspergillus terricola and Aspergillus ochraceus, isolated from Brazilian soil, were cultivated in Vogel and Adams media supplemented with 20 different carbon sources, at 30 °C, under static conditions, for 120 and 144 h, respectively. High levels of cellulase-free xylanase were produced in birchwood or oat spelt xylan-media. Wheat bran was the most favorable agricultural residue for xylanase production. Maximum activity was obtained at 60 °C and pH 6.5 for A. terricola, and 65 °C and pH 5.0 for A. ochraceus. A. terricola xylanase was stable for 1 h at 60 °C and retained 50% activity after 80 min, while A. ochraceus xylanase presented a t50 of 10 min. The xylanases were stable in an alkali pH range. Biobleaching of 10 U/g dry cellulose pulp resulted in 14.3% delignification (A. terricola) and 36.4% (A. ochraceus). The brightness was 2.4–3.4% ISO higher than the control. Analysis in SEM showed defibrillation of the microfibrils. Arabinase traces and β-xylosidase were detected which might act synergistically with xylanase.




Similar content being viewed by others
References
Polizeli MLTM, Rizzatti ACS, Monti R, Terenzi HF, Jorge JA, Amorim DS (2005) Xylanases from fungi: properties and industrial applications. Appl Microbiol Biotechnol 67:577–591
Coelho GD, Carmona EC (2003) Xylanolytic complex from Aspergillus giganteus: production and characterization. J Basic Microbiol 43(4):269–277
Viikari L, Kantelinen A, Sundquist J, Linko M (1994) Xylanases in bleaching: From an idea to the industry. FEMS Microbiol Rev 13:335–350
Bailey MJ, Viikari L (1993) Effect of pH on production of xylanase by Trichoderma reesei on xylan and cellulose-based media. Appl Microbiol Biotechnol 40:224–229
Christov LP, Szakacs G, Balakrishnan H (1999) Production, partial characterization and use of fungal cellulase-free xylanases in pulp bleaching. Process Biochem 34:511–517
Bhat MK (2000) Cellulases and related enzymes in biotechnology. Biotech Adv 18:355–383
Rizzatti ACS, Jorge JA, Terenzi HF, Rechia CGV, Polizeli MLTM (2001) Purification and properties of a thermostable extracellular β-D-xylosidase produced by a thermotolerant Aspergillus phoenicis. J Ind Microbiol Biotechnol 4:265–275
Adams PR (1990) Mycelial amylase activities of thermophilic species of Rhizomucor, Humicola and Papulaspora. Mycopathologia 112:35–37
Khanna P, Sundari SS, Kumar NJ (1995) Production, isolation and partial purification of xylanase from Aspergillus sp. World J Microbiol Biotechnol 11:242–243
Wiseman A (1975) Handbook of enzyme biotechnology. Wiley, New York
Vogel HJ (1964) Distribution of lysine pathways among fungi: evolutionary implications. Am Nat 98:435–446
Peralta RM, Terenzi HF, Jorge JA (1990) β-D-Glycosidase activities of Humicola grisea: biochemical and kinetic characterization of a multifunctional enzyme. Biochem Biophys Acta 1033:243–249
Lowry OH, Rosebrough NJ, Farr AL, Randal RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Miller GH (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–429
Kersters-Hilderson H, Claeyssens M, Doorslaer EV, Saman E, Bruyne CK (1982) β-D xylosidase from Bacillus pumilus. Methods Enzymol 83:631–639
Test Methods TAPPI (1996) Technical association of the pulp and paper industry. TAPPI Press, Atlanta
Kulkarni N, Shendye A, Rao M (1999) Molecular and biotechnological aspects of xylanases. FEMS Microbiol Rev 23:411–456
Shah AR, Madamwar D (2005) Xylanase production by a newly isolated Aspergillus foetidus strain and its characterization. Process Biochem 40:1763–1771
Rizzatti ACS, Sandrim VC, Jorge JA, Terenzi HF, Polizeli MLTM (2004) Influence of temperature on the properties of the xylanolytic enzymes of the thermotolerant fungus Aspergillus phoenicis. J Ind Microbiol Biotechnol 31:88–93
Sandrim VC, Rizzatti ACS, Terenzi HF, Jorge JA, Milagres AMF, Polizeli MLTM (2005) Purification and biochemical characterization of two xylanases produced by Aspergillus caespitosus and their potential for kraft pulp bleaching. Process Biochem 40:1823–1828
Peixoto-Nogueira SC, Michelin M, Betini JHA, Jorge JA, Terenzi HF, Polizeli MLTM (2009) Production of xylanase by Aspergilli using alternative carbon sources: application of the crude extract on cellulose pulp biobleaching. J Ind Microbiol Biotechnol 36(1):149–155
Marques de Souza CG, Girardo NS, Costa MAF, Peralta RM (1999) Influence of growth conditions on the production of xylanolytic enzymes by Aspergillus flavus. J Basic Microbiol 39(3):155–160
Yuan Q, Wang J, Zhang H, Qian Z (2005) Effect of temperature shift on production of xylanase by Aspergillus niger. Process Biochem 40:3255–3257
Fang HY, Chang SM, Hsieh MC, Fang TJ (2007) Production, optimization growth conditions and properties of the xylanase from Aspergillus carneus M34. J Mol Catal B Enzym 49:36–42
Dobrev GT, Pishtiyski IG, Stanchev VS, Mircheva R (2007) Optimization of nutrient medium containing agricultural wastes for xylanase production by Aspergillus niger B03 using optimal composite experimental design. Bioresour Technol 98:2671–2678
Souza DF, Souza CGM, Peralta RM (2001) Effect of easily metabolizable sugars in the production of xylanase by Aspergillus tamarii in solid-state fermentation. Process Biochem 36:835–838
Carmona EC, Brochetto-Braga MR, Pizzirani-Kleiner AA, Jorge JA (1998) Purification and biochemical characterization of an endoxylanase from Aspergillus versicolor. FEMS Microbiol Lett 166:311–315
Techapun C, Poosaran N, Watanabe M, Sasaki K (2003) Thermostable and alkaline-tolerant microbial cellulase-free xylanases produced from agricultural wastes and properties required for use in pulp bleaching bioprocess: a review. Process Biochem 38:1327–1340
Bim MA, Franco TT (2000) Extraction in aqueous two-phase systems of alkaline xylanase produced by Bacillus pumilus and its application in kraft pulp bleaching. J Chromatogr B 743:349–356
Duarte MCT, Silva EC, Gomes IMB, Ponezi AN, Portugal EP, Vicente JR, Davanzo E (2003) Xylan-hydrolyzing enzyme system from Bacillus pumilus CBMAI 0008 and its effects on Eucalyptus grandis kraft pulp for pulp bleaching improvement. Bioresour Technol 88:9–15
Raghukumar C, Muraleedharan U, Gaud VR, Mishra R (2004) Xylanases of marine fungi of potential use for biobleaching of paper pulp. J Ind Microbiol Biotechnol 31:433–441
Betini JHA, Michelin M, Peixoto-Nogueira, SC, Jorge JA, Terenzi HF, Polizeli MLTM (2009) Xylanases from Aspergillus niger, Aspergillus niveus and Aspergillus ochraceus produced under solid-state fermentation and their application in cellulose pulp bleaching. Bioprocess Biosyst Eng 32:819–824. doi:10.1007/s00449-009-0308-y
Salles BC, Medeiros RG, Bao SN, Silva FG Jr, Filho EXF (2005) Effect of celulase-free xylanases from Acrophialophora nainiana and Humicola grisea var. thermoidea on eucalyptus kraft pulp. Process Biochem 40:343–349
Bajpai P (1999) Application of enzymes in the pulp and paper industry. Biotechnol Prog 15:147–157
Viikari L, Suurnaki A, Buchert J (1996) Enzyme-aided bleaching of kraft pulp: fundamental mechanisms and practical applications. In: Jeffries TW, Viikari L (eds) Enzyme pulp and paper processing. ACS Symposium Series 655, Washington, DC
Polizeli MLTM (2009) Properties and commercial applications of xylanases from fungi. In: Rai M (ed) Advances in fungal biotechnology. I.K. International Publisher, New Delhi
Acknowledgments
This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and the Conselho de Desenvolvimento Científico e Tecnológico (CNPq). J.A.J., H.F.T., and M.L.T.M.P. are research fellows of CNPq. M.M. is a recipient of a FAPESP fellowship, and this work is part of her doctoral thesis. We thank Ricardo Alarcon and Maurício de Oliveira for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Michelin, M., Peixoto-Nogueira, S.C., Betini, J.H.A. et al. Production and properties of xylanases from Aspergillus terricola Marchal and Aspergillus ochraceus and their use in cellulose pulp bleaching. Bioprocess Biosyst Eng 33, 813–821 (2010). https://doi.org/10.1007/s00449-009-0403-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-009-0403-0