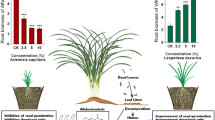
Abstract
Allelopathy has a profound impact on the germination and growth of plants, influencing the establishment of plant populations and shaping community ecological patterns. However, the allelopathic potential of many grassland species remains poorly understood. In this study, we prepared aqueous extracts from 17 herbaceous plants to investigate their allelopathic effects on the seed germination and seedling growth of Leymus chinensis, a dominant grassland species. Our results revealed that the response of L. chinensis to allelopathic compounds was dependent on the specific plant species, extract concentration, and target plant organ. Notably, Fabaceae plants exhibited a stronger allelopathic potential than Poaceae, Asteraceae, and other plant families. Moreover, we observed that root growth of L. chinensis was more sensitive to allelopathy than shoot growth, and seed germination was more affected than seedling growth. Generally, the germination of L. chinensis was strongly inhibited as the donor plant extract concentration increased. The leachate of Fabaceae plants inhibited the seedling growth of L. chinensis at concentrations ranging from 0.025 to 0.1 g mL−1. On the other hand, the leachate from other families’ plants exhibited either inhibitory or hormetic effects on the early growth of L. chinensis, promoting growth at 0.025 g mL−1 and hindering it at concentrations between 0.05 and 0.1 g mL−1. These findings highlight the significant allelopathic potential of grassland plants, which plays a critical role in establishing plant populations and associated ecological processes. In addition, they shed light on the coexistence of other plants with dominant plants in the community.






Similar content being viewed by others
Availability of data and material
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
References
Andersen CP, King G, Plocher M et al (2016) Germination and early plant development of ten plant species exposed to titanium dioxide and cerium oxide nanoparticles. Environ Toxicol Chem 35:2223–2229. https://doi.org/10.1002/etc.3374
Bertness MD, Callaway R (1994) Positive interactions in communities. Trends Ecol Evol 9:191–193. https://doi.org/10.1016/0169-5347
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889. https://doi.org/10.2307/2261425
Bradford KJ (2002) Applications of hydrothermal time to quantifying and modeling seed germination and dormancy. Weed Sci 50:248–260. https://doi.org/10.1614/0043-1745(2002)050[0248:aohttq]2.0.co;2
Bruce Williamson G, Richardson D (1988) Bioassays for allelopathy: Measuring treatment responses with independent controls. J Chem Ecol 14:181–187. https://doi.org/10.1007/bf01022540
Callaway RM, Walker LR (1997) Competition and facilitation: a synthetic approach to interactions in plant communities. Ecology 78:1958–1965. https://doi.org/10.2307/2265936
Callaway RM, Brooker RW, Choler P et al (2002) Positive interactions among alpine plants increase with stress. Nature 417:844–848. https://doi.org/10.1038/nature00812
Chen L, Li J, Zhu Y et al (2022) Caffeic acid, an allelochemical in Artemisia argyi, inhibits weed growth via suppression of mitogen-activated protein kinase signaling pathway and the biosynthesis of gibberellin and phytoalexin. Front Plant Sci 12:802198. https://doi.org/10.3389/fpls.2021.802198
Chen L, Yang XG, Wang L, Song NP (2017) Allelopathic effects of Caragana intermedia on monocot and dicot plant species and identification of allelochemical. AJ 42:253–264. https://doi.org/10.26651/allelo.j./2017-42-2-1121
Cheng F, Cheng Z (2015) Research progress on the use of plant allelopathy in agriculture and the physiological and ecological mechanisms of allelopathy. Front Plant Sci 6:1020. https://doi.org/10.3389/fpls.2015.01020
Chon S-U, Nelson CJ (2010) Allelopathy in Compositae plants. A Review Agron Sustain Dev 30:349–358. https://doi.org/10.1051/agro/2009027
Chon S-U, Kim Y-M, Lee J-C (2003) Herbicidal potential and quantification of causative allelochemicals from several Compositae weeds. Weed Res 43:444–450. https://doi.org/10.1046/j.0043-1737.2003.00361.x
Connell JH (1983) On the prevalence and relative importance of interspecific competition: evidence from field experiments. Am Nat 122:661–696. https://doi.org/10.1086/284165
Dai Z-C, Wang X-Y, Qi S-S et al (2016) Effects of leaf litter on inter-specific competitive ability of the invasive plant Wedelia trilobata. Ecol Res 31:367–374. https://doi.org/10.1007/s11284-016-1344-0
Dar BA, Al-Rowaily SL, Assaeed AM et al (2017) Allelopathic potential of Argemone ochroleuca from different habitats on seed germination of native species and cultivated crops. Pak J Bot 5:1841–1848
Đorđević T, Đurović-Pejčev R, Stevanović M, et al (2022) Phytotoxicity and allelopathic potential of Juglans regia L. leaf extract. Front Plant Sci 13:986740. https://doi.org/10.3389/fpls.2022.986740
Einhellig FA (1994) Mechanism of action of allelochemicals in allelopathy. American Chemical Society, Washington, DC, In., pp 96–116
Ens EJ, French K, Bremner JB (2008) Evidence for allelopathy as a mechanism of community composition change by an invasive exotic shrub, Chrysanthemoides monilifera spp. rotundata. Plant Soil 316(1–2):125–137. https://doi.org/10.1007/s11104-008-9765-3
Ghimire BK, Ghimire B, Yu CY, Chung I-M (2019) Allelopathic and autotoxic effects of Medicago sativa—derived allelochemicals. Plants 8:233. https://doi.org/10.3390/plants8070233
Herranz JM, Ferrandis P, Copete MA et al (2005) Effect of allelopathic compounds produced by Cistus ladanifer on germination of 20 Mediterranean taxa. Plant Ecol 184:259–272. https://doi.org/10.1007/s11258-005-9071-6
Islam A, KatoNoguchi H (2013) Plant growth inhibitory activity of medicinal plant Hyptis suaveolens: could allelopathy be a cause? Emir J Food Agric 25:692. https://doi.org/10.9755/ejfa.v25i9.16073
Lankau RA (2010) Intraspecific variation in allelochemistry determines an invasive species’ impact on soil microbial communities. Oecologia 165:453–463. https://doi.org/10.1007/s00442-010-1736-8
Li XF, Wang J, Huang D et al (2010) Allelopathic potential of Artemisia frigida and successional changes of plant communities in the northern China steppe. Plant Soil 341:383–398. https://doi.org/10.1007/s11104-010-0652-3
Liu G, Li X, Qi D et al (2016) Evaluation and utilization of Leymus chinensis germplasm resources. Chin Sci Bull 61:271–281
Liu GS, Qi DM, Shu QY (2004) Seed germination characteristics in the perennial grass species Leymus chinensis. Seed Sci Technol 32:717–725. https://doi.org/10.15258/sst.2004.32.3.07
Liu G, Wang D, Shi F et al (2022) Review of study on germplasm resources of Leymus chinensis and its inspirations. Chin J Grassl 44:1–9. https://doi.org/10.16742/j.zgcdxb.20220043
Liu S, Ma Z, Zhang Y et al (2022b) Astragalus sinicus incorporated as green manure for weed control in corn. Front Plant Sci 13:829421. https://doi.org/10.3389/fpls.2022.829421
Ma H-Y, Liang Z-W, Wang Z-C et al (2008) Lemmas and endosperms significantly inhibited germination of Leymus chinensis (Trin.) Tzvel. (Poaceae). J Arid Environ 72:573–578. https://doi.org/10.1016/j.jaridenv.2007.06.013
Meiners SJ (2014) Functional correlates of allelopathic potential in a successional plant community. Plant Ecol 215:661–672. https://doi.org/10.1007/s11258-014-0331-1
Meiners SJ, Kong C-H, Ladwig LM et al (2012) Developing an ecological context for allelopathy. Plant Ecol 213:1221–1227. https://doi.org/10.1007/s11258-012-0078-5
Muller CH (1966) The role of chemical inhibition (allelopathy) in vegetational composition. Bull Torrey Bot Club 93:332. https://doi.org/10.2307/2483447
Nakahisa K, Tsuzuki E, Mitsumizo T (1993) Study on the allelopathy of alfalfa (Medicago sativa L.): I. Observation of allelopathy and survey for substances inducing growth inhibition. JPN J Crop Sci 62:294–299. https://doi.org/10.1626/jcs.62.294
Nambara E, Okamoto M, Tatematsu K et al (2010) Abscisic acid and the control of seed dormancy and germination. Seed Sci Res 20:55–67. https://doi.org/10.1017/s0960258510000012
Nandula VK, Eubank TW, Poston DH et al (2006) Factors affecting germination of horseweed (Conyza canadensis). Weed Sci 54:898–902. https://doi.org/10.1614/ws-06-006r2.1
Newman EI, Rovira AD (1975) Allelopathy Among Some British Grassland Species. J Ecol 63:727. https://doi.org/10.2307/2258598
Nishida N, Tamotsu S, Nagata N et al (2005) Allelopathic effects of volatile monoterpenoids produced by Salvia leucophylla: Inhibition of cell proliferation and DNA synthesis in the root apical meristem of Brassica campestris seedlings. J Chem Ecol 31:1187–1203. https://doi.org/10.1007/s10886-005-4256-y
Omezzine F, Haouala R (2013) Effect of Trigonella foenum-graecum L. development stages on some phytochemicals content and allelopathic potential. Sci Hortic 160:335–344. https://doi.org/10.1016/j.scienta.2013.06.023
Pellissier F (1994) Effect of phenolic compounds in humus on the natural regeneration of spruce. Phytochemistry 36:865–867. https://doi.org/10.1016/s0031-9422(00)90452-7
Quan X, Qiao Y, Chen M et al (2021) Comprehensive evaluation of the allelopathic potential of Elymus nutans. Ecol Evol 11:12389–12400. https://doi.org/10.1002/ece3.7982
Quayyum HA, Mallik AU, Lee PF (1999) Allelopathic potential of aquatic plants associated with wild rice (Zizania palustris): I. Bioassay with plant and lake sediment samples. J Chem Ecol 25:209–220. https://doi.org/10.1023/a:1020853620102
Rasmussen JA, Einhellig FA (1977) Synergistic inhibitory effects of p-coumaric and ferulic acids on germination and growth of grain sorghum. J Chem Ecol 3:197–205. https://doi.org/10.1007/bf00994146
Reigosa MJ, Sánchez-Moreiras A, González L (1999) Ecophysiological approach in allelopathy. CRC Crit Rev Plant Sci 18:577–608. https://doi.org/10.1016/s0735-2689(99)00392-5
Rice EL (1984) Allelopathy. Academic Press, New York
Roberts EH (1986) Seeds. Physiology of development and germination (book). Plant Cell Environ 9:356–356. https://doi.org/10.1111/1365-3040.ep11611812
Rudrappa T, Bonsall J, Gallagher JL et al (2007) Root-secreted allelochemical in the noxious weed Phragmites Australis deploys a reactive oxygen species response and microtubule assembly disruption to execute rhizotoxicity. J Chem Ecol 33:1898–1918. https://doi.org/10.1007/s10886-007-9353-7
Russo VM, Webber CL, Myers DL (1997) Kenaf extract affects germination and post-germination development of weed, grass and vegetable seeds. Ind Crop Prod 6:59–69. https://doi.org/10.1016/s0926-6690(96)00206-3
Schandry N, Becker C (2020) Allelopathic plants: models for studying plant–interkingdom interactions. Trends Plant Sci 25:176–185. https://doi.org/10.1016/j.tplants.2019.11.004
Shi X, Huang H, Zhou M et al (2021) Paeonol attenuated vascular fibrosis through regulating Treg/Th17 balance in a gut microbiota-dependent manner. Front Pharmacol 12:765482. https://doi.org/10.3389/fphar.2021.765482
Svensson JR, Nylund GM, Cervin G et al (2012) Novel chemical weapon of an exotic macroalga inhibits recruitment of native competitors in the invaded range. J Ecol 101:140–148. https://doi.org/10.1111/1365-2745.12028
Teerarak M, Laosinwattana C, Charoenying P (2010) Evaluation of allelopathic, decomposition and cytogenetic activities of Jasminum officinale L. f. var. grandiflorum (L.) Kob. on bioassay plants. Bioresour Technol 101:5677–5684. https://doi.org/10.1016/j.biortech.2010.02.038
Thorpe AS, Thelen GC, Diaconu A, Callaway RM (2009) Root exudate is allelopathic in invaded community but not in native community: field evidence for the novel weapons hypothesis. J Ecol 97:641–645. https://doi.org/10.1111/j.1365-2745.2009.01520.x
Tilman D (1982) Resource competition and community structure. Princeton University Press, Princeton. https://doi.org/10.1515/9780691209654
Tilman D (1994) Competition and biodiversity in spatially structured habitats. Ecology 75:2–16. https://doi.org/10.2307/1939377
Wang C, Xiao H, Zhao L et al (2016) The allelopathic effects of invasive plant Solidago canadensis on seed germination and growth of Lactuca sativa enhanced by different types of acid deposition. Ecotoxicology 25:555–562. https://doi.org/10.1007/s10646-016-1614-1
Wang C, Jiang K, Wu B et al (2018a) Silver nanoparticles with different particle sizes enhance the allelopathic effects of Canada goldenrod on the seed germination and seedling development of lettuce. Ecotoxicology 27:1116–1125. https://doi.org/10.1007/s10646-018-1966-9
Wang C, Wu B, Jiang K (2018b) Allelopathic effects of Canada goldenrod leaf extracts on the seed germination and seedling growth of lettuce reinforced under salt stress. Ecotoxicology 28:103–116. https://doi.org/10.1007/s10646-018-2004-7
Wang X, Wang J, Zhang R et al (2018c) Allelopathic effects of aqueous leaf extracts from four shrub species on seed germination and initial growth of Amygdalus pedunculata Pall. Forests 9:711. https://doi.org/10.3390/f9110711
Wang X, Zhang R, Wang J et al (2021) The effects of leaf extracts of four tree species on Amygdalus pedunculata seedlings growth. Front Plant Sci 11:587579. https://doi.org/10.3389/fpls.2020.587579
Wang C, Liu Z, Wang Z et al (2022a) Effects of autotoxicity and allelopathy on seed germination and seedling growth in Medicago truncatula. Front Plant Sci 13:908426. https://doi.org/10.3389/fpls.2022.908426
Wang K, Wang T, Ren C et al (2022b) Aqueous extracts of three herbs allelopathically inhibit lettuce germination but promote seedling growth at low concentrations. Plants 11:486. https://doi.org/10.3390/plants11040486
Williams RD, Hoagland RE (1982) The effects of naturally occurring phenolic compounds on seed germination. Weed Sci 30:206–212. https://doi.org/10.1017/s0043174500062342
Wu A-P, Li Z-L, He F-F et al (2015) Screening allelochemical-resistant species of the alien invasive Mikania micrantha for restoration in South China. PLoS ONE 10:e0132967. https://doi.org/10.1371/journal.pone.0132967
Yamaguchi S (2008) Gibberellin metabolism and its regulation. Annu Rev Plant Biol 59:225–251. https://doi.org/10.1146/annurev.arplant.59.032607.092804
Yang Y, Liu G, Zhang B (1995) An analysis of age structure and the strategy for asexual propagation of Aneurolepidium chinense population. Acta Bot Sin 37:147–153
Yu F, Huang X, Ma Y (2008) Allelopathic effect of different parts and plants of Astragalus adsurgens at different growth stages. Acta Prat Sin 17:76–83
Xu Y, Chen X, Ding L, Kong C-H (2023) Allelopathy and allelochemicals in grasslands and forests. Forests 14:562. https://doi.org/10.3390/f14030562
Zhang S, Jin Y, Zhu W et al (2010) Baicalin released from Scutellaria baicalensis induces autotoxicity and promotes soilborn pathogens. J Chem Ecol 36:329–338. https://doi.org/10.1007/s10886-010-9760-z
Zhang RM, Zuo ZJ, Gao PJ et al (2012) Allelopathic effects of VOCs of Artemisia frigida Willd. on the regeneration of pasture grasses in Inner Mongolia. J Arid Environ 87:212–218. https://doi.org/10.1016/j.jaridenv.2012.04.008
Zhang Y, Tang S, Liu K et al (2014) The allelopathic effect of Potentilla acaulis on the changes of plant community in grassland, northern China. Ecol Res 30:41–47. https://doi.org/10.1007/s11284-014-1203-9
Zhang Z, Liu Y, Yuan L et al (2020) Effect of allelopathy on plant performance: a meta-analysis. Ecol Lett 24:348–362. https://doi.org/10.1111/ele.13627
Zheng Y-L, Feng Y-L, Zhang L-K et al (2014) Integrating novel chemical weapons and evolutionarily increased competitive ability in success of a tropical invader. New Phytol 205:1350–1359. https://doi.org/10.1111/nph.13135
Zuo Z-J, Zhang R-M, Gao P-J et al (2011) Allelopathic effects of Artemisia frigida Willd. on growth of pasture grasses in Inner Mongolia. China Biochem Syst Ecol 39:377–383. https://doi.org/10.1016/j.bse.2011.05.010
Acknowledgements
We thank Jinying Lvu for her assistance with the experimental equipment provided and the anonymous reviewers for their helpful comments.
Funding
This research was funded by the National Key Research and Development Program of China, grant number 2018YFF0213405, and the National Natural Science Foundation of China, grant number 31772654.
Author information
Authors and Affiliations
Contributions
KW (Kaili Wang) and KW (Kun Wang) conceived and designed the experiments. KW (Kaili Wang), PD, ZM, JH, and QG performed the experiments. KW (Kaili Wang) analyzed the data and wrote the manuscript. KL and LG provided revised advice. YR and DH coordinated the project. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Additional information
Communicated by Wentao Luo.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, K., Dou, P., Miao, Z. et al. Seed germination and seedling growth response of Leymus chinensis to the allelopathic influence of grassland plants. Oecologia 204, 899–913 (2024). https://doi.org/10.1007/s00442-024-05539-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-024-05539-6