Abstract
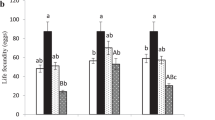
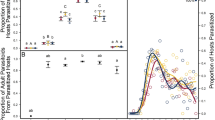
Temperature alters host suitability for parasitoid development through direct and indirect pathways. Direct effects depend on ambient temperatures experienced by a single host individual during its lifetime. Indirect effects (or parental effects) occur when thermal conditions met by a host parental generation affect the way its offspring will interact with parasitoids. Using the complex involving eggs of the moth Lobesia botrana as hosts for the parasitoid Trichogramma cacoeciae, we developed an experimental design to disentangle the effects of (1) host parental temperature (temperature at which the host parental generation developed and laid host eggs) and (2) host offspring temperature (temperature at which host eggs were incubated following parasitism, i.e. direct thermal effects) on this interaction. The host parental generation was impacted by temperature experienced during its development: L. botrana females exposed to warmer conditions displayed a lower pupal mass but laid more host eggs over a 12-h period. Host parental temperature also affected the outcomes of the interaction. Trichogramma cacoeciae exhibited lower emergence rates but higher hind tibia length on emergence from eggs laid under warm conditions, even if they were themselves exposed to cooler temperatures. Such indirect thermal effects might arise from a low nutritional quality and/or a high immunity of host eggs laid in warm conditions. By contrast with host parental temperature, offspring temperature (direct thermal effects) did not significantly affect the outcomes of the interaction. This work emphasises the importance of accounting for parental thermal effects to predict the future of trophic dynamics under global warming scenarios.



Similar content being viewed by others
References
Abdel-latief M, Hilker M (2008) Innate immunity: eggs of Manduca sexta are able to respond to parasitism by Trichogramma evanescens. Insect Biochem Mol Biol 38:136–145. https://doi.org/10.1016/J.IBMB.2007.10.001
Bahar MH, Soroka JJ, Dosdall LM (2012) Constant versus fluctuating temperatures in the interactions between Plutella xylostella (Lepidoptera: Plutellidae) and its larval parasitoid Diadegma insulare (Hymenoptera: Ichneumonidae). Environ Entomol 41:1653–1661. https://doi.org/10.1603/EN12156
Barnay O, Hommay G, Gertz C, Kienlen JC, Schubert G, Marro JP, Pizzol J, Chavigny P (2001) Survey of natural populations of Trichogramma (Hym., Trichogrammatidae) in the vineyards of Alsace (France). J Appl Entomol 125:469–477. https://doi.org/10.1046/j.1439-0418.2001.00575.x
Beaumont LJ, Hughes L, Pitman AJ (2008) Why is the choice of future climate scenarios for species distribution modelling important? Ecol Lett 11:1135–1146. https://doi.org/10.1111/j.1461-0248.2008.01231.x
Delava E, Fleury F, Gibert P (2016) Effects of daily fluctuating temperatures on the Drosophila–Leptopilina boulardi parasitoid association. J Therm Biol 60:95–102. https://doi.org/10.1016/j.jtherbio.2016.06.012
Donelson JM, Salinas S, Munday PL, Shama LNS (2018) Transgenerational plasticity and climate change experiments: where do we go from here? Glob Change Biol 24:13–34. https://doi.org/10.1111/gcb.13903
Doyon J, Boivin G (2005) The effect of development time on the fitness of female Trichogramma evanescens. J Insect Sci 5:4–9. https://doi.org/10.1093/jis/5.1.4
Eggert H, Diddens-de Buhr MF, Kurtz J (2015) A temperature shock can lead to trans-generational immune priming in the red flour beetle, Tribolium castaneum. Ecol Evol 5:1318–1326. https://doi.org/10.1002/ece3.1443
Fournier F, Boivin G (2000) Comparative dispersal of Trichogramma evanescens and Trichogramma pretiosum (Hymenoptera: Trichogrammatidae) in relation to environmental conditions. Environ Entomol 29:55–63. https://doi.org/10.1603/0046-225X-29.1.55
Furlong MJ, Zalucki MP (2017) Climate change and biological control: the consequences of increasing temperatures on host–parasitoid interactions. Curr Opin Insect Sci 20:39–44. https://doi.org/10.1016/j.cois.2017.03.006
Geister TL, Lorenz MW, Hoffmann KH, Fischer K (2009) Energetics of embryonic development: effects of temperature on egg and hatchling composition in a butterfly. J Comp Physiol B 179:87–98. https://doi.org/10.1007/s00360-008-0293-5
Godfray HCJ (1994) Parasitoids: behavioral and evolutionary ecology. Princeton University Press, Princeton
Hance T, van Baaren J, Vernon P, Boivin G (2007) Impact of extreme temperatures on parasitoids in a climate change perspective. Annu Rev Entomol 52:107–126. https://doi.org/10.1146/annurev.ento.52.110405.091333
Harris RMB, Grose MR, Lee G, Bindoff NL, Porfirio LL, Fox-Hughes P (2014) Climate projections for ecologists. Wires Clim Change 5:621–637. https://doi.org/10.1002/wcc.291
Howe RW (1967) Temperature effects on embryonic development in insects. Annu Rev Entomol 12:15–42. https://doi.org/10.1146/annurev.en.12.010167.000311
Hutchison WD, Moratorio M, Martin JM (1990) Morphology and biology of Trichogrammatoidea bactrae (Hymenoptera: Trichogrammatidae), imported from Australia as a parasitoid of pink bollworm (Lepidoptera: Gelechiidae) eggs. Ann Entomol Soc Am 83:46–54. https://doi.org/10.1093/aesa/83.1.46
Iltis C, Martel G, Thiéry D, Moreau J, Louâpre P (2018) When warmer means weaker: high temperatures reduce behavioural and immune defences of the larvae of a major grapevine pest. J Pest Sci 91:1315–1326. https://doi.org/10.1007/s10340-018-0992-y
Iltis C, Louâpre P, Pecharová K, Thiéry D, Zito S, Bois B, Moreau J (2019) Are life-history traits equally affected by global warming? A case study combining a multi-trait approach with fine-grain climate modeling. J Insect Physiol 117:103916. https://doi.org/10.1016/j.jinsphys.2019.103916
Janowitz SA, Fischer K (2011) Opposing effects of heat stress on male versus female reproductive success in Bicyclus anynana butterflies. J Therm Biol 36:283–287. https://doi.org/10.1016/J.JTHERBIO.2011.04.001
Jeffs CT, Lewis OT (2013) Effects of climate warming on host–parasitoid interactions. Ecol Entomol 38:209–218. https://doi.org/10.1111/een.12026
Kishani Farahani H, Ashouri A, Zibaee A, Abroon P, Alford L (2016) The effect of host nutritional quality on multiple components of Trichogramma brassicae fitness. Bull Entomol Res 106:633–641. https://doi.org/10.1017/S000748531600033X
Moreau J, Benrey B, Thiéry D (2006) Grape variety affects larval performance and also female reproductive performance of the European grapevine moth Lobesia botrana (Lepidoptera: Tortricidae). Bull Entomol Res 96:205–212. https://doi.org/10.1079/BER2005417
Moreau J, Richard A, Benrey B, Thiéry D (2009) Host plant cultivar of the grapevine moth Lobesia botrana affects the life history traits of an egg parasitoid. Biol Control 50:117–122. https://doi.org/10.1016/j.biocontrol.2009.03.017
Moreau J, Monceau K, Thiéry D (2016) Larval food influences temporal oviposition and egg quality traits in females of Lobesia botrana. J Pest Sci 89:439–448. https://doi.org/10.1007/s10340-015-0695-6
Moreno F, Pérez-Moreno I, Marco V (2009) Effects of Lobesia botrana (Lepidoptera: Tortricidae) egg age, density, and UV treatment on parasitism and development of Trichogramma cacoeciae (Hymenoptera: Trichogrammatidae). Environ Entomol 38:1513–1520. https://doi.org/10.1603/022.038.0520
Muller K, Arenas L, Thiéry D, Moreau J (2016) Direct benefits from choosing a virgin male in the European grapevine moth, Lobesia botrana. Anim Behav 114:165–172. https://doi.org/10.1016/J.ANBEHAV.2016.02.005
Nardi JB (2004) Embryonic origins of the two main classes of hemocytes—granular cells and plasmatocytes—in Manduca sexta. Dev Genes Evol 214:19–28. https://doi.org/10.1007/s00427-003-0371-3
Olson DM, Andow DA (1998) Larval crowding and adult nutrition effects on longevity and fecundity of female Trichogramma nubilale Ertle & Davis (Hymenoptera: Trichogrammatidae). Environ Entomol 27:508–514. https://doi.org/10.1093/ee/27.2.508
Pizzol J, Pintureau B (2008) Effect of photoperiod experienced by parents on diapause induction in Trichogramma cacoeciae. Entomol Exp Appl 127:72–77. https://doi.org/10.1111/j.1570-7458.2008.00671.x
Pizzol J, Khoualdia O, Ferran A, Chavigny P, Vanlerbeghe-Masutti F (2005) A single molecular marker to distinguish between strains of Trichogramma cacoeciae. Biocontrol Sci Technol 15:527–531. https://doi.org/10.1080/09583150500088934
Pizzol J, Desneux N, Wajnberg E, Thiéry D (2012) Parasitoid and host egg ages have independent impact on various biological traits in a Trichogramma species. J Pest Sci 85:489–496. https://doi.org/10.1007/s10340-012-0434-1
Pomfret JC, Knell RJ (2006) Immunity and the expression of a secondary sexual trait in a horned beetle. Behav Ecol 17:466–472. https://doi.org/10.1093/beheco/arj050
Schreven SJJ, Frago E, Stens A, de Jong PW, van Loon JPA (2017) Contrasting effects of heat pulses on different trophic levels, an experiment with a herbivore-parasitoid model system. PLoS One 12:e0176704. https://doi.org/10.1371/journal.pone.0176704
Seehausen LM, Cusson M, Régnière J, Bory M, Stewart D, Djoumad A, Smith SM, Martel V (2017) High temperature induces downregulation of polydnavirus gene transcription in lepidopteran host and enhances accumulation of host immunity gene transcripts. J Insect Physiol 98:126–133. https://doi.org/10.1016/J.JINSPHYS.2016.12.008
Sgrò CM, Terblanche JS, Hoffmann AA (2016) What can plasticity contribute to insect responses to climate change? Annu Rev Entomol 61:433–451. https://doi.org/10.1146/annurev-ento-010715-023859
Stireman JO, Dyer LA, Janzen DH, Singer MS, Lill JT, Marquis J, Ricklefs RE, Gentry GL, Hallwachs W, Coley PD, Barone JA, Greeney HF, Connahs H, Barbosa P, Morais HC, Diniz IR (2005) Climatic unpredictability and parasitism of caterpillars: implications of global warming. Proc Natl Acad Sci USA 102:17384–17387. https://doi.org/10.1073/pnas.0508839102
Strand MR, Pech LL (1995) Immunological basis for compatibility in parasitoid–host relationships. Annu Rev Entomol 40:31–56. https://doi.org/10.1146/annurev.en.40.010195.000335
Thiéry D, Desneux N (2018) Host plants of the polyphagous grape berry moth Lobesia botrana during larval stage modulate moth egg quality and subsequent parasitism by the parasitoid Trichogramma cacoeciae. Entomol Gen 38:47–59. https://doi.org/10.1127/entomologia/2018/0675
Thiéry D, Moreau J (2005) Relative performance of European grapevine moth (Lobesia botrana) on grapes and other hosts. Oecologia 143:548–557. https://doi.org/10.1007/s00442-005-0022-7
Thiéry D, Monceau K, Moreau J (2014) Larval intraspecific competition for food in the European grapevine moth Lobesia botrana. Bull Entomol Res 104:517–524. https://doi.org/10.1017/S0007485314000273
Thiéry D, Louâpre P, Muneret L, Rusch A, Sentenac G, Vogelweith F, Iltis C, Moreau J (2018) Biological protection against grape berry moths. A review. Agron Sustain Dev 38:15. https://doi.org/10.1007/s13593-018-0493-7
Thomson LJ, Macfadyen S, Hoffmann AA (2010) Predicting the effects of climate change on natural enemies of agricultural pests. Biol Control 52:296–306. https://doi.org/10.1016/J.BIOCONTROL.2009.01.022
Trauer-Kizilelma U, Hilker M (2015) Impact of transgenerational immune priming on the defence of insect eggs against parasitism. Dev Comp Immunol 51:126–133. https://doi.org/10.1016/J.DCI.2015.03.004
Triggs AM, Knell RJ (2012) Parental diet has strong transgenerational effects on offspring immunity. Funct Ecol 26:1409–1417. https://doi.org/10.1111/j.1365-2435.2012.02051.x
van Baaren J, Le Lann C, van Alphen J (2010) Consequences of climate change for aphid-based multi-trophic systems. In: Kindlmann P, Dixon AFG, Michaud JP (eds) Aphid biodiversity under environmental change. Springer, Dordrecht, pp 55–68. https://doi.org/10.1007/978-90-481-8601-3_4
Vinson SB, Iwantsch GF (1980) Host suitability for insect parasitoids. Annu Rev Entomol 25:397–419. https://doi.org/10.1146/annurev.en.25.010180.002145
Visser B, Ellers J (2008) Lack of lipogenesis in parasitoids: a review of physiological mechanisms and evolutionary implications. J Insect Physiol 54:1315–1322. https://doi.org/10.1016/J.JINSPHYS.2008.07.014
Vogelweith F, Thiéry D, Moret Y, Colin E, Motreuil S, Moreau J (2014) Defense strategies used by two sympatric vineyard moth pests. J Insect Physiol 64:54–61. https://doi.org/10.1016/j.jinsphys.2014.03.009
Walker M, Jones TH (2001) Relative roles of top–down and bottom–up forces in terrestrial tritrophic plant-insect herbivore-natural enemy systems. Oikos 93:177–187. https://doi.org/10.1034/j.1600-0706.2001.930201.x
Woestmann L, Saastamoinen M (2016) The importance of trans-generational effects in Lepidoptera. Curr Zool 62:489–499. https://doi.org/10.1093/cz/zow029
Acknowledgements
We are grateful to thank Géraldine Groussier for providing the parasitoid strains used in the experiments, Sébastien Zito and Benjamin Bois for providing the climatic data. We acknowledge the valuable contribution of Alexandre Bauer, Aude Balourdet, Hugo Baali, Léa Bariod and Martin Pêcheur during the experiments. We would also like to thank the handling editor George Heimpel and the two anonymous reviewers for their insightful comments and helpful suggestions on the manuscript. This work was supported by the Conseil Régional de Bourgogne Franche-Comté through the Plan d’Actions Régional pour l’Innovation (PARI) and two other funding sources (FABER LOUAPRE-AGREE-BGS and VALEACLIM-BOIS), and the European Union through the PO FEDER-FSE Bourgogne 2014/2020 programs.
Author information
Authors and Affiliations
Contributions
All authors conceived and designed the experiments. CI and CM: performed the experiments; CI, CM and PL: analysed the data; CI, PL and JM: led the writing of the manuscript. All authors critically revised the intellectual content of the draft and gave their approbation for the final version to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Additional information
Communicated by George Heimpel.
Rights and permissions
About this article
Cite this article
Iltis, C., Moreau, J., Manière, C. et al. Where you come from matters: temperature influences host–parasitoid interaction through parental effects. Oecologia 192, 853–863 (2020). https://doi.org/10.1007/s00442-020-04613-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04613-z