Abstract
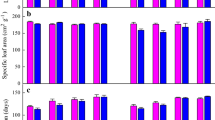
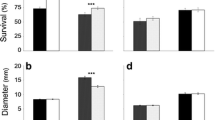
Invasive populations often shift phenotypically during introduction. Moreover, they are postulated to show an increased phenotypic plasticity compared with their native counterparts, which could be advantageous. However, less is known about trait selection across populations along the invasion gradient in response to environmental factors, such as increasing drought caused by climate change. In this study, we investigated the impacts of drought on growth, regrowth, and various leaf traits in plants of different origin. Therefore, seeds of 18 populations of the perennial Tanacetum vulgare were collected along the invasion gradient (North America, invasive; West Europe, archaeophyte; East Europe, native) and grown in competition with the grass Poa pratensis under control or dry conditions in a common garden. Above-ground biomass was cut once and the regrowth was measured as an indicator for tolerance over a second growth period. Initially, drought had little effects on growth of T. vulgare, but after cutting, plants grew more vigorously. Against expectations, phenotypic plasticity was not higher in invasive populations, but even reduced in one trait, which may be attributable to ecological constraints imposed by multiple stress conditions. Trait responses reflected the range expansion and invasion gradient and were influenced by the latitudinal origin of populations. Populations of invaded ranges may be subject to faster and more extensive genetic mixing or had less time to undergo and reflect selective processes.



Similar content being viewed by others
References
Andersson S (1991) Geographical variation and genetic analysis of leaf shape in Crepis tectorum (Asteraceae). Plant Syst Evol 178:247–258
Blossey B, Nötzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889
Bossdorf O, Auge H, Lafuma L, Rogers WE, Siemann E, Prati D (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11
CABI (2015) Tanacetum vulgare. In: Invasive Species Compendium. CAB International, Wallingford. http://www.cabi.org. Accessed 30 Nov 2015
Campitelli BE, Stinchcombe JR (2013) Testing potential selective agents acting on leaf shape in Ipomoea hederacea: predictions based on an adaptive leaf shape cline. Ecol Evol 3:2409–2423
Catford JA, Jansson R, Nilsson C (2009) Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Div Distrib 15:22–40
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought; from genes to the whole plant. Funct Plant Biol 30:239–264
Christensen S, Heimes C, Agerbirk N, Kuzina V, Olsen C, Hauser T (2014) Different geographical distributions of two chemotypes of Barbarea vulgaris that differ in resistance to insects and a pathogen. J Chem Ecol 40:491–501
Clements DR, Ditommaso A (2011) Climate change and weed adaptation: can evolution of invasive plants lead to greater range expansion than forecasted? Weed Res 51:227–240
Colautti RI, Maron JL, Barrett SCH (2009) Common garden comparisons of native and introduced plant populations: latitudinal clines can obscure evolutionary inferences. Evol Appl 2:187–199
Colomer-Ventura F, Martínez-Vilalta J, Zuccarini P, Escolà A, Armengot L, Castells E (2015) Contemporary evolution of an invasive plant is associated with climate but not with herbivory. Funct Ecol 29:1475–1485
Comas LH, Becker SR, Cruz VMV, Byrne PF, Dierig DA (2013) Root traits contributing to plant productivity under drought. Front Plant Sci 4:442
Cornelius R, Haug D (1991) Zur Plastizität des reproduktiven Aufwandes bei Conyza canadensis und Tanacetum vulgare. In: Schmid B, Stöcklin J (eds) Populationsbiologie der Pflanzen. Birkhäuser, Basel, Switzerland, pp 87–95
Ebeling SK, Stöcklin J, Hensen I, Auge H (2011) Multiple common garden experiments suggest lack of local adaptation in an invasive ornamental plant. J Plant Ecol 4:209–220
Erfmeier A, Tsaliki M, Roß CA, Bruelheide H (2011) Genetic and phenotypic differentiation between invasive and native Rhododendron (Ericaceae) taxa and the role of hybridization. Ecol Evol 1:392–407
Fornoni J (2011) Ecological and evolutionary implications of plant tolerance to herbivory. Funct Ecol 25:399–407
Gallardo B (2014) Europe’s top 10 invasive species: relative importance of climatic, habitat and socio-economic factors. Ethol Ecol Evol 26:130–151
Gard B, Bretagnolle F, Dessaint F, Laitung B (2013) Invasive and native populations of common ragweed exhibit strong tolerance to foliar damage. Basic Appl Ecol 14:28–35
Geng YP, van Klinken RD, Sosa A, Li B, Chen JK, Xu CY (2016) The relative importance of genetic diversity and phenotypic plasticity in determining invasion success of a clonal weed in the USA and China. Front Plant Sci. doi:10.3389/fpls.2016.00213
Gioria M, Osborne BA (2014) Resource competition in plant invasions: emerging patterns and research needs. Front Plant Sci 5:501
Godoy O, Valladares F, Castro-Díez P (2011) Multispecies comparison reveals that invasive and native plants differ in their traits but not in their plasticity. Funct Ecol 25:1248–1259
Grotkopp E, Rejmanek M (2007) High seedling relative growth rate and specific leaf area are traits of invasive species: phylogenetically independent contrasts of woody angiosperms. Am J Bot 94:526–532
Hinz HL, Schwarzlaender M (2004) Comparing invasive plants from their native and exotic range: what can we learn for biological control. Weed Technol 18:1533–1541
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular 347:1–32
Hogenbirk JC, Wein RW (1992) Temperature effects on seedling emergence from boreal wetland soils: implications for climate change. Aquat Bot 42:361–373
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inferences in general parametric models. Biometric J 50:346–363
IPCC (2012) Managing the risks of extreme events and disasters to advance climate change adaptation. A special report of working groups I and II of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jacobs J (2008) Ecology and management of Common Tansy (Tanacetum vulgare L.). In: Invasive Species. Technical note No. MT-18. Bozeman, Montana: United States Department of Agriculture
Kaufman SR, Smouse PE (2001) Comparing indigenous and introduced populations of Melaleuca quinquenervia (Cav.) Blake: response of seedlings to water and pH levels. Oecologia 127:487–494
Keskitalo M, Lindén A, Valkonen JPT (1998) Genetic and morphological diversity of Finnish tansy (Tanacetum vulgare L., Asteraceae). Theor Appl Genet 96:1141–1150
Kleine S, Müller C (2014) Drought stress and leaf herbivory affect root terpenoid concentrations and growth of Tanacetum vulgare. J Chem Ecol 40:1115–1125
Kühn I, Klotz S (2002) Systematik, Taxonomie und Nomenklatur. In: Klotz S, Kühn I, Durka W (eds) BIOLFLOR—Eine Datenbank mit biologisch-ökologischen Merkmalen zur Flora von Deutschland. Schriftenreihe für Vegetationskunde. Bonn, Germany
Kumschick S, Hufbauer RA, Alba C, Blumenthal DM (2013) Evolution of fast-growing and more resistant phenotypes in introduced common mullein (Verbascum thapsus). J Ecol 101:378–387
Leimu R, Koricheva J (2006) A meta-analysis of tradeoffs between plant tolerance and resistance to herbivores: combining the evidence from ecological and agricultural studies. Oikos 112:1–9
Lemoine NP, Burkepile DE, Parker JD (2016) Quantifying differences between native and introduced species. Trends Ecol Evol 31:372–381
Lin T, Klinkhamer PGL, Vrieling K (2015) Parallel evolution in an invasive plant: effect of herbivores on competitive ability and regrowth of Jacobaea vulgaris. Ecol Lett 18:668–676
Liu B, Liu ZM, Wang LX, Wang ZM (2014) Responses of rhizomatous grass Phragmites communis to wind erosion: effects on biomass allocation. Plant Soil 380:389–398
Machado RAR, Ferrieri AP, Robert CAM, Glauser G, Kallenbach M, Baldwin IT, Erb M (2013) Leaf-herbivore attack reduces carbon reserves and regrowth from the roots via jasmonate and auxin signaling. New Phytol 200:1234–1246
Mack R (1991) The commercial seed trade: an early disperser of weeds in the United States. Econ Bot 45:257–273
Mack RN (2003) Plant naturalizations and invasions in the Eastern United States: 1634–1860. Ann Miss Bot Garden 90:77–90
Mack RN, Erneberg M (2002) The United States naturalized flora: largely the product of deliberate introductions. Ann Mo Bot Gard 89:176–189
Martinkova J, Klimes L, Klimesova J (2011) Multiple regenerative strategies of short-lived species: an effect on geographical distribution, preference of human-made habitats and invasive status. Folia Geobot 46:181–189
Mony C, Mercier E, Bonis A, Bouzille JB (2010) Reproductive strategies may explain plant tolerance to inundation: a mesocosm experiment using six marsh species. Aquat Bot 92:99–104
Munné-Bosch S (2014) Perennial roots to immortality. Plant Physiol 166:720–725
Münzbergová Z, Křivánek M, Bucharová A, Juklíčková V, Herben T (2005) Ramet performance in two tussock plants: do the tussock-level parameters matter? Flora 200:275–284
Nicotra AB, Leigh A, Boyce CK, Jones CS, Niklas KJ, Royer DL, Tsukaya H (2011) The evolution and functional significance of leaf shape in the angiosperms. Funct Plant Biol 38:535–552
Orians CM, Ward D (2010) Evolution of plant defenses in nonindigenous environments. Annu Rev Entomol 55:439–459
Pankoke H, Müller C (2013) Impact of defoliation on the regrowth capacity and the shoot metabolite profile of Plantago lanceolata L. Plant Phys Biochem 71:325–333
Picotte J, Rhode J, Cruzan M (2009) Leaf morphological responses to variation in water availability for plants in the Piriqueta caroliniana complex. Plant Ecol 200:267–275
Pinheiro J, Bates D, DebRoy S, Sarkar D (2014) nlme: linear and nonlinear mixed effects models. R Package Version 3:1–117
R Development Core Team (2014) R: a language and environment for statistical computing, Vol. 3.0.3. R Foundation for Statistical Computing, Vienna
Rebele F (2000) Competition and coexistence of rhizomatous perennial plants along a nutrient gradient. Plant Ecol 147:77–94
Richards CL, Bossdorf O, Muth NZ, Gurevitch J, Pigliucci M (2006) Jack of all trades, master of some? On the role of phenotypic plasticity in plant invasions. Ecol Lett 9:981–993
Richardson DM, Pyšek P (2012) Naturalization of introduced plants: ecological drivers of biogeographical patterns. New Phytol 196:383–396
Samis KE, Murren CJ, Bossdorf O, Donohue K, Fenster CB, Malmberg RL, Purugganan MD, Stinchcombe JR (2012) Longitudinal trends in climate drive flowering time clines in North American Arabidopsis thaliana. Ecol Evol 2:1162–1180
Schmitz G (1996) Phytophagenkomplexe von Artemisia vulgaris L. und Tanacetum vulgare L. (Asteraceae) und deren Beeinflussung durch zunehmende Urbanität der Standorte, PhD Thesis. Rheinischen Friedrich-Wilhelms-Universität, Bonn
Schwachtje J, Baldwin IT (2008) Why does herbivore attack reconfigure primary metabolism? Plant Phys 146:845–851
Tiffin P (2002) Competition and time of damage affect the pattern of selection acting on plant defense against herbivores. Ecology 83:1981–1990
Tsukaya H (2002) The leaf index: heteroblasty, natural variation, and the genetic control of polar processes of leaf expansion. Plant Cell Physiol 43:372–378
Valladares F, Sanchez-Gómez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94:1103–1116
Valladares F, Gianoli E, Gómez JM (2007) Ecological limits to plant phenotypic plasticity. New Phytol 176:749–763
van der Putten WH et al (2000) Plant species diversity as a driver of early succession in abandoned fields: a multi-site approach. Oecologia 124:91–99
van Kleunen M, Weber E, Fischer M (2010) A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol Lett 13:235–245
Wolf V, Berger U, Gassmann A, Müller C (2011) High chemical diversity of a plant species is accompanied by increased chemical defence in invasive populations. Biol Invas 13:2091–2102
Zwicke M, Picon-Cochard C, Morvan-Bertrand A, Prud’homme MP, Volaire F (2015) What functional strategies drive drought survival and recovery of perennial species from upland grassland? Ann Bot 116:1001–1015
Acknowledgments
The authors thank Simon Waldherr, Felix Lansing, and Caroline Pons for support in sowing, watering, and harvest. Detlef Balten and Gebhard Sewing are thanked for support in plant transport to the common garden. Ruth Jacobs as well as two anonymous reviewers are thanked for valuable comments on the manuscript. The kind suppliers of seeds from various T. vulgare populations are listed in Wolf et al. (2011) and are gratefully acknowledged.
Author contribution statement
The conception and design of the study were performed by SK and CM. SK and LW carried out the common garden experiment. SK performed the statistical analysis of the paper and SK and CM wrote the manuscript. The final version was approved by all authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Hermann Heilmeier.
This study advances our understanding of plant responses to current major challenges induced by anthropogenic disturbance such as species invasions and climate change. It demonstrates that plants of an invasive perennial grown in competition under different drought conditions grew even more vigorously after cutting, highlighting the pronounced tolerance of this species.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kleine, S., Weissinger, L. & Müller, C. Impact of drought on plant populations of native and invasive origins. Oecologia 183, 9–20 (2017). https://doi.org/10.1007/s00442-016-3706-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3706-2