Abstract
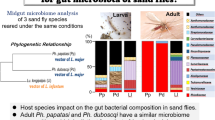
Leishmaniasis is a zoonotic vector-borne disease with worldwide distribution. All current approaches in leishmaniasis control or development of vaccines/cures showed only limited success. Recently, paratransgenesis has been marked as a promising strategy for leishmaniasis control. Thus, the investigations of the gut microbial content of sand flies have gained popularity. Gut microbial composition of the laboratory colony of Phlebotomus papatasi was investigated via microbial culturomics approach which refers to the combination of multiple culture conditions and different selective and/or enriched culture mediums, followed by 16S rDNA sequencing. Investigations were conducted on three offspring generations, with six samplings of immature stages (four larval samplings, one pre-pupa, one pupa) and samplings of adults before and after blood feeding. The aim was to determine if microbiome changes during the sand fly development and to identify bacteria with transstadial potential. The presence of 8 bacterial taxa (Bacillus sp., Terribacillus sp., Staphylococcus sp., Alcaligenes sp., Microbacterium sp., Leucobacter sp., Ochrobactrum sp. and Enterobacter sp.), 2 fungi (Fusarium sp. and Acremonium sp.) and 1 yeast (Candida sp.) were recorded. Gram-positive bacteria were more diverse, but gram-negative bacteria were more abundant. All taxa were recorded among immature stage samples, while only one bacterium was detected in adults. Microbial diversity among larval samples was stable, with a steady decrease in pre-pupa and pupa, resulting in the survival of only Ochrobactrum sp. in adults. Abundance of microbes was higher when larvae were actively feeding, with a gradual decrease after larvae stopped feeding and commenced pupation. Ochrobactrum sp. is the bacteria with transstadial potential, worthy of future in-depth analysis for the application in paratransgenic approach for the control of Leishmania sp.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abbasi I, Lopo T, de Queiroz A, Kirstein OD et al (2018) Plant-feeding phlebotomine sand flies, vectors of leishmaniasis, prefer Cannabis sativa. Proc Natl Acad Sci U S A 115:11790–11795. https://doi.org/10.1073/pnas.1810435115
Abdellahi L, Iraji F, Mahmoudabadi A, Hejazi SH (2022) Vaccination in leishmaniasis: a review article. Iran Biomed J 26:1–35. https://doi.org/10.52547/ibj.26.1.35
Apisarnthanarak A, Kiratisin P, Mundy LM (2005) Evaluation of Ochrobactrum intermedium bacteremia in a patient with bladder cancer. Diagn Microbiol Infect Dis 53:153–155. https://doi.org/10.1016/j.diagmicrobio.2005.05.014
Arenas R, Torres-Guerrero E, Quintanilla-Cedillo MR, Ruiz-Esmenjaud J (2017) Leishmaniasis: a review. F1000Research 6:1–15. https://doi.org/10.12688/f1000research.11120.1
Aujoulat F, Romano-Bertrand S, Masnou A et al (2014) Niches, population structure and genome reduction in Ochrobactrum intermedium: clues to technology-driven emergence of pathogens. PLoS One 9. https://doi.org/10.1371/journal.pone.0083376
Azambuja P, Feder D, Garcia ES (2004) Isolation of Serratia marcescens in the midgut of Rhodnius prolixus: impact on the establishment of the parasite Trypanosoma cruzi in the vector. Exp Parasitol 107:89–96. https://doi.org/10.1016/j.exppara.2004.04.007
Coico R (2005) Gram staining. Current protocols in microbiology. Appendix 3C. A.3C.1-A.3C.2. https://doi.org/10.1002/9780471729259.mca03cs00
Diba K, Kordbacheh P, Mirhendi SH et al (2007) Identification of Aspergillus species using morphological characteristics. Pakistan J Med Sci 23:867–872
Karimian F, Koosha M, Choubdar N, OshaghiI MA (2022) Comparative analysis of the gut microbiota of sand fly vectors of zoonotic visceral leishmaniasis (ZVL) in Iran; host-environment interplay shapes diversity. PLoS Neglected Trop Dis Neglected Trop Dis 16:e0010609
Frank JA, Reich CI, Sharma S et al (2008) Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl Environ Microbiol 74:2461–2470. https://doi.org/10.1128/AEM.02272-07
Gonzales-Ceron L, Santillan F, Rodriguez MH et al (2003) Bacteria in midguts of field-collected Anopheles albimanus block Plasmodium vivax sporogonic development. J Med Entomol 40:371–374. https://doi.org/10.1603/0022-2585-40.3.371
Guernaoui S, Garcia D, Gazanion E et al (2011) Bacterial flora as indicated by PCR-temperature gradient gel electrophoresis (TGGE) of 16S rDNA gene fragments from isolated guts of phlebotomine sand flies (Diptera: Psychodidae). J Vector Ecol 36. https://doi.org/10.1111/j.1948-7134.2011.00124.x
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98 citeulike-article-id:691774
Heerman M, Weng JL, Hurwitz I et al (2015) Bacterial infection and immune responses in lutzomyia longipalpis sand fly larvae midgut. PLoS Negl Trop Dis 9. https://doi.org/10.1371/journal.pntd.0003923
Hurwitz I, Fieck A, Read A et al (2011a) Paratransgenic control of vector borne diseases. Int J Biol Sci 7:1334–1344. https://doi.org/10.7150/ijbs.7.1334
Hurwitz I, Hillesland H, Fieck A et al (2011b) The paratransgenic sand fly: a platform for control of Leishmania transmission. Parasites and Vectors 4:82. https://doi.org/10.1186/1756-3305-4-82
Ishii K, Adachi T, Hara T et al (2014) Identification of a Serratia marcescens virulence factor that promotes hemolymph bleeding in the silkworm, Bombyx mori. J Invertebr Pathol 117:61–67. https://doi.org/10.1016/j.jip.2014.02.001
Johnson JS, Spakowicz DJ, Hong BY et al (2019) Evaluation of 16S rRNA gene sequencing for species and strain-level microbiome analysis. Nat Commun 10. https://doi.org/10.1038/s41467-019-13036-1
Kakumanu ML, Marayati BF, Schal C et al (2021) Oviposition-site selection of phlebotomus papatasi (Diptera: Psychodidae) sand flies: attraction to bacterial isolates from an attractive rearing medium. J Med Entomol 58:518–527. https://doi.org/10.1093/jme/tjaa198
Kämpfer P, Citron DM, Goldstein EJC, Scholz HC (2007) Difficulty in the identification and differentiation of clinically relevant Ochrobactrum species [2]. J Med Microbiol 56:1571–1573. https://doi.org/10.1099/jmm.0.47350-0
Karakuş M, Karabey B, Orçun Kalkan Ş et al (2017) Midgut bacterial diversity of wild populations of Phlebotomus (P.) papatasi, the vector of zoonotic cutaneous leishmaniasis (ZCL) in Turkey. Sci Rep 7:14812. https://doi.org/10.1038/s41598-017-13948-2
Kaur M, Chadha P, Kaur S, Kaur A (2021) Aspergillus flavus induced oxidative stress and immunosuppressive activity in Spodoptera litura as well as safety for mammals. BMC Microbiol 21. https://doi.org/10.1186/s12866-021-02249-4
Kumar S, Bin LZ, Sanyour-Doyel N et al (2022) Efficient gene targeting in soybean using Ochrobactrum haywardense-mediated delivery of a marker-free donor template. Plant Physiol 189:585–594. https://doi.org/10.1093/plphys/kiac075
Lawyer P, Killick-Kendrick M, Rowland T et al (2017) Laboratory colonization and mass rearing of phlebotomine sand flies (Diptera, Psychodidae). Parasite 24:42. https://doi.org/10.1051/parasite/2017041
Louradour I, Monteiro CC, Inbar E et al (2017) The midgut microbiota plays an essential role in sand fly vector competence for Leishmania major. Cell Microbiol 19. https://doi.org/10.1111/cmi.12755
Maleki-Ravasan N, Oshaghi MA, Afshar D et al (2015) Aerobic bacterial flora of biotic and abiotic compartments of a hyperendemic zoonotic cutaneous leishmaniasis (ZCL) focus. Parasites and Vectors 8:63. https://doi.org/10.1186/s13071-014-0517-3
Malta J, Heerman M, Weng JL et al (2017) Midgut morphological changes and autophagy during metamorphosis in sand flies. Cell Tissue Res 368:513–529. https://doi.org/10.1007/s00441-017-2586-z
Marayati BF, Schal C, Ponnusamy L et al (2015) Attraction and oviposition preferences of Phlebotomus papatasi (Diptera: Psychodidae), vector of Old-World cutaneous leishmaniasis, to larval rearing media. Parasites and Vectors 8. https://doi.org/10.1186/s13071-015-1261-z
Möller LVM, Arends JP, Harmsen HJM et al (1999) Ochrobactrum intermedium infection after liver transplantation. J Clin Microbiol 37:241–244. https://doi.org/10.1128/jcm.37.1.241-244.1999
Monteiro CC, Villegas LEM, Campolina TB et al (2016) Bacterial diversity of the American sand fly Lutzomyia intermedia using high-throughput metagenomic sequencing. Parasites and Vectors 9. https://doi.org/10.1186/s13071-016-1767-z
Moraes CS, Lucena SA, Moreira BHS et al (2012) Relationship between digestive enzymes and food habit of Lutzomyia longipalpis (Diptera: Psychodidae) larvae: characterization of carbohydrases and digestion of microorganisms. J Insect Physiol 58:1136–1145. https://doi.org/10.1016/j.jinsphys.2012.05.015
Mukhopadhyay J, Braig HR, Rowton ED, Ghosh K (2012) Naturally occurring culturable aerobic gut flora of adult Phlebotomus papatasi, vector of Leishmania major in the old world. PLoS One 7:e35748. https://doi.org/10.1371/journal.pone.0035748
Nasrabadi M, Azarm A, Molaeezadeh M et al (2021) Monitoring and mapping of insecticide resistance in vectors of visceral leishmaniasis in the world. J Infect Dis Prev Med 9:1000251
Ogodo AC, Agwaranze DI, Daji M, Aso RE (2022) Microbial techniques and methods: basic techniques and microscopy. Anal Tech Biosci from Basics to Appl 201–220. https://doi.org/10.1016/B978-0-12-822654-4.00003-8
Papadopoulos C, Karas PA, Vasileiadis S et al (2020) Host species determines the composition of the prokaryotic microbiota in phlebotomus sandflies. Pathogens 9. https://doi.org/10.3390/pathogens9060428
Peterkova-Koci K, Robles-Murguia M, Ramalho-Ortigao M, Zurek L (2012) Significance of bacteria in oviposition and larval development of the sand fly Lutzomyia longipalpis. Parasites and Vectors 5. https://doi.org/10.1186/1756-3305-5-145
Public Health England (2014) Detection and enumeration of bacteria in swabs and other environmental samples. PHE Microbiology Services Food, Water & Environmental Microbiology Methods Working Group Year of report: 2014, pp 1–21
Ratcliffe NA, Furtado Pacheco JP, Dyson P et al (2022) Overview of paratransgenesis as a strategy to control pathogen transmission by insect vectors. Parasites and Vectors 15:112. https://doi.org/10.1186/s13071-021-05132-3
Ready PD (2013) Biology of phlebotomine sand flies as vectors of disease agents. Annu Rev Entomol 58:227–250
Romo Bechara N, Wasserberg G, Raymann K (2022) Microbial ecology of sand fly breeding sites: aging and larval conditioning alter the bacterial community composition of rearing substrates. Parasites and Vectors 15. https://doi.org/10.1186/s13071-022-05381-w
RStudio T (2020) RStudio: integrated development for R, vol 42. RStudio, Inc, Boston, MA, p 14
Ruiz-Postigo JA, Jain S, Madjou S et al (2022) Global leishmaniasis surveillance: 2021, assessing the impact of the COVID-19 pandemic. Wkly Epidemiol Rec 45:575–590
Sachivkina N, Podoprigora I, Bokov D (2021) Morphological characteristics of Candida albicans, Candida krusei, Candida guilliermondii, and Candida glabrata biofilms, and response to farnesol. Vet World 14(6):1608–1614. https://doi.org/10.14202/vetworld.2021
Sales KGDS, Costa PL, De Morais RCS et al (2015) Identification of phlebotomine sand fly blood meals by real-time PCR. Parasites and Vectors 8. https://doi.org/10.1186/s13071-015-0840-3
Sant’Anna MRV, Darby AC, Brazil RP et al (2012) Investigation of the bacterial communities associated with females of Lutzomyia sand fly species from South America. PLoS One 7. https://doi.org/10.1371/journal.pone.0042531
Sant’Anna MRV, Diaz-Albiter H, Aguiar-Martins K et al (2014) Colonisation resistance in the sand fly gut: Leishmania protects Lutzomyia longipalpis from bacterial infection. Parasites and Vectors 7:329. https://doi.org/10.1186/1756-3305-7-329
Seye F, Faye O, Ndiaye M et al (2009) Pathogenicity of the fungus, Aspergillus clavatus, isolated from the locust, Oedaleus senegalensis, against larvae of the mosquitoes Aedes aegypti, Anopheles gambiae and Culex quinquefasciatus. J Insect Sci 9. https://doi.org/10.1673/031.009.5301
Silva-Filha MHNL, Romão TP, Rezende TMT et al (2021) Bacterial toxins active against mosquitoes: mode of action and resistance. Toxins (Basel) 13(8):523. https://doi.org/10.3390/toxins13080523
Summerbell RC, Gueidan C, Schroers HJ et al (2011) Acremonium phylogenetic overview and revision of Gliomastix, Sarocladium, and Trichothecium. Stud Mycol. https://doi.org/10.3114/sim.2011.68.06
Summerell BA (2019) Resolving Fusarium: current status of the Genus. Annu Rev Phytopathol 25:57:323-339. https://doi.org/10.1146/annurev-phyto-082718-100204
Tabbabi A, Mizushima D, Yamamoto D, Kato H (2022) Sand flies and their microbiota. Parasitologia 2:71–87
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Vivero RJ, Castañeda-Monsalve VA, Romero LR et al (2021) Gut microbiota dynamics in natural populations of pintomyia evansi under experimental infection with leishmania infantum. Microorganisms 9. https://doi.org/10.3390/microorganisms9061214
Vivero RJ, Jaramillo NG, Cadavid-Restrepo G et al (2016) Structural differences in gut bacteria communities in developmental stages of natural populations of Lutzomyia evansi from Colombia’s Caribbean coast. Parasites and Vectors 9. https://doi.org/10.1186/s13071-016-1766-0
Vivero RJ, Villegas-Plazas M, Cadavid-Restrepo GE et al (2019) Wild specimens of sand fly phlebotomine Lutzomyia evansi, vector of leishmaniasis, show high abundance of Methylobacterium and natural carriage of Wolbachia and Cardinium types in the midgut microbiome. Sci Rep 9. https://doi.org/10.1038/s41598-019-53769-z
Volf P, Kiewegová A, Nemec A (2002) Bacterial colonisation in the gut of Phlebotomus duboscqi (Diptera: Psychodidae): transtadial passage and the role of female diet. Folia Parasitol (Praha). https://doi.org/10.14411/fp.2002.014
Volf P, Volfova V (2011) Establishment and maintenance of sand fly colonies. J Vector Ecol 36:S1–S9. https://doi.org/10.1111/j.1948-7134.2011.00106.x
World Health Organisation (2023) Leishmaniasis fact sheet. https://www.who.int/news-room/factsheets/detail/leishmaniasis. Accessed 12 Dec 2023
Wijerathna T, Gunathunga S, Gunathilaka N (2020) Recent developments and future directions in the paratransgenesis based control of Leishmania transmission. Biol Control 145:104260. https://doi.org/10.1016/j.biocontrol.2020.104260
Acknowledgements
The authors would like to thank Sefika Özcan and Elif Hatice Ayten for their immense help and support in sand fly colony maintenance.
Funding
Paratransgenesis as a weapon against leishmaniasis (PartoLeish). European Commission Horizon 2020 Marie Skłodowska-Curie Actions and The Scientific and Technological Research Council of Türkiye (TÜBİTAK) Cofund program (Co-Funded Brain Circulation Scheme2 - CoCirculation2; TÜBİTAK 2236; Project No: 121C042)
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, funding acquisition and writing of original draft were performed by S.V.; formal analysis and investigation were done by S.V., B.E.S., A.D.U. and C.K.; review and edition of the manuscript: S.V., B.E.S., A.D.U., A.Y., C.K., B.A.; S.V. and B.A. were supervising the work. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Sutherland Maciver
Disclaimer
The entire responsibility of the publication/paper belongs to the owner of the publication/paper. The financial support received from TÜBİTAK does not mean that the content of the publication is approved in a scientific sense by TÜBİTAK.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vaselek, S., Sarac, B.E., Uzunkaya, A.D. et al. Identification of Ochrobactrum as a bacteria with transstadial transmission and potential for application in paratransgenic control of leishmaniasis. Parasitol Res 123, 82 (2024). https://doi.org/10.1007/s00436-023-08087-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-023-08087-9