Abstract
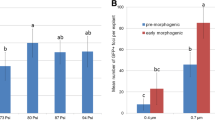
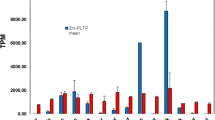
The presence of selectable marker genes and vector backbone sequences has affected the safe assessment of transgenic plants. In this study, the ovary-drip method for directly generating vector- and selectable marker-free transgenic plants was described, by which maize was transformed with a linear GFP cassette (Ubi-GFP-nos). The key features of this method center on the complete removal of the styles and the subsequent application of a DNA solution directly to the ovaries. The movement of the exogenous DNA was monitored using fluorescein isothiocyanate-labeled DNA, which showed that the time taken by the exogenous DNA to enter the ovaries was shortened compared to that of the pollen-tube pathway. This led to an improved transformation frequency of 3.38% compared to 0.86% for the pollen-tube pathway as determined by PCR analysis. The use of 0.05% surfactant Silwet L-77 + 5% sucrose as a transformation solution further increased the transformation frequency to 6.47%. Southern blot analysis showed that the transgenic plants had low transgene copy number and simple integration pattern. Green fluorescence was observed in roots and immature embryos of transgenic plants by fluorescence microscopy. Progeny analysis showed that GFP insertions were inherited in T1 generation. The ovary-drip method would become a favorable choice for directly generating vector- and marker-free transgenic maize expressing functional genes of agronomic interest.







Similar content being viewed by others
Abbreviations
- FITC:
-
Fluorescein isothiocyanate
- GFP:
-
Green fluorescent protein
- Ubi promoter:
-
Ubiquitin promoter
- Nos terminator:
-
Nopaline synthase terminator
References
Agrawal PK, Kohli A, Twyman RM, Christou P (2005) Transformation of plants with multiple cassettes generates simple transgene integration patterns and high expression levels. Mol Breed 16:247–260
Bent AF (2000) Arabidopsis in planta transformation. Uses, mechanisms, and prospects for transformation of other species. Plant Physiol 124:1540–1547
Christou P (1996) Transformation technology. Trends Plant Sci 1:423–431
Clough S, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Cluster PD, O’Dell M, Metzlaff RB (1994) Details of T-DNA structural organization from a transgenic Petunia population exhibiting co-suppression. Plant Mol Biol 32:1197–1203
Daniell H (1999) GM crops: public perception and scientific solutions. Trends Plant Sci 4:467–469
Demeke T, Hucl P, Båga M, Caswell K, Leung N, Chibbar RN (1999) Transgene inheritance and silencing in hexaploid wheat. Theor Appl Genet 99:947–953
De Vetten N, Wolters AM, Raemakers K, Van Der Meer I, Ter Stege R, Heeres E, Heeres P, Visser R (2003) A transformation method for obtaining marker-free plants of a cross-pollinating and vegetatively propagated crop. Nat Biotechnol 21:439–442
Filipecki M, Malepszy S (2006) Unintended consequences of pant transformation: a molecular insight. J Appl Genet 4:277–286
Fu XD, Duc LT, Fontana S, Bong BB, Tinjuangjun P, Sudhakar D, Twyman RM, Christou P, Kohli A (2000) Linear transgene constructs lacking vector backbone sequences generate low-copy-number transgenic plants with simple integration patterns. Transgenic Res 9:11–19
Gao XR, Wang GK, Su Q, Wang Y, An LJ (2007) Phytase expression in transgenic soybeans: stable transformation with a vector-less construct. Biotechnol Lett 29:1781–1787
Horvath H, Jensen LG, Wong OT, Kohl E, Ullrich SE, Cochren J, Kannangars CG, Von Wettstein D (2001) Stability of transgene expression, field performance and recombination breeding of transformed barley lines. Theor Appl Genet 102:1–11
Hou WS, Guo SD, Lu M (2003) Development of transgenic wheat with Galanthus nivalis agglutinin gene (sgna) via the pollen tube pathway. Chin Bull Bot 20:198–204
Hu CY, Wang LJ (1999) In planta soybean transformation technologies developed in China: procedure, confirmation and field performance. In Vitro Cell Dev Biol Plant 35:417–420
Kohli A, Griffiths S, Palacios N, Twyman RM, Vain P, Laurie DA, Christou P (1999) Molecular characterization of transforming plasmid rearrangements in transgenic rice reveals a recombination hotspot in the CaMV 35S promoter and confirms the predominance of microhomology-mediated recombination. Plant J 17:591–601
Kondrak M, Ingrid M, Meer VD, Banfalvi Z (2006) Generation of marker- and backbone-free transgenic potatoes by site-specific recombination and a bi-functional marker gene in a non-regular one-border Agrobacterium transformation vector. Transgenic Res 15:729–737
Kononov ME, Bassuner B, Gelvin SB (1997) Integration of T-DNA binary vector “backbone” sequence into the tobacco genome: evidence for multiple complex patterns of integration. Plant J 11:945–957
Loc NT, Tinjuangjun P, Gatehouse AMR, Christou P, Gatehouse JA (2002) Linear transgene constructs lacking vector backbone sequences generate transgenic rice plants which accumulate higher levels of proteins conferring insect resistance. Mol Breed 9:231–244
Luo ZX, Wu RA (1988) Simple method for the transformation of rice via the pollen-tube pathway. Plant Mol Biol Rep 6:165–174
Miki B, McHugh S (2004) Selectable marker genes in transgenic plants: applications, alternatives and biosafety. J Biotechnol 107:193–232
Poirier Y, Ventre G, Nawrath C (2000) High-frequency linkage of co-expressing T-DNA in transgenic Arabidopsis thaliana transformed by vacuum-infiltration of Agrobacterium tumefaciens. Theor Appl Genet 100:487–493
Puchta H (2000) Removing selectable marker genes: taking the shortcut. Trends Plant Sci 5:273–274
Ramanathan V, Veluthambi K (1996) Transfer of non-T-DNA portions of Agrobacterium tumefaciens Ti plamid pTIA6 from the left terminus of T-L-DNA. Plant Mol Biol 28:1149–1154
Rasco GS, Riley R, Cannell M, Barcelo P, Lazzeri PA (2001) Procedures allowing the transformation of range of European elite wheat (Triticum aestivum L.) varieties via particle bombardment. J Exp Bot 52:865–874
Reichandt M, Rogers S (1994) Preparation of plant DNA using CTAB. In: Ausubel FM, Bernt R, Struhl K (eds) Current protocols in molecular biology [M]. Sarah Greene, Brooklyn, NY, pp 233–237
Romano A, Raemakers K, Bernardi J, Visser R, Mooibroek H (2003) Transgene organization in potato after particle bombardment-mediated (co-)transformation using plasmids and gene cassettes. Transgenic Res 12:461–473
Sachs ES, Benedict JH, Stelly DM (1998) Expression and segregation of genes encoding Cry1A insecticidal proteins in cotton. Crop Sci 38:1–11
Shen JH, Li HR, Li YF, Yin H, Lian YQ (1987) Cytologic observations on the double fertilization in maize. Acta Bot Sin 29:480–485
Siemens J, Schieder O (1996) Transgenic plants: genetic transformation—recent developments and state of the art. Plant Tissue Cult Biotechnol 2:66–75
Tangmitcharoen S, Owens JN (1997) Floral biology, pollination, pistil receptivity, and pollen tube growth of teak (Tectona grandis Linn f.). Ann Bot 79:227–241
Tingay S, McElroy D, Kalla R, Fieg S, Wang M, Thomas S, Brettell R (1997) Agrobacterium tumefaciens-mediated barley transformation. Plant J 11:1369–1376
Ulian EC, Magill JM, Smith RH (1994) Expression and inheritance pattern of two foreign genes in petunia. Theor Appl Genet 88:433–440
Vain P, James VA, Worland B, Snape JW (2002) Transgene behavior across two generation in a large random population of transgenic rice plants produced by particle bombardment. Theor Appl Genet 105:878–889
Volodymyr VR, Dang TV, Evelyn K (2005) Multiple gene co-integration in Arabidopsis thaliana predominantly occurs in the same genetic locus after simultaneous in planta transformation with distinct Agrobacterium tumefaciens strains. Plant Sci 168:1515–1523
Wu W, Su Q, Xia XY, Wang Y, Luan YS, An LJ (2008) The Suaeda liaotungensis Kitag betaine aldehyde dehydrogenase gene improves salt tolerance of transgenic maize mediated with minimum linear length of DNA fragment. Euphytica 159:17–25
Yin ZM, Plader W, Malepszy S (2004) Transgene inheritance in plants. J Appl Genet 45:127–144
Zechendorf B (1994) What the public thinks about biotechnology. Biotechnology 12:870–875
Zeng JZ, Wang DJ, Wu YQ, Zhang J, Zhou WJ, Zhu XP, Xu NJ (1994) Transgenic wheat plants obtained with pollen tube pathway method. Sci Chin 37:319–325
Zhang YS, Yin XY, Yang AF, Li GS, Zhang JR (2005) Stability of inheritance of transgenes in maize (Zea mays L.) line produced using different transformation methods. Euphytica 144:11–22
Zhao L, Wang CL, Zong SY, Huang JL, Gong ZZ (2003) Herbicide resistant transgenic rice (Oryza sativa L.) transformed by pollen-tube pathway method and its inheritance. Chin Biotechnol 23:92–97
Zhou GY, Weng J, Zeng Y, Huang J, Qian S, Liu G (1983) Introduction of exogenous DNA into cotton embryos. Methods Enzymol 101:433–481
Zhou GY, Weng J, Gong Z, Zeng Y, Yang W, Shen W (1988) Molecular breeding of agriculture: a technique for introducing exogenous DNA into plants after self-pollination. Sci Agric Sin 21:1–6
Zhou J, Mi XJ, Cui JZ (2006) Non-Mendelian inheritance of transgene in plant. Mol Plant Breed 4:614–620
Zuo KJ, Zhang XL, Nie YC (2002) Sequence analysis of Bt insertion flanking fragments in transgenic Bt cotton. Acta Genet Sin 29:735–740
Acknowledgments
This work was supported by grants from the Key Projects of Science and Technology Council of Liaoning Province (no. 2006208001). The authors specially thank Dr. Alan K. Chang (Dalian University of Technology) for critically reviewing this manuscript and helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, A., Su, Q. & An, L. Ovary-drip transformation: a simple method for directly generating vector- and marker-free transgenic maize (Zea mays L.) with a linear GFP cassette transformation. Planta 229, 793–801 (2009). https://doi.org/10.1007/s00425-008-0871-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-008-0871-5