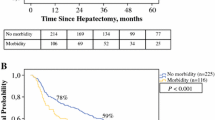
Abstract
Purpose
The aim of this study was to identify predictive risk factors associated with 90-day mortality after hepatic resection (HR) in hepatocellular carcinoma (HCC).
Methods
All patients undergoing elective resection for HCC from a single- institutional and prospectively maintained database were included. Multivariate regression analysis was conducted to identify pre- and intraoperative as well as histopathological predictive factors of 90-day mortality after elective HR.
Results
Between August 2004 and October 2021, 196 patients were enrolled (148 male /48 female). The median age of the study cohort was 68.5 years (range19-84 years). The rate of major hepatectomy (≥ 3 segments) was 43.88%. Multivariate analysis revealed patient age ≥ 70 years [HR 2.798; (95% CI 1.263–6.198); p = 0.011], preoperative chronic renal insufficiency [HR 3.673; (95% CI 1.598–8.443); p = 0.002], Child–Pugh Score [HR 2.240; (95% CI 1.188–4.224); p = 0.013], V-Stage [HR 2.420; (95% CI 1.187–4.936); p = 0.015], and resected segments ≥ 3 [HR 4.700; (95% 1.926–11.467); p = 0.001] as the major significant determinants of the 90-day mortality.
Conclusion
Advanced patient age, pre-existing chronic renal insufficiency, Child–Pugh Score, extended hepatic resection, and vascular tumor involvement were identified as significant predictive factors of 90-day mortality. Proper patient selection and adjustment of treatment strategies could potentially reduce short-term mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is the most common primary hepatic malignancy and a leading cause of cancer-related death worldwide [1, 2]. Hepatic resection (HR) and liver transplantation (LT) constitute the cornerstones of curative intended treatment even in advanced tumor stages [3, 4]. However, surgery-related mortality in HCC has been reported to range from 2% to 32%, depending on pre-existing cirrhosis and the extent of resection [5,6,7]. Morbidity rates after surgery range between 10% to almost 50% [8]. Advances in surgical techniques, significant improvements in perioperative care and proper patient selection in highly specialized and high-volume hepato-pancreato-biliary (HPB) centers have resulted in a remarkable reduction of perioperative mortality and morbidity [9, 10]. In order to avoid underestimation of postoperative mortality, the 90-day mortality rate has been proposed as a reliable measure of operative quality in hepatic surgery for malignancy since delayed mortality may not be registered within the first 30 postoperative days [11, 12]. Various clinical risk models and predictive scores of postoperative mortality in hepatic surgery have been described [13,14,15,16,17,18]. However their validity and accuracy in HCC must be questioned as these scores are constituted of a portfolio of arbitrarily chosen pre,-intra- and postoperative data/variables which were derived from heterogeneous study populations and tumor entities undergoing various types of parenchyma resection. The (model for end stage liver disease) MELD Score was therefore introduced to properly discriminate patients at risk of postoperative mortality in HCC patients with cirrhosis [19], whereas its predictive capacity diminished in non-cirrhotic patients [20].
Interestingly, only a few studies specifically analyzed the 90-day mortality outcome and its contributing parameters in elective HCC resection [21,22,23,24] resulting in a paucity of reliable and reproducible predictive mortality factors. Of note, three of these studies originated from Asian centers [21, 22, 24], one study only included preoperative clinical variables [24], while one study exclusively analyzed patients with major hepatectomy [22].
Hence, our primary goal was therefore to identify factors associated with 90-day mortality in a representative cohort of patients undergoing curative intent surgery for HCC. Identifying these factors will facilitate appropriate risk stratification and patient selection in order to minimize surgical morbidity and mortality by modifying adjustable parameters and considering alternative treatment options.
Material and methods
All patients with HCC undergoing curative hepatic resection at the Department of General, Visceral, Thorax and Pediatric Surgery, University Hospital Duesseldorf, Germany between August 2004 and October 2021 were considered eligible from a prospectively maintained database. The exclusion criteria included patient age < 18 years, missing information regarding 90-day mortality, surgical exploration without parenchymal resection, and mixed typed tumors on final histology examination. Prior to the study initiation, the approval of the local ethics committee at the Heinrich-Heine-University Duesseldorf, Germany was granted (study-no.: 2021–1800- KFogU). All reported procedures and steps were in accordance with the principles of the latest version of the Declaration of Helsinki. All parts of this article were strictly subjected to the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) checklist for the reporting of observational Studies [25]. Data acquisition was organized and performed in four sections:
-
1)
Preoperative: demographic and patient- related characteristics [age, gender, body mass index (BMI), ASA score (American Society of Anesthesiologists)], comorbidities, underlying hepatic disease and damage, baseline laboratory findings (including liver function tests, total blood count, renal parameters, albumin, α-fetoprotein, and hepatitis serology), MELD Score, Child–Pugh classification, preoperative imaging studies with tumor load and location, volumetric liver calculation in case of insufficient future liver remnant volume (FLRV), and alternative therapy concepts.
-
2)
Intraoperative: type and extent of resection, biliary reconstruction, duration of surgery (min), number of transfused blood units, intraoperative complications.
-
3)
Postoperative: morbidity including bile leakage, intra-abdominal abscess formation, cholangitis, sepsis, wound infection, notification of liver failure according to the International Study Group of Liver Surgery (ISGLS) criteria [26], and 90-day mortality as the primary endpoint.
-
4)
Histology and tumor stage: total number of tumors, tumor diameter (mm), TNM classification based on Union internationale contre le cancer (UICC) 8th edition [27], tumor grading, distant metastasis, resection margin, perineural and lymphangio-invasion, nodal and vascular involvement.
The current available terminology of hepatectomy was applied to classify type and extent of resection [28]. Major hepatectomy was categorized as the resection of ≥ 3segments. Postoperative morbidity was defined and stratified based on the Clavien-Dindo classification [29]. The status of preoperative chronic renal insufficiency was determined according to the current nephrological guidelines with a glomerular filtration rate (GFR) < 60 ml/min per 1·73 m2 or markers of kidney damage of at least 3 months duration [30].
Each HCC patient was discussed in a multidisciplinary tumor board and the indication for surgical resection was confirmed by an expert panel of gastroenterologists, hepatobiliary surgeons, pathologists, radiotherapists and radiologists. Preoperative work-up included helical computed tomography (CT) scanning of the chest, abdomen, and pelvis. If necessary additional magnetic resonance imaging (MRI) scans of the liver were obtained for appropriate planning. Patients with extensive tumor burden and a prospective FLRV of < 30% were candidates for augmentation techniques using either portal venous embolization (PVE) or in situ split plus portal vein ligation (ISLT) at the discretion of the surgical team involved. Technical aspects and our institutional approach of in situ splitting of hepatic parenchyma has been discussed extensively [31, 32]. Moreover, we described the successful application of ISLT as a rescue procedure after insufficient growth with PVE [33]. Intraoperative hepatic ultrasound evaluation was routinely conducted to assess tumor extent and resectability, and to rule out undetected tumor nodules. Of note, the Pringle’s maneuver was not routinely applied for hepatic resection at our institution. Parenchyma dissection was done with the cavitron ultrasonic surgical aspirator (CUSA®; Valleylab, Boulder, Colorado, USA). Hepato-duodenal ligament lymphadenectomy was conducted for oncological and/or preparatory reasons and to visualize the hilar vascular and biliary anatomy in case of extended resections. After complex biliary reconstruction a decompressing T-Drain was inserted in some cases for optimal drainage and pressure reduction within the biliary tract. The retrieved specimen was subjected to precise histopathological examination. Tumor grading was performed according to Edmondson and Steiner [34]. Additionally, vascular and lymphangiovascular invasion, and the resection margins were determined by macroscopic and/or microscopic evaluation.
Statistical analysis and variable selection
Statistical analysis was performed using the SPSS 25.0 software program (Statistical Package for Social Sciences; SPSS Inc., Chicago, IL, USA). To assess normal distribution of continuous data, the Shapiro–Wilk test was applied. Subsequently, the t-test was used for normal distributed data, while the Mann–Whitney U test was carried out for data that did not exhibit a normal distribution. Continuous data were expressed as median and standard deviation (SD). Categorical variables were summarized as frequencies (%) and compared using the chi-square or Fisher’s exact test. The 90-day mortality was defined as death within the 90-days interval from the initial hepatic resection. Patients were divided into two groups according to the occurrence of 90-day mortality. To identify potential risk factors for 90-day mortality based on pre- and intraoperative as well as histopathological parameters, Kaplan–Meier curves were generated and evaluated using the log-rank test. In addition, hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated using a univariate Cox regression analysis. All relevant clinical and pathological variables with a p-value ≤ 0.1 were included into a multivariable Cox regression analysis. The forward stepwise selection was used to create a final model. Multiple imputation was used to replace missing values in our dataset. Variables with at least 20% missing values were excluded from the analysis. In all analyses, a p-value of < 0.05 indicated statistical significance.
Results
Patient characteristics and intraoperative data
A total of 196 (148 male/ 48 female) patients underwent curative HR for HCC at our department between 2004 and 2021. Preoperative patient characteristics, pathology reports and the intraoperative course are summarized in Table 1. The median age of the entire cohort was 68.5 years (range19-84 years). Ninety-three patients (47.45%) were seventy years or older. The majority of patients were classified as ASA III/IV (66.33%). Hepatitis B and C were evident in 44 (22.45%) and 58 patients (29.59%) respectively. Twenty- three patients (11.73%) had intermediate or advanced liver cirrhosis CHILD B and C while Child A cirrhosis was recorded in 150 (76.53%) patients. Thirty-nine patients (19.90%) had a history of chronic preoperative alcohol abuse. The most common comorbidities included cardio-pulmonary disease (56.63%), followed by diabetes mellitus type 2 (35.20%), and chronic renal insufficiency (14.80%). In 111 patients (56.63%) the MELD Score was at least as high as the median of 8. A single HCC lesion was noted in 116 patients (59.18%) whereas ≥ 2 lesions were recorded in 80 cases (40.82%). The rate of bilobular tumor burden was 29.59% (58 patients). Eighty-six patients (43.88%) underwent extended resections of ≥ 3 segments and complex biliary reconstructions were performed in 15 patients (7.65%). The median operative time in the entire cohort was 307 min (range 70–815 min) and a prolonged surgical procedure extending the median value was recorded in half of all patients (50%). Fifteen patients (7.65%) with a critical FLRV of less than 30%, required PVE or ISLT as hepatic augmentation techniques prior to extended resection. Rescue ISLT was necessary in 4 patients after unsuccessful PVE considering the insufficient volume gain. After histological examination, an advanced T-Stage (III/IV) was observed in 41 patients (20.92%). Thirty-two patients (16.33%) had high grade tumors (Grade III/IV). The rate of lymphangio-and vascular invasion was 5.10% and 17.35% respectively. Distant metastasis were observed in 5 patients (2.55%). In 35 patients (17.86%) R0-tumor clearance was achieved by a narrow resection margin (< 0.1 cm).
Postoperative course
The postoperative course is depicted in Table 2. The most prevalent postoperative morbidity was ISGLS grade B/C liver failure (32.65%). Wound infections were observed in 24 patients (12.24%). Twenty-one patients suffered from bile leakage (10.71%). In addition, intra-abdominal abscess formation was noted in 8.16% of the cases. Other infectious complications included sepsis (11.73%), and cholangitis (7.65%). The overall rate of severe complications (CD ≥ 3a) was 40.31%. Thirty patients died within the 90-day time interval from surgical intervention accounting for a 90-day mortality rate of 15.30% as the primary endpoint. Interestingly, when patients were stratified by study years, the 90-day mortality rate decreased from 19.69% in the time interval 2004–2015 to 7.8% in patients undergoing HR between 2016 and 2021.
Uni-and multivariate analyses of predictive factors of 90-days mortality
First, a univariate analysis was performed to identify pre- and intraoperative as well as histopathological parameters that are associated with 90-day mortality. All variables with a p-value ≤ 0.1 were then included into a multivariate regression analysis. Accordingly, the following parameters were included into the multivariate analysis: age ≥ 70 years, ASA score I/II vs. III/IV, AST ≥ 52.5 U/l, hemoglobin ≥ 13.3 g/dl, Child–Pugh Score, chronic renal insufficiency, uni- versus bilobular lesion(s), T-Stage I/II vs. III/IV, M-Stage, L-Stage, V-Stage, UICC Stage ISLT/PVE, segments ≥ 3 resection, biliary reconstruction, operative time ≥ 307 min, intraoperative transfusion (supplementary Table 1). Subsequently, the final multivariate analysis revealed age ≥ 70 years [HR 2.798; (95% CI 1.263–6.198); p = 0.011], chronic renal insufficiency [HR 3.673; (95% CI 1.598–8.443); p = 0.002], Child–Pugh Score [HR 2.240; (95% CI 1.188–4.224); p = 0.013], V-Stage [HR 2.420; (95% CI 1.187–4.936); p = 0.015], and segments ≥ 3 [HR 4.700; (95% 1.926–11.467); p = 0.001] as significant predictive factors associated with 90-day mortality (Table 3).
Discussion
The results of our single institutional study with 196 included patients demonstrate that the 90-day mortality rate after HR for HCC is significantly associated with advanced patient age (≥ 70 years), preoperative existent chronic renal insufficiency, CHILD–Pugh Score, vascular tumor involvement, and major hepatectomy with ≥ 3 resected segments. We deliberately omitted all postoperative factors from our analysis, as the main focus relied on pathological and (modifiable) pre- and intraoperative variables that allow for proper risk stratification and thus patient-tailored therapy.
The overall 90-day mortality rate in our cohort was 15.30%, which is consistent with a previous western report [23] but notably higher than Asian studies [35,36,37]. Results from a large German database query [38] demonstrated a hospital mortality rate of 9.3% in HCC patients following resection which might be a potential underestimation of the mortality data within 90 days, given our 30-day mortality rate of 6.63%. A recently published meta-analysis revealed a weighted 90-day mortality rate of 4.2% (range 3%–5.4%) among 8474 included patients with a significant level of heterogeneity [39].
Hepatic resection is still regarded as an effective and potentially curative method in HCC treatment. With the aging population and demographic changes, the percentage of elderly patients with pre-existing liver disease including HCC considered for hepatectomy is increasing [40]. However, the role of HR in the elderly population is still controversial based on conflicting results [41, 42]. Besides, a universal definition of “elderly” has not been described and studies used different cut-off values to stratify for patient age [43]. It has been previously shown that older patients over 70 years with liver cirrhosis undergoing hepatic resection are at increased risk of an unfavorable short-term outcome [44].These results were even confirmed in non-cirrhotic elderly patients after ≥ 2 segment resections [45]. Another large scaled study with 27.094 patients from Japan also depicted advanced patient age (≥ 70 years) as a significant risk factor of postoperative mortality in HCC [41].
The potential explanations rely on the underlying fragility and impaired physiological reserve capacities in this patient subgroup, especially in response to major surgical trauma [46], pre-existing liver deterioration related to liver cirrhosis and hepatitis [47], and the higher incidence of relevant comorbidities [41]. Of note, in our study almost half of the patients (47.45%) were aged 70 and above. The rate of major hepatic resection in the elderly group was 47.31%.
Another significant determinant of 90-day mortality was pre-existing renal insufficiency. Chronic kidney disease was reported in 29 patients (14.80%). The mortality rate in this patient cohort was 31.03%. Indeed, in the literature the impact of chronic renal insufficiency on short-and long term survival after HCC resection has been discussed with differing results [48, 49]. A recent meta-analysis [50] revealed that chronic kidney disease was associated with higher rates of postoperative complications and decreased overall survival. In another study, Shirata et al. [49] could demonstrate similar 90-day mortality rates in patients with chronic renal disease and Child–Pugh A cirrhosis in comparison to patients without renal impairment. In our cohort, the majority patients with renal disease had liver cirrhosis Child–Pugh A (79.31%). The 90-day mortality rate in this subgroup of Child–Pugh A patients with renal insufficiency was 21.73%, in contrast to Shirata et al. [49], who reported a 1.9% mortality rate following HR. Noteworthy, the rate of major hepatectomy in chronic renal disease patients was higher in our study (31.03% versus 27%) as compared to Shirata et al. [49] and a considerable proportion of our patients in this subgroup were classified ASA III/IV (75.86%). In the current study, the Child–Pugh classification was identified as a predictive factor of 90-day mortality. This is in line with a previously published work from Singapore analyzing 244 HCC patients [21]. The Child–Pugh score is a well-established clinical tool based on laboratory and clinical findings which helps to assess the severity of liver dysfunction and to predict postoperative outcome in hepatic surgery [51]. However, due to several limitations [52], other models such as the albumin–bilirubin (ALBI) score have demonstrated more accuracy in predicting overall survival in HCC resection [53]. In a large multicenter study, Beradi and colleagues [54] evaluated the 90-day mortality rate following 253 mostly minor hepatic resections in Child-B cirrhotic patients. The 90-day mortality rate of the entire cohort was 4.3% while a significant difference in 90-day mortality rate was observed depending on the extent of surgical resection (minor resection 3.3% versus major resection 10.3%; p = 0.04). In another European single center study [55] the in-hospital mortality rate of patients receiving HR for HCC stratified by Child–Pugh A, and B-C classification was 4.7% and 21.3% respectively (p = 0.0003).
The in-hospital mortality rate of the 23 included Child–Pugh B-C in our cohort was 13.04%, whereas the 90-day mortality rate increased to 39%. These numbers again highlight the importance of evaluating 90-day mortality instead of in-hospital and/or 30-day mortality to avoid underestimation and underreporting of the “true” operative related deaths [12, 56].
Of note, the rate of 11.22% Child–Pugh B patients in our study was higher than in the above mentioned studies of Beradi et al. (6.9%) [54] and Lei et al. (8.2%) [21]. The extent and type of resection has been proposed as a significant predictive factor and has subsequently been incorporated into risk scoring tools of in-hospital [57] and 90-day mortality [15, 23]. In contrast, a recent meta-analysis with 43 included studies could not demonstrate short-term survival benefits although long-term survival was significantly influenced by the resection strategy [58]. In our study the 90-day mortality rate of 86 patients undergoing major hepatectomy (≥ 3 segments) was 25.58% in comparison to just 7.27% in the group with minor resections (p < 0.0001). It is important to outline that this observation may be confounded by the remnant liver volume and the underlying liver function/disease as the majority of patients (88.27%) had various degrees of liver cirrhosis. Vascular infiltration negatively affects long-term survival in many studies [55, 59,60,61]. Interestingly, our study highlighted the presence of vascular tumor infiltration as a predictive factor of 90-day mortality. Of note, the rate of vascular tumor involvement included hepatic vein (HV), portal vein (PV), and inferior vena cava (IVC) infiltration rates of 12.24%, 2.04%, and 1.53% respectively. The high mortality rate of this subgroup is potentially linked to the greater proportion of extended hepatectomies (67.64%) with respect to anatomical and oncological considerations.
The presented results here must be interpreted taking into account the included study population and the proportion of minor and major resections performed. Over the years, we have followed a more liberal operative strategy with radical resections, even in older and comorbid patients, which might be an explanation of the overall higher mortality rate in the entire cohort compared to other reports. Noteworthy, we have observed a decline in the 90-day mortality rate from 19.69% to 7.8% since 2016. The same observation of improved outcome data was also reported by other studies [10, 62]. This trend in our cohort is largely attributed to careful patient selection for surgery, continuous technical modifications with less aggressive parenchyma resection, introduction of hepatobiliary scintigraphy, multidisciplinary approaches, and adjustment of modifiable pre-operative parameters. In fact, the rate of major hepatectomies in our department has fallen by almost 20% since 2016.
Our study has some important shortcomings with regard to its retrospective study design and the relatively small sample size. All data and results were derived from a single-western institution over an almost 20-years study period and are potentially not applicable to Asian cohorts with lower operative mortality rates. Additionally, technical refinements and changes in perioperative management and practice, which were not considered in our analysis, may have influenced the patient outcome after HR. Potential selection bias due to personal preferences and institutional guidelines, as well as missing value bias can further reduce the validity of the presented results. Furthermore, evolving non-surgical treatment modalities are not adequately addressed in this study setting. Finally, our data and results must be interpreted in the context of a natural evolution and improved learning curve in hepatic surgery from a European institution. Therefore, they could be an orientation and aid for comparable Western centers to adapt their surgical treatment strategy accordingly.
Conclusion
Advanced patient age, pre-existing chronic renal insufficiency, Child–Pugh Score, extended hepatic resection, and vascular tumor involvement were identified as significant predictive factors of 90-day mortality following elective HR in HCC. Hence, proper patient selection and adjustment of modifiable pre-and intraoperative parameters could reduce the 90-day mortality rate as a surrogate marker of surgical quality and safety. Larger scaled and multi-institutional studies with comparable patient cohorts are needed to further validate the presented results.
Data availability
All the used and/or analyzed datasets in this study are available from the corresponding author on reasonable request.
References
Cancer today. http://gco.iarc.fr/today/home. Accessed 24 Nov 2023
Llovet JM, Kelley RK, Villanueva A et al (2021) Hepatocellular carcinoma. Nat Rev Dis Primers 7:6. https://doi.org/10.1038/s41572-020-00240-3
Glantzounis GK, Paliouras A, Stylianidi M-C et al (2018) The role of liver resection in the management of intermediate and advanced stage hepatocellular carcinoma. A systematic review. Eur J Surg Oncol 44:195–208. https://doi.org/10.1016/j.ejso.2017.11.022
Pinna AD, Yang T, Mazzaferro V et al (2018) Liver transplantation and hepatic resection can achieve cure for hepatocellular carcinoma. Ann Surg 268:868–875. https://doi.org/10.1097/SLA.0000000000002889
Farges O, Malassagne B, Flejou JF et al (1999) Risk of major liver resection in patients with underlying chronic liver disease: a reappraisal. Ann Surg 229:210–215. https://doi.org/10.1097/00000658-199902000-00008
Shimada M, Rikimaru T, Sugimachi K et al (2000) The importance of hepatic resection for hepatocellular carcinoma originating from nonfibrotic liver. J Am Coll Surg 191:531–537. https://doi.org/10.1016/s1072-7515(00)00705-5
Dokmak S, Ftériche FS, Borscheid R et al (2013) 2012 Liver resections in the 21st century: we are far from zero mortality. HPB (Oxford) 15:908–915. https://doi.org/10.1111/hpb.12069
Xie Q-S, Chen Z-X, Zhao Y-J et al (2021) Systematic review of outcomes and meta-analysis of risk factors for prognosis after liver resection for hepatocellular carcinoma without cirrhosis. Asian J Surg 44:36–45. https://doi.org/10.1016/j.asjsur.2020.08.019
Ardito F, Famularo S, Aldrighetti L et al (2020) The impact of hospital volume on failure to rescue after liver resection for hepatocellular carcinoma: analysis from the HE.RC.O.LE.S. Ital Registry Ann Surg 272:840–846. https://doi.org/10.1097/SLA.0000000000004327
Ariizumi S-I, Katagiri S, Kotera Y et al (2022) Improved mortality, morbidity, and long-term outcome after anatomical hepatectomy with the glissonean pedicle approach in patients with hepatocellular carcinoma: 30 years’ experience at a single institute. Ann Surg 275:947–954. https://doi.org/10.1097/SLA.0000000000004311
Mise Y, Vauthey J-N, Zimmitti G et al (2015) Ninety-day postoperative mortality is a legitimate measure of hepatopancreatobiliary surgical quality. Ann Surg 262:1071–1078. https://doi.org/10.1097/SLA.0000000000001048
Schiergens TS, Dörsch M, Mittermeier L et al (2015) Thirty-day mortality leads to underestimation of postoperative death after liver resection: a novel method to define the acute postoperative period. Surgery 158:1530–1537. https://doi.org/10.1016/j.surg.2015.07.019
Chang C-M, Yin W-Y, Su Y-C et al (2014) Preoperative risk score predicting 90-day mortality after liver resection in a population-based study. Medicine 93:e59. https://doi.org/10.1097/MD.0000000000000059
Hyder O, Pulitano C, Firoozmand A et al (2013) A risk model to predict 90-day mortality among patients undergoing hepatic resection. J Am Coll Surg 216:1049–1056. https://doi.org/10.1016/j.jamcollsurg.2013.01.004
Dasari BVM, Hodson J, Sutcliffe RP et al (2019) Developing and validating a preoperative risk score to predict 90-day mortality after liver resection. J Surg Oncol 119:472–478. https://doi.org/10.1002/jso.25350
Malinchoc M, Kamath PS, Gordon FD et al (2000) A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology 31:864–871. https://doi.org/10.1053/he.2000.5852
Pugh RN, Murray-Lyon IM, Dawson JL et al (1973) Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 60:646–649. https://doi.org/10.1002/bjs.1800600817
Simons JP, Hill JS, Ng SC et al (2009) Perioperative mortality for management of hepatic neoplasm: a simple risk score. Ann Surg 250:929–934. https://doi.org/10.1097/SLA.0b013e3181bc9c2f
Teh SH, Christein J, Donohue J et al (2005) Hepatic resection of hepatocellular carcinoma in patients with cirrhosis: Model of End-Stage Liver Disease (MELD) score predicts perioperative mortality. J Gastrointest Surg 9:1207–1215. https://doi.org/10.1016/j.gassur.2005.09.008. (discussion 1215)
Teh SH, Sheppard BC, Schwartz J, Orloff SL (2008) Model for End-stage Liver Disease score fails to predict perioperative outcome after hepatic resection for hepatocellular carcinoma in patients without cirrhosis. Am J Surg 195:697–701. https://doi.org/10.1016/j.amjsurg.2007.05.054
Lei GY, Shen L, Junnarkar SP et al (2021) Predictors of 90-day mortality following hepatic resection for hepatocellular carcinoma. Visc Med 37:102–109. https://doi.org/10.1159/000510811
Soong R-S, Chen Y-C, Chou T-C et al (2022) A novel predictive scoring system for 90-day mortality among patients with hepatocellular cell carcinoma receiving major hepatectomy. Cancers (Basel) 14:1398. https://doi.org/10.3390/cancers14061398
Qi Y, LeVan TD, Haynatzki G et al (2020) Development of an Integer-based risk score to predict 90-day mortality after hepatectomy in patients with hepatocellular carcinoma. Am J Clin Oncol 43:640–647. https://doi.org/10.1097/COC.0000000000000724
Yin Y, Cheng J-W, Chen F-Y et al (2021) A novel preoperative predictive model of 90-day mortality after liver resection for huge hepatocellular carcinoma. Ann Transl Med 9:774. https://doi.org/10.21037/atm-20-7842
von Elm E, Altman DG, Egger M et al (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335:806–808. https://doi.org/10.1136/bmj.39335.541782.AD
Rahbari NN, Garden OJ, Padbury R et al (2011) Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 149:713–724. https://doi.org/10.1016/j.surg.2010.10.001
Brierley JD, Gospodarowicz MK, Wittekind C (2016) TNM classification of malignant tumours, 8th ed. Wiley-Blackwell
Nagino M, DeMatteo R, Lang H et al (2021) Proposal of a new comprehensive notation for hepatectomy: the “new world” terminology. Ann Surg 274:1–3. https://doi.org/10.1097/SLA.0000000000004808
Clavien PA, Barkun J, de Oliveira ML et al (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250:187–196. https://doi.org/10.1097/SLA.0b013e3181b13ca2
Webster AC, Nagler EV, Morton RL, Masson P (2017) Chronic kidney disease. Lancet 389:1238–1252. https://doi.org/10.1016/S0140-6736(16)32064-5
Knoefel WT, Gabor I, Rehders A et al (2013) In situ liver transection with portal vein ligation for rapid growth of the future liver remnant in two-stage liver resection. Br J Surg 100:388–394. https://doi.org/10.1002/bjs.8955
Alexander A, Lehwald-Tywuschik N, Rehders A et al (2023) Optimizing growth of the future liver remnant and making in-situ liver transsection safe-a standardized approach to ISLT or ALPPS. Curr Oncol 30:3277–3288. https://doi.org/10.3390/curroncol30030249
Lehwald-Tywuschik N, Vaghiri S, Schulte Am Esch J et al (2020) In situ split plus portal vein ligation (ISLT) - a salvage procedure following inefficient portal vein embolization to gain adequate future liver remnant volume prior to extended liver resection. BMC Surg 20:63. https://doi.org/10.1186/s12893-020-00721-y
Edmondson HA, Steiner PE (1954) Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 7:462–503. https://doi.org/10.1002/1097-0142(195405)7:3%3c462::aid-cncr2820070308%3e3.0.co;2-e
Zhong J, Ke Y, Gong W et al (2014) Hepatic resection associated with good survival for selected patients with intermediate and advanced-stage hepatocellular carcinoma. Ann Surg 260:329–340. https://doi.org/10.1097/SLA.0000000000000236
Kokudo T, Hasegawa K, Matsuyama Y et al (2016) Survival benefit of liver resection for hepatocellular carcinoma associated with portal vein invasion. J Hepatol 65:938–943. https://doi.org/10.1016/j.jhep.2016.05.044
Torzilli G, Makuuchi M, Inoue K et al (1999) No-mortality liver resection for hepatocellular carcinoma in cirrhotic and noncirrhotic patients: is there a way? A prospective analysis of our approach. Arch Surg 134:984–992. https://doi.org/10.1001/archsurg.134.9.984
Filmann N, Walter D, Schadde E et al (2019) Mortality after liver surgery in Germany. Br J Surg 106:1523–1529. https://doi.org/10.1002/bjs.11236
Vasavada B, Patel H (2022) Postoperative mortality after liver resection for hepatocellular carcinoma—A systematic review. Metanalysis and metaregression of studies published in the last 5 years. Surg Pract 26:123–130. https://doi.org/10.1111/1744-1633.12569
Santambrogio R, Barabino M, Scifo G et al (2017) Effect of age (over 75 Years) on postoperative complications and survival in patients undergoing hepatic resection for hepatocellular carcinoma. J Gastrointest Surg 21:657–665. https://doi.org/10.1007/s11605-016-3354-1
Okinaga H, Yasunaga H, Hasegawa K et al (2018) Short-term outcomes following hepatectomy in elderly patients with hepatocellular carcinoma: an analysis of 10,805 septuagenarians and 2,381 octo- and nonagenarians in Japan. Liver Cancer 7:55–64. https://doi.org/10.1159/000484178
Tan J-T, Zhao C, Peng N-F et al (2016) Association between age and overall survival of patients with hepatocellular carcinoma after hepatic resection. J Surg Oncol 114:966–970. https://doi.org/10.1002/jso.24434
Mizuguchi T, Kawamoto M, Meguro M et al (2015) Impact of aging on morbidity and mortality after liver resection: a systematic review and meta-analysis. Surg Today 45:259–270. https://doi.org/10.1007/s00595-014-0863-y
Faber W, Stockmann M, Schirmer C et al (2014) Significant impact of patient age on outcome after liver resection for HCC in cirrhosis. Eur J Surg Oncol 40:208–213. https://doi.org/10.1016/j.ejso.2013.10.018
Faber W, Sharafi S, Stockmann M et al (2014) Patient age and extent of liver resection influence outcome of liver resection for hepatocellular carcinoma in non-cirrhotic liver. Hepatogastroenterology 61:1925–1930
Pasetto LM, Lise M, Monfardini S (2007) Preoperative assessment of elderly cancer patients. Crit Rev Oncol Hematol 64:10–18. https://doi.org/10.1016/j.critrevonc.2007.08.001
Mizuguchi T, Kawamoto M, Meguro M et al (2014) Preoperative liver function assessments to estimate the prognosis and safety of liver resections. Surg Today 44:1–10. https://doi.org/10.1007/s00595-013-0534-4
Orii T, Takayama T, Haga I et al (2008) Efficacy of a liver resection for hepatocellular carcinoma in patients with chronic renal failure. Surg Today 38:329–334. https://doi.org/10.1007/s00595-007-3634-1
Shirata C, Hasegawa K, Kokudo T et al (2018) Liver resection for hepatocellular carcinoma in patients with renal dysfunction. World J Surg 42:4054–4062. https://doi.org/10.1007/s00268-018-4698-3
Liu X-Y, Zhao Z-Q, Cheng Y-X et al (2022) Does chronic kidney disease really affect the complications and prognosis after liver resection for hepatocellular carcinoma? A Meta-Analysis Front Surg 9:870946. https://doi.org/10.3389/fsurg.2022.870946
Morandi A, Risaliti M, Montori M et al (2023) Predicting post-hepatectomy liver failure in HCC patients: a review of liver function assessment based on laboratory tests scores. Medicina (Kaunas) 59:1099. https://doi.org/10.3390/medicina59061099
Durand F, Valla D (2005) Assessment of the prognosis of cirrhosis: Child-Pugh versus MELD. J Hepatol 42(Suppl):S100-107. https://doi.org/10.1016/j.jhep.2004.11.015
Wang Y-Y, Zhong J-H, Su Z-Y et al (2016) Albumin-bilirubin versus Child-Pugh score as a predictor of outcome after liver resection for hepatocellular carcinoma. Br J Surg 103:725–734. https://doi.org/10.1002/bjs.10095
Berardi G, Morise Z, Sposito C et al (2020) Development of a nomogram to predict outcome after liver resection for hepatocellular carcinoma in Child-Pugh B cirrhosis. J Hepatol 72:75–84. https://doi.org/10.1016/j.jhep.2019.08.032
Capussotti L, Muratore A, Amisano M et al (2005) Liver resection for hepatocellular carcinoma on cirrhosis: analysis of mortality, morbidity and survival–a European single center experience. Eur J Surg Oncol 31:986–993. https://doi.org/10.1016/j.ejso.2005.04.002
Egger ME, Ohlendorf JM, Scoggins CR et al (2015) Assessment of the reporting of quality and outcome measures in hepatic resections: a call for 90-day reporting in all hepatectomy series. HPB (Oxford) 17:839–845. https://doi.org/10.1111/hpb.12470
Simons JP, Ng SC, Hill JS et al (2010) In-hospital mortality from liver resection for hepatocellular carcinoma: a simple risk score. Cancer 116:1733–1738. https://doi.org/10.1002/cncr.24904
Moris D, Tsilimigras DI, Kostakis ID et al (2018) Anatomic versus non-anatomic resection for hepatocellular carcinoma: a systematic review and meta-analysis. Eur J Surg Oncol 44:927–938. https://doi.org/10.1016/j.ejso.2018.04.018
Lee Y-H, Hsu C-Y, Huang Y-H et al (2014) Vascular invasion in hepatocellular carcinoma: prevalence, determinants and prognostic impact. J Clin Gastroenterol 48:734–741. https://doi.org/10.1097/MCG.0b013e3182a8a254
Hsieh C-H, Wei C-K, Yin W-Y et al (2015) Vascular invasion affects survival in early hepatocellular carcinoma. Mol Clin Oncol 3:252–256. https://doi.org/10.3892/mco.2014.420
Jonas S, Bechstein WO, Steinmüller T et al (2001) Vascular invasion and histopathologic grading determine outcome after liver transplantation for hepatocellular carcinoma in cirrhosis. Hepatology 33:1080–1086. https://doi.org/10.1053/jhep.2001.23561
Taketomi A, Kitagawa D, Itoh S et al (2007) Trends in morbidity and mortality after hepatic resection for hepatocellular carcinoma: an institute’s experience with 625 patients. J Am Coll Surg 204:580–587. https://doi.org/10.1016/j.jamcollsurg.2007.01.035
Acknowledgements
Not applicable
Funding
Open Access funding enabled and organized by Projekt DEAL. Open Access funding enabled and organized by Project DEAL.
Author information
Authors and Affiliations
Contributions
Study conception and design (S.V., and L.D.), Acquisition of data (S.V., S.A.S., and D.P.), Analysis and interpretation of data (S.V., L.D., S.K., and A.A.), Statistical analysis (L.D., and S.V.), Drafting of manuscript (S.V., L.D., and A.A.), Critical revision of manuscript (N.L.T., and W.T.K.). All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the local institutional review board (Heinrich-Heine-University, Duesseldorf, Germany; study-no.: 2021–1800- KFogU). All procedures performed in this study were in accordance with the ethical standards in the 1964 Declaration of Helsinki and its later amendments. Informed consent was waived because no data regarding the cases were disclosed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vaghiri, S., Lehwald-Tywuschik, N., Prassas, D. et al. Predictive factors of 90-day mortality after curative hepatic resection for hepatocellular carcinoma: a western single-center observational study. Langenbecks Arch Surg 409, 149 (2024). https://doi.org/10.1007/s00423-024-03337-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-024-03337-5