Abstract
Background
Adenoid cystic carcinoma (ACC) is a rare type of cancer commonly occurring in salivary glands. It is characterized by slow but infiltrative growth, nerve infiltration and overall poor prognosis, with late recurrence and distant metastasis. The treatment of ACC is still limited to surgery and/or (adjuvant) radiotherapy. Till now no promising systemic therapy option exists. However, various studies deliver promising results after treatment with anti-angiogenetic agents, such as anti-EGFR-antibody Cetuximab or Tyrosinkinase inhibitor Lenvatinib.
Methods
By using of immunohistological methods we analyzed and compared the macrophage and lymphocyte populations, vascularization, and PD-L1-status in 12 ACC of the salivary glands.
Results
All cases showed a significant elevation of macrophages with M2 polarization and a higher vascularization in ACC compared to normal salivary gland tissue. The CD4/CD8 quotient was heterogenous. ACC does not show relevant PD-L1 expression.
Conclusions
The predominant M2 polarization of macrophages in ACC could be responsible for elevated vascularization, as already been proved in other cancer types, that M2 macrophages promote angiogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Salivary gland malignancies are overall rare and almost one fifth of them are adenoid cystic carcinomas (ACC) [1]. Based on its growing pattern, ACC is divided in three histological types, namely, cribriform, tubular and solid. In total the cribriform type has the best and solid type the worst prognosis [2]. An infiltration of nerves is also a typical feature of ACC and is correlated with a poorer outcome of the disease [3].
Former studies have shown an elevated expression of epidermal growth factor receptor (EGFR) in ACC [4, 5]. This suggests a high tendency to neovascularization, so that ACC might be susceptible to anti-angiogenetic agents, such as Cetuximab or Lenvatinib. On the other hand ACC show a weak to no programmed death ligand-1 (PD-L1) expression [6], a decisive biomarker for the use of Pembrolizumab in head and neck squamous cell carcinoma, which makes the benefit from immune checkpoint inhibitors questionable. ACC are low immunogenic tumors with low infiltration rates through CD8-positive lymphocytes [6], which is a crucial part of the anticancer immune response in the tumor microenvironment that can affect the outcome [7,8,9]. Furthermore, the CD4/CD8 ratio is in some cancer types, like the triple negative breast cancer, a useful prognostic tool [10], but can also be used as indicator to monitor the course of a therapy with immune checkpoint inhibitors [11]. However, its relevance in ACC is not yet analyzed.
Angiogenesis is a critical part of tumor growth and progression in general [12] and previous reports indirectly suggest elevated angiogenesis in ACC [13, 14], whereas a similar immmunohistological study did not show any significant difference between intratumoral and peritumoral vascularization [15]. Macrophages are a heterogenous group of cells that polarise to a M1 or M2 phenotype and are able to retain their plasticity and transform according to environmental signals [16,17,18]. M2-polarized macrophages, which have been linked to angiogenesis and cancer growth in pancreas [19], are also strongly represented in ACC [20, 21] and perhaps, at least partially, responsible for enhanced angiogenesis in this cancer. Till Dato no study to our knowledge compared both aspects.
In the era of personalized medicine, where monoclonal antibodies revolutionised oncology, the treatment of ACC does not appear to profit from these developments, as its curative therapy is still limited to surgery with/or radiation [22] and the therapy outcome is still not satisfactory. Up to now, no systemic treatment seems to be able to achieve satisfactory results [23,24,25].
In our study we used immunohistochemical methods to investigate the immunological microenvironment including vascularization of tumor and normal salivary gland tissue to examine if there are any abnormalities, that could be used as basis for translational development of new therapy regimens for ACC.
Materials and methods
Patients
Twelve Patients (8 female, 4 male) with mean age at time of diagnosis of 59.5 years, treated at the University Hospital of Regensburg, Germany were retrospectively included in this study (Table 1). The diagnosis of ACC was reviewed and confirmed. Tissue from the pathological routine diagnostics from these 12 primary ACC of the salivary glands was used for examination. After achieving a positive ethics votum (22-2753-104) an in situ immunohistochemical characterization of the immunological tumor environment was performed. For the in-situ characterization, standard routine diagnostic procedures and antibodies were applied. First, a new hematoxylin–eosin slide was prepared (Fig. 1).
The tumor and residual salivary gland tissue were identified. In three tissue samples no normal salivary gland tissue was available for examination. Then, immunohistochemical reactions for PD-L1 (Dako Anti-Human PD-L1 Clone 22C3), CD4 (Ventana anti-CD4 Rabbit Monoclonal Primary Antibody Clone SP35) and CD8 (Ventana anti-CD8 Rabbit Monoclonal Primary Antibody, Clone SP57) for lymphocytes, CD68 (Dako Anti-Human CD68, Clone KP1) for macrophages and CD163 (NovocastraTM Lyophilized Mouse Monoclonal Antibody CD163, Clone 10D6) for M2-macrophages were performed. In addition, a CD31 (Dako Monoclonal Mouse Anti-Human CD31, Clone JC70) reaction was used to identify the vessel endothelial cells. All immunohistochemical stains were performed on tissue sections (2–4 µm thickness), prepared from-formalin-fixed (4% neutral buffered formalin) paraffin-embedded tissue blocks. Immunohistochemical staining was performed using a Roche Ventana Benchmark Ultra automated slide stainer (Ventana Medical Systems, Roche, France).
All slides were scanned (3DHISTECH Ltd.Pannoramic slide scanner 250) and evaluated using a virtual microscopy software (3DHISTECH Ltd. Case Viewer Ver.2.2) (Figs. 2, 3).
Comparison of the vascularization and immune cells in ACC and normal salivary gland tissue. Dots representing the mean number of counted cells or vessels per high power field (HPF). a CD68 + CD163− macrophages (M1) versus CD68 + CD163 + macrophages (M2) only in tumor tissue. b CD68 + macrophages, c CD31 + vessels and d CD8 + and CD4 + lymphocytes in tumor and normal salivary gland tissue. p < 0.05; **p < 0.005; ***p < 0.001
For PD-L1 the three common diagnostic scores [tumor proportion score (TPS), Immune Cell Score (IC) and combined positive Score (CPS)] were assessed according the guidelines by an experienced consultant pathologist [26].
To quantify the lymphocytes, ten high power fields (HPF) of the invasion tumor front were examined and the positive cells were counted. The averages and then the quotient of CD4 to CD8 lymphocytes were calculated.
The CD68 reaction was used to visualize all macrophages. To evaluate the quantity of the macrophages ten HPF of the invasion tumor front and normal salivary gland tissue were examined and the positive cells were counted. Then, the mean value (Confidence interval 95%, CI 95%) of macrophages within the tumor front and the salivary gland tissue was calculated. The macrophages were then further characterized concerning their subpopulations. M2-macrophages were detected using an anti-CD163-antibody. Utilising the same, previously described procedure for the quantification of CD68-positive macrophages, the M2-macrophages were also quantified. Using the numbers of CD68-positive-macrophages and CD163-positive–M2-macrophages the number of M1-macrophages per HPF was calculated.
For the quantification of blood vessels, the endothelial cells were marked using a CD31-reaction. The vessel count in cross sections was performed within the tumour and normal salivary gland tissue, examining the same ten HPF, respectively, followed by calculation of the mean values like previously described.
Statistical analysis
All acquired results were documented in Microsoft Excel 16.58 (Microsoft Corporation, Redmond, WA, U.S.A.). The calculations and graphs were also produced with GraphPad Prism version 9.3.1 for MacOS (GraphPad Software, San Diego, California USA). To investigate if there are significant differences between M1 and M2 macrophages, but also vessels and CD4 + and CD8 + lymphocytes in tumor and healthy salivary gland tissue, we applied the Wilcoxon signed ranged test. A p value ≤ 0.05 in two tailed tests was considered as statistically significant.
Results
The majority of macrophages in ACC shows an M2 polarization
In eight samples statistically significant more macrophages were observed in the tumor front in comparison with normal salivary gland tissue (Fig. 2a). In one sample nearly the same number of macrophages was found within the salivary gland tissue and the tumor, whereby this sample also showed signs of sialadenitis. For the three remaining samples a comparison of macrophages within the tumor and salivary gland was not possible due to lack of tumor free salivary gland tissue. Despite this, the macrophages of the tumor were calculated in all cases and further characterized in subpopulations of M1 and M2 macrophages. Interestingly, the majority of macrophages was polarized to an M2-phenotype (p = 0.0005), whereas only a small number of macrophages represented the M1-phenotype (Figs. 2b and 3).
Elevated intratumoral vascularization compared to normal salivary gland tissue
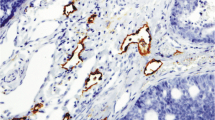
In the analyzed tissues, in which both tumor and normal salivary gland tissue were available, we observed a much higher vascularization in the tumor compared to normal tissue. Intratumoral vessels were in average elevated by 2.89 times in the tumor tissue compared to local salivary gland tissue (p = 0.0039) (Figs. 2c and 4).
Heterogeneous lymphocytes infiltration in ACC
Besides three cases, where no normal salivary gland tissue was available and another one in which no more tissue was available for examination, the intratumoral CD4 positive lymphocytes where elevated from 1.7 to 9.9 times compared to normal tissue, but the presence of CD4-positive lymphocytes was not in the same range. In six cases the range of intratumoral CD8-positive lymphocytes was elevated up to 17 times. In general though, comparing the number of lymphocytes in the tumor and normal tissue, revealed no statistically significant difference (Fig. 2d). After evaluating and comparing the quotient of CD4-to-CD8 intratumoral lymphocytes, we observed heterogenous results. In four out of eleven cases more CD4 lymphocytes were found and in seven cases the CD8 lymphocytes compared to CD4 lymphocytes dominated.
PD-1 status in ACC
In terms of PD-L1 status no relevant expression was observed. More specifically all cases showed a TPS of 0. The vast majority of the samples (10 out of 12) also showed an IC and CPS of 0. In two cases we observed slightly positive IC and CPS scores with a maximum of 2% and 3%, respectively (Table 1). This shows that the tumor cells show no immuno-histochemical expression of PD-L1. In addition, the immunal cells show no or minimal positivity for the immunohistochemical reaction for PD-L1.
Discussion
In the present study we clearly demonstrated an elevated number of macrophages in the tumor environment in comparison with normal salivary gland tissue but even more interesting is the finding of the almost complete polarization of macrophages in CD163-positive–M2-macrophages. It is well-known that the polarization of macrophages affects in various ways the behavior of tumors [27, 28]. M2 macrophages promote tumor aggression and progress [19, 29] in contrary to M1-macrophages which have an antitumoral effect [28]. They are also known for their role in the tumor angiogenesis [19, 30]. This is also consistent with our findings, showing significantly more vessels within the tumor tissue compared to normal salivary gland tissue (p = 0.0039), as well as significantly more M2 macrophages in comparison with M1 macrophages.
Recently Yang et al. showed in their experiments, that in ACC the mechanism of CCL2/CCR2 axis in the interactions between tumor cells and tumor-associated macrophages during the progression of salivary ACC, promotes a polarization to M2 tumor-associated macrophages [20], which explains the significantly higher polarization (p = 0.0005) of macrophages in the M2-subpopulation found in our study.
Adenoid cystic carcinomas are rare tumors and represent a challenge for clinicians. Confirming already performed studies, ACC are negative or extremely low positive for PD-L1 (all three scores), also suggesting a failure of immune check point inhibitor therapy.
The CD4/CD8-ratio of tumor infiltrating lymphocytes possesses in some malignancies—like gastric cancer [31] or as previously mentioned in the triple negative breast carcinoma [10]—a predictive value. In our study the ACC shows great heterogeneity in this matter and of course a larger collective is necessary to proof its clinical value.
These results, especially concerning the elevated macrophage infiltrate of the tumor front, their obvious polarization in M2 Macrophages and the also significantly elevated neovascularization, could help to develop new strategies of treating ACC. Potential inhibition of the angiogenesis, using well-known anti-angiogenetic agents may bring an additional effect regarding the cure of ACC. Our results support various studies and reports involving Cetuximab in the treatment of ACC showing promising results [32,33,34,35,36]. In addition, the tyrosine kinase inhibitor (TKI) Lenvatinib achieved good results in the treatment of patients with recurrent or metastasized ACC [37, 38], as well as other TKI-like sorafenib [39, 40] and axitinib [41]. However, there is a lack of prospective randomized controlled phase III studies that confirm the effectiveness of such therapies, mainly because of the rarity and slow growth of these tumors. Furthermore, controlling the population of M2-macrophages in tumor is already being investigated as a potential cancer treatment with promising results, but this concept is still far from finding its way to routine daily practice [42]. Nevertheless, targeting tumor-associated macrophages might be a breakthrough in the treatment of ACC, especially in patients with metastatic or advanced disease, which lack options of effective therapy. As soon as such substances are available, they should be investigated in animal models and also phase II and III studies to find their way in the clinical practice if they prove to be effective. It is, therefore, necessary that large cancer centers cooperate and perform together such studies to collect the essential number of patients, so that highly qualitative and representative results can lead to novel therapies for ACC. ACC is a complicated deadly systemic disease so a wider therapeutical approach, targeting various features of the tumor itself or its environment is in our opinion the key to treat this tumor entity or at least control its course.
In the study of Li et al. showed recently that macrophage migration inhibitory factor was overexpressed in ACC. MIF might be a risk factor for ACC patients [43] and seems also to promote the perineural invasion in ACC patients [44]. Until now there are no clear data, how this factor affects the polarization of macrophages. This is a key point for examining in further studies.
Next step is to examine the general effect of M2-macrophages in ACC using molecular pathology techniques, where protein and RNA-based studies shall be performed.
We are aware that the small number of examined cases poses as a limitation of our study. However, all cases showed statistically consistent results, so that the power of the results is credible and confirmed. It is, therefore, necessary to perform larger studies to confirm our results and taking in consideration the rarity of ACC this would be easier with the cooperation of other large cancer centers.
Conclusion
The fact that the ACC invasion front is being dominated by a large number of macrophages with M2 phenotype and also the highly elevated angiogenesis in the tumor tissue maybe the clue to treat a stubborn until today disease. Larger studies that require cooperation of many cancer centers are urgently required and we are willing to be a part of such national or international initiative.
Data availability
The data sets presented in this article are not freely available because of patient confidentiality and participant privacy terms.
References
Hellquist H, Skalova A (2014) Adenoid cystic carcinoma. Springer, Berlin, pp 221–260
Du F, Zhou CX, Gao Y (2016) Myoepithelial differentiation in cribriform, tubular and solid pattern of adenoid cystic carcinoma: a potential involvement in histological grading and prognosis. Ann Diagn Pathol 22:12–17. https://doi.org/10.1016/j.anndiagpath.2016.03.001
Liu X, Yang X, Zhan C, Zhang Y, Hou J, Yin X (2020) Perineural invasion in adenoid cystic carcinoma of the salivary glands: where we are and where we need to go. Front Oncol 10:1493. https://doi.org/10.3389/fonc.2020.01493
Bell D, Roberts D, Kies M, Rao P, Weber RS, El-Naggar AK (2010) Cell type-dependent biomarker expression in adenoid cystic carcinoma. Cancer 116(24):5749–5756. https://doi.org/10.1002/cncr.25541
Nightingale J, Lum B, Ladwa R, Simpson F, Panizza B (2021) Adenoid cystic carcinoma: a review of clinical features, treatment targets and advances in improving the immune response to monoclonal antibody therapy. Biochim Biophys Acta Rev Cancer 1875(2):188523. https://doi.org/10.1016/j.bbcan.2021.188523
Mosconi C, de Arruda JAA, de Farias ACR, Oliveira GAQ, de Paula HM, Fonseca FP et al (2019) Immune microenvironment and evasion mechanisms in adenoid cystic carcinomas of salivary glands. Oral Oncol 88:95–101. https://doi.org/10.1016/j.oraloncology.2018.11.028
Afonina IS, Cullen SP, Martin SJ (2010) Cytotoxic and non-cytotoxic roles of the CTL/NK protease granzyme B. Immunol Rev 235(1):105–116. https://doi.org/10.1111/j.0105-2896.2010.00908.x
Wang J, Li R, Cao Y, Gu Y, Fang H, Fei Y et al (2021) Intratumoral CXCR5+CD8+T associates with favorable clinical outcomes and immunogenic contexture in gastric cancer. Nat Commun. https://doi.org/10.1038/s41467-021-23356-w
Oshi M, Asaoka M, Tokumaru Y, Yan L, Matsuyama R, Ishikawa T et al (2020) CD8 T cell score as a prognostic biomarker for triple negative breast cancer. Int J Mol Sci 21(18):6968. https://doi.org/10.3390/ijms21186968
Wang K, Shen T, Siegal GP, Wei S (2017) The CD4/CD8 ratio of tumor-infiltrating lymphocytes at the tumor-host interface has prognostic value in triple-negative breast cancer. Hum Pathol 69:110–117. https://doi.org/10.1016/j.humpath.2017.09.012
Shindo G, Endo T, Onda M, Goto S, Miyamoto Y, Kaneko T (2013) Is the CD4/CD8 ratio an effective indicator for clinical estimation of adoptive immunotherapy for cancer treatment? J Cancer Ther 04(08):1382–1390. https://doi.org/10.4236/jct.2013.48164
Zuazo-Gaztelu I, Casanovas O (2018) Unraveling the role of angiogenesis in cancer ecosystems. Front Oncol 8:248. https://doi.org/10.3389/fonc.2018.00248
House SL, Castro AM, Lupu TS, Weinheimer C, Smith C, Kovacs A et al (2016) Endothelial fibroblast growth factor receptor signaling is required for vascular remodeling following cardiac ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 310(5):H559–H571. https://doi.org/10.1152/ajpheart.00758.2015
Myoken Y, Myoken Y, Okamoto T, Sato JD, Kan M, Mckeehan WL et al (1996) Immunohistochemical study of overexpression of fibroblast growth factor-1 (fgf-1), fgf-2, and fgf receptor-1 in human malignant salivary gland tumours. J Pathol 178(4):429–436. https://doi.org/10.1002/(SICI)1096-9896(199604)178:4%3c429::AID-PATH495%3e3.0.CO;2-1
KoochekDezfuli M, Seyedmajidi M, Nafarzadeh S, Yazdani F, Bijani A (2019) Angiogenesis and lymphangiogenesis in salivary gland adenoid cystic carcinoma and mucoepidermoid carcinoma. Asian Pacific J Cancer Prev. 20(12):3547–3553. https://doi.org/10.31557/apjcp.2019.20.12.3547
Stout RD, Jiang C, Matta B, Tietzel I, Watkins SK, Suttles J (2005) Macrophages sequentially change their functional phenotype in response to changes in microenvironmental influences. J Immunol 175(1):342–349. https://doi.org/10.4049/jimmunol.175.1.342
Stout RD, Suttles J (2004) Functional plasticity of macrophages: reversible adaptation to changing microenvironments. J Leukoc Biol 76(3):509–513. https://doi.org/10.1189/jlb.0504272
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8(12):958–969. https://doi.org/10.1038/nri2448
Yang Y, Guo Z, Chen W, Wang X, Cao M, Han X et al (2021) M2 macrophage-derived exosomes promote angiogenesis and growth of pancreatic ductal adenocarcinoma by targeting E2F2. Mol Ther 29(3):1226–1238. https://doi.org/10.1016/j.ymthe.2020.11.024
Yang Z, Li H, Wang W, Zhang J, Jia S, Wang J et al (2019) CCL2/CCR2 axis promotes the progression of salivary adenoid cystic carcinoma via recruiting and reprogramming the tumor-associated macrophages. Front Oncol 9:231. https://doi.org/10.3389/fonc.2019.00231
Linxweiler M, Kuo F, Katabi N, Lee M, Nadeem Z, Dalin MG et al (2020) The immune microenvironment and neoantigen landscape of aggressive salivary gland carcinomas differ by subtype. Clin Cancer Res 26(12):2859–2870. https://doi.org/10.1158/1078-0432.ccr-19-3758
Rodriguez-Russo CA, Junn JC, Yom SS, Bakst RL (2021) Radiation therapy for adenoid cystic carcinoma of the head and neck. Cancers 13(24):6335. https://doi.org/10.3390/cancers13246335
Mahmood U, Bang A, Chen YH, Mak RH, Lorch JH, Hanna GJ et al (2021) A randomized phase 2 study of pembrolizumab with or without radiation in patients with recurrent or metastatic adenoid cystic carcinoma. Int J Radiat Oncol Biol Phys 109(1):134–144. https://doi.org/10.1016/j.ijrobp.2020.08.018
Papaspyrou G, Hoch S, Rinaldo A, Rodrigo JP, Takes RP, van Herpen C et al (2011) Chemotherapy and targeted therapy in adenoid cystic carcinoma of the head and neck: a review. Head Neck 33(6):905–911. https://doi.org/10.1002/hed.21458
Laurie SA, Ho AL, Fury MG, Sherman E, Pfister DG (2011) Systemic therapy in the management of metastatic or locally recurrent adenoid cystic carcinoma of the salivary glands: a systematic review. Lancet Oncol 12(8):815–824. https://doi.org/10.1016/s1470-2045(10)70245-x
Schildhaus HU (2018) Der prädiktive Wert der PD-L1-Diagnostik. Pathologe 39(6):498–519. https://doi.org/10.1007/s00292-018-0507-x
Rhee I (2016) Diverse macrophages polarization in tumor microenvironment. Arch Pharmacal Res 39(11):1588–1596. https://doi.org/10.1007/s12272-016-0820-y
Pan Y, Yu Y, Wang X, Zhang T (2020) Tumor-associated macrophages in tumor immunity. Front Immunol 11:583084. https://doi.org/10.3389/fimmu.2020.583084
Suarez-Lopez L, Sriram G, Kong YW, Morandell S, Merrick KA, Hernandez Y et al (2018) MK2 contributes to tumor progression by promoting M2 macrophage polarization and tumor angiogenesis. Proc Natl Acad Sci U S A 115(18):E4236–E4244. https://doi.org/10.1073/pnas.1722020115
Jetten N, Verbruggen S, Gijbels MJ, Post MJ, De Winther MPJ, Donners MMPC (2014) Anti-inflammatory M2, but not pro-inflammatory M1 macrophages promote angiogenesis in vivo. Angiogenesis 17(1):109–118. https://doi.org/10.1007/s10456-013-9381-6
Zurlo IV, Schino M, Strippoli A, Calegari MA, Cocomazzi A, Cassano A et al (2022) Predictive value of NLR, TILs (CD4+/CD8+) and PD-L1 expression for prognosis and response to preoperative chemotherapy in gastric cancer. Cancer Immunol Immunother 71(1):45–55. https://doi.org/10.1007/s00262-021-02960-1
Caballero M, Sosa AE, Tagliapietra A, Grau JJ (2013) Metastatic adenoid cystic carcinoma of the salivary gland responding to cetuximab plus weekly paclitaxel after no response to weekly paclitaxel alone. Head Neck 35(2):E52–E54. https://doi.org/10.1002/hed.21870
Hitre E, Budai B, Takácsi-Nagy Z, Rubovszky G, Tóth E, Remenár É et al (2013) Cetuximab and platinum-based chemoradio- or chemotherapy of patients with epidermal growth factor receptor expressing adenoid cystic carcinoma: a phase II trial. Br J Cancer 109(5):1117–1122. https://doi.org/10.1038/bjc.2013.468
Jensen AD, Krauss J, Weichert W, Debus J, Münter MW (2010) RadioImmunotherapy for adenoid cystic carcinoma: a single-institution series of combined treatment with cetuximab. Radiat Oncol 5(1):102. https://doi.org/10.1186/1748-717x-5-102
Hitre E, Budai B, Takacsi-Nagy Z, Rubovszky G, Toth E, Remenar E et al (2013) Cetuximab and platinum-based chemoradio- or chemotherapy of patients with epidermal growth factor receptor expressing adenoid cystic carcinoma: a phase II trial. Br J Cancer 109(5):1117–1122. https://doi.org/10.1038/bjc.2013.468
Mueller SK, Haderlein M, Lettmaier S, Agaimy A, Haller F, Hecht M et al (2022) Targeted therapy, chemotherapy, immunotherapy and novel treatment options for different subtypes of salivary gland Cancer. J Clin Med 11(3):720. https://doi.org/10.3390/jcm11030720
Tchekmedyian V, Sherman EJ, Dunn L, Tran C, Baxi S, Katabi N et al (2019) Phase II study of lenvatinib in patients with progressive, recurrent or metastatic adenoid cystic carcinoma. J Clin Oncol 37(18):1529–1537. https://doi.org/10.1200/jco.18.01859
Feeney L, Jain Y, Beasley M, Donnelly O, Kong A, Moleron R et al (2021) Centralised RECIST assessment and clinical outcomes with lenvatinib monotherapy in recurrent and metastatic adenoid cystic carcinoma. Cancers 13(17):4336. https://doi.org/10.3390/cancers13174336
Thomson DJ, Silva P, Denton K, Bonington S, Mak SK, Swindell R et al (2015) Phase II trial of sorafenib in advanced salivary adenoid cystic carcinoma of the head and neck. Head Neck 37(2):182–187. https://doi.org/10.1002/hed.23577
Locati LD, Perrone F, Cortelazzi B, Bergamini C, Bossi P, Civelli E et al (2016) A phase II study of sorafenib in recurrent and/or metastatic salivary gland carcinomas: translational analyses and clinical impact. Eur J Cancer 69:158–165. https://doi.org/10.1016/j.ejca.2016.09.022
Locati LD, Cavalieri S, Bergamini C, Resteghini C, Alfieri S, Calareso G et al (2019) Phase II trial with axitinib in recurrent and/or metastatic salivary gland cancers of the upper aerodigestive tract. Head Neck 41(10):3670–3676. https://doi.org/10.1002/hed.25891
Hu G, Guo M, Xu J, Wu F, Fan J, Huang Q et al (2019) Nanoparticles targeting macrophages as potential clinical therapeutic agents against cancer and inflammation. Front Immunol 10:1998. https://doi.org/10.3389/fimmu.2019.01998
Li C, Chen Q, Tian Z, Li S, Gong Z, Lin Z et al (2019) Expression of MIF, Beclin1, and LC3 in human salivary gland adenoid cystic carcinoma and its prognostic value. Medicine (Baltimore) 98(20):e15402-e. https://doi.org/10.1097/MD.0000000000015402
Zhang M, Li ZF, Wang HF, Wang SS, Yu XH, Wu JB et al (2019) MIF promotes perineural invasion through EMT in salivary adenoid cystic carcinoma. Mol Carcinog 58(6):898–912. https://doi.org/10.1002/mc.22979
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: IM, CB, AM. Methodology: IM, AM. Validation: IM, JK, CB and AM. Formal analysis: IM, AM. Investigation: IM, AM. Resources: TE, JK, PB, CB, CB. Data curation: IM, TE, AM. Writing—original draft preparation: IM, AM. Writing—review and editing, all authors. Visualization: IM, AM. Supervision: JK, CB, AM. Project administration: AM, CB. Funding acquisition, CB and CB. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Michaelides, I., Künzel, J., Ettl, T. et al. Adenoid cystic carcinoma of the salivary glands: a pilot study of potential therapeutic targets and characterization of the immunological tumor environment and angiogenesis. Eur Arch Otorhinolaryngol 280, 2937–2944 (2023). https://doi.org/10.1007/s00405-023-07884-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-023-07884-3