Abstract
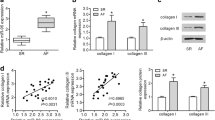
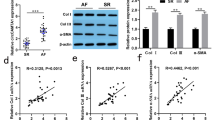
Atrial fibrosis is important for the pathogenesis of atrial fibrillation (AF) but the underlying signal transduction is incompletely understood. We therefore studied the role of microRNA-21 (miR-21) and its downstream target Sprouty 1 (Spry1) during atrial fibrillation. Left atria (LA) from patients with AF showed a 2.5-fold increased expression of miR-21 compared to matched LA of patients in sinus rhythm. Increased miR-21 expression correlated positively with atrial collagen content and was associated with a reduced protein expression of Spry1 and increased expression of connective tissue growth factor (CTGF), lysyl oxidase and Rac1-GTPase. Neonatal cardiac fibroblasts treated with angiotensin II (AngII) or CTGF showed an increased miR-21 and decreased Spry1 expression. Pretreatment with an inhibitor of Rac1 GTPase, NSC23766, reduced the AngII-induced upregulation of miR-21. A small molecule inhibitor of lysyl oxidase, BAPN, prevented the AngII as well as the CTGF-induced miR-21 expression. Transgenic mice with cardiac overexpression of Rac1, which develop spontaneous AF and atrial fibrosis with increasing age, showed upregulation of miR-21 expression associated with reduced Spry1 expression. miR-21 expression and signalling in vivo were prevented by long-term treatment of the mice with statins. Direct inhibition of miR-21 by antagomir-21 prevented fibrosis of the atrial myocardium post-myocardial infarction. Left atria of patients with atrial fibrillation are characterized by upregulation of miR-21 und reduced expression of Spry1. Activation of Rac1 by angiotensin II leads to a CTGF- and lysyl oxidase-mediated increase of miR-21 expression contributing to structural remodelling of the atrial myocardium.






Similar content being viewed by others
References
Adam O, Frost G, Custodis F, Sussman MA, Schäfers HJ, Böhm M, Laufs U (2007) Role of Rac1 GTPase activation in atrial fibrillation. J Am Coll Cardiol 50:359–367. doi:10.1016/j.jacc.2007.03.041
Adam O, Lavall D, Theobald K, Hohl M, Grube M, Ameling S, Sussman MA, Rosenkranz S, Kroemer HK, Schäfers HJ, Böhm M, Laufs U (2010) Rac1-induced connective tissue growth factor regulates connexin 43 and N-cadherin expression in atrial fibrillation. J Am Coll Cardiol 55:469–480. doi:10.1016/j.jacc.2009.08.064
Adam O, Theobald K, Lavall D, Grube M, Kroemer HK, Ameling S, Schäfers HJ, Böhm M, Laufs U (2011) Increased lysyl oxidase expression and collagen cross-linking during atrial fibrillation. J Mol Cell Cardiol 50:678–685. doi:10.1016/j.yjmcc.2010.12.019
Allessie M, Ausma J, Schotten U (2002) Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc Res 54:230–246 (pii:S0008636302002584)
Ambros V (2004) The functions of animal microRNAs. Nature 431:350–355. doi:10.1038/nature02871
Antoniades C, Bakogiannis C, Leeson P, Guzik TJ, Zhang MH, Tousoulis D, Antonopoulos AS, Demosthenous M, Marinou K, Hale A, Paschalis A, Psarros C, Triantafyllou C, Bendall J, Casadei B, Stefanadis C, Channon KM (2011) Rapid, direct effects of statin treatment on arterial redox state and nitric oxide bioavailability in human atherosclerosis via tetrahydrobiopterin-mediated endothelial nitric oxide synthase coupling. Circulation 124:335–345. doi:10.1161/CIRCULATIONAHA.110.985150
Antoniades C, Bakogiannis C, Tousoulis D, Reilly S, Zhang MH, Paschalis A, Antonopoulos AS, Demosthenous M, Miliou A, Psarros C, Marinou K, Sfyras N, Economopoulos G, Casadei B, Channon KM, Stefanadis C (2010) Preoperative atorvastatin treatment in CABG patients rapidly improves vein graft redox state by inhibition of Rac1 and NADPH-oxidase activity. Circulation 122:S66–S73. doi:10.1161/CIRCULATIONAHA.109.927376
Barringhaus KG, Zamore PD (2009) MicroRNAs: regulating a change of heart. Circulation 119:2217–2224. doi:10.1161/CIRCULATIONAHA.107.715839
Boldt A, Scholl A, Garbade J, Resetar ME, Mohr FW, Gummert JF, Dhein S (2006) ACE-inhibitor treatment attenuates atrial structural remodeling in patients with lone chronic atrial fibrillation. Basic Res Cardiol 101:261–267. doi:10.1007/s00395-005-0571-2
Boldt A, Wetzel U, Lauschke J, Weigl J, Gummert J, Hindricks G, Kottkamp H, Dhein S (2004) Fibrosis in left atrial tissue of patients with atrial fibrillation with and without underlying mitral valve disease. Heart 90:400–405. doi:10.1136/hrt.2003.015347
Bonauer A, Carmona G, Iwasaki M, Mione M, Koyanagi M, Fischer A, Burchfield J, Fox H, Doebele C, Ohtani K, Chavakis E, Potente M, Tjwa M, Urbich C, Zeiher AM, Dimmeler S (2009) MicroRNA-92a controls angiogenesis and functional recovery of ischemic tissues in mice. Science 324:1710–1713. doi:10.1126/science.1174381
Boon RA, Seeger T, Heydt S, Fischer A, Hergenreider E, Horrevoets AJ, Vinciguerra M, Rosenthal N, Sciacca S, Pilato M, van Heijningen P, Essers J, Brandes RP, Zeiher AM, Dimmeler S (2011) MicroRNA-29 in aortic dilation: implications for aneurysm formation. Circ Res 109(10):1115–1119. doi:10.1161/CIRCRESAHA.111.255737
Casci T, Vinos J, Freeman M (1999) Sprouty, an intracellular inhibitor of Ras signaling. Cell 96:655–665 (pii:S0092-8674(00)80576-0)
Custodis F, Eberl M, Kilter H, Böhm M, Laufs U (2006) Association of RhoGDIalpha with Rac1 GTPase mediates free radical production during myocardial hypertrophy. Cardiovasc Res 71:342–351. doi:10.1016/j.cardiores.2006.04.005
Dobrev D, Carlsson L, Nattel S (2012) Novel molecular targets for atrial fibrillation therapy. Nat Rev Drug Discov 11:275–291. doi:10.1038/nrd3682
Dobrev D, Nattel S (2010) New antiarrhythmic drugs for treatment of atrial fibrillation. Lancet 375:1212–1223. doi:10.1016/S0140-6736(10)60096-7
Du T, Zamore PD (2005) microPrimer: the biogenesis and function of microRNA. Development 132:4645–4652. doi:10.1242/dev.02070
Fiedler J, Jazbutyte V, Kirchmaier BC, Gupta SK, Lorenzen J, Hartmann D, Galuppo P, Kneitz S, Pena JT, Sohn-Lee C, Loyer X, Soutschek J, Brand T, Tuschl T, Heineke J, Martin U, Schulte-Merker S, Ertl G, Engelhardt S, Bauersachs J, Thum T (2011) MicroRNA-24 regulates vascularity after myocardial infarction. Circulation 124:720730. doi:10.1161/CIRCULATIONAHA.111.039008
Ghildiyal M, Zamore PD (2009) Small silencing RNAs: an expanding universe. Nat Rev Genet 10:94–108. doi:10.1038/nrg2504
Girmatsion Z, Biliczki P, Bonauer A, Wimmer-Greinecker G, Scherer M, Moritz A, Bukowska A, Goette A, Nattel S, Hohnloser SH, Ehrlich JR (2009) Changes in microRNA-1 expression and IK1 up-regulation in human atrial fibrillation. Heart Rhythm 6:1802–1809. doi:10.1016/j.hrthm.2009.08.035
Goette A, Arndt M, Rocken C, Spiess A, Staack T, Geller JC, Huth C, Ansorge S, Klein HU, Lendeckel U (2000) Regulation of angiotensin II receptor subtypes during atrial fibrillation in humans 96. Circulation 101:2678–2681. doi:10.1161/01.CIR.101.23.2678
Hacohen N, Kramer S, Sutherland D, Hiromi Y, Krasnow MA (1998) Sprouty encodes a novel antagonist of FGF signaling that patterns apical branching of the Drosophila airways. Cell 92:253–263 (S0092-8674(00)80919-8)
Hanafusa H, Torii S, Yasunaga T, Nishida E (2002) Sprouty1 and Sprouty2 provide a control mechanism for the Ras/MAPK signalling pathway. Nat Cell Biol 4:850–858. doi:10.1038/ncb867
Hasin T, Elhanani O, Abassi Z, Hai T, Aronheim A (2011) Angiotensin II signaling up-regulates the immediate early transcription factor ATF3 in the left but not the right atrium. Basic Res Cardiol 106:175–187. doi:10.1007/s00395-010-0145-9
Hornstra IK, Birge S, Starcher B, Bailey AJ, Mecham RP, Shapiro SD (2003) Lysyl oxidase is required for vascular and diaphragmatic development in mice. J Biol Chem 278:14387–14393. doi:10.1074/jbc.M210144200M210144200
Ihm SH, Chang K, Kim HY, Baek SH, Youn HJ, Seung KB, Kim JH (2010) Peroxisome proliferator-activated receptor-gamma activation attenuates cardiac fibrosis in type 2 diabetic rats: the effect of rosiglitazone on myocardial expression of receptor for advanced glycation end products and of connective tissue growth factor. Basic Res Cardiol 105:399–407. doi:10.1007/s00395-009-0071-x
Kagan HM, Li W (2003) Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem 88:660–672. doi:10.1002/jcb.10413
Kawano H, Do YS, Kawano Y, Starnes V, Barr M, Law RE, Hsueh WA (2000) Angiotensin II has multiple profibrotic effects in human cardiac fibroblasts 39. Circulation 101:1130–1137. doi:10.1161/01.CIR.101.10.1130
Krützfeldt J, Rajewsky N, Braich R, Rajeev KG, Tuschl T, Manoharan M, Stoffel M (2005) Silencing of microRNAs in vivo with ‘antagomirs’. Nature 438:685–689. doi:10.1038/nature04303
Kuehbacher A, Urbich C, Zeiher AM, Dimmeler S (2007) Role of Dicer and Drosha for endothelial microRNA expression and angiogenesis. Circ Res 101:59–68. doi:10.1161/CIRCRESAHA.107.153916
Landthaler M, Yalcin A, Tuschl T (2004) The human DiGeorge syndrome critical region gene 8 and its D. melanogaster homolog are required for miRNA biogenesis. Curr Biol 14:2162–2167. doi:10.1016/j.cub.2004.11.001
Laufs U, Kilter H, Konkol C, Wassmann S, Böhm M, Nickenig G (2002) Impact of HMG CoA reductase inhibition on small GTPases in the heart. Cardiovasc Res 53:911–920. doi:10.1016/S0008-6363(01)00540-5
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75:843–854 (pii:0092-8674(93)90529-Y)
Li D, Fareh S, Leung TK, Nattel S (1999) Promotion of atrial fibrillation by heart failure in dogs: atrial remodeling of a different sort 1. Circulation 100:87–95. doi:10.1161/01.CIR.100.1.87
Li D, Shinagawa K, Pang L, Leung TK, Cardin S, Wang Z, Nattel S (2001) Effects of angiotensin-converting enzyme inhibition on the development of the atrial fibrillation substrate in dogs with ventricular tachypacing-induced congestive heart failure. Circulation 104:2608–2614. doi:10.1161/hc4601.099402
Li W, Nellaiappan K, Strassmaier T, Graham L, Thomas KM, Kagan HM (1997) Localization and activity of lysyl oxidase within nuclei of fibrogenic cells. Proc Natl Acad Sci USA 94:12817–12822. doi:10.1073/pnas.94.24.12817
Liao JK, Laufs U (2005) Pleiotropic effects of statins. Annu Rev Pharmacol Toxicol 45:89–118. doi:10.1146/annurev.pharmtox.45.120403.095748
Lopez B, Gonzalez A, Hermida N, Valencia F, de Teresa E, Diez J (2010) Role of lysyl oxidase in myocardial fibrosis. From basic science to clinical aspects. Am J Physiol Heart Circ Physiol 299:H1–H9. doi:10.1152/ajpheart.00335.2010
Lopez B, Querejeta R, Gonzalez A, Beaumont J, Larman M, Diez J (2009) Impact of treatment on myocardial lysyl oxidase expression and collagen cross-linking in patients with heart failure. Hypertension 53:236–242. doi:10.1161/HYPERTENSIONAHA.108.125278
Lu Y, Zhang Y, Wang N, Pan Z, Gao X, Zhang F, Shan H, Luo X, Bai Y, Sun L, Song W, Xu C, Wang Z, Yang B (2010) MicroRNA-328 contributes to adverse electrical remodeling in atrial fibrillation. Circulation 122:2378–2387. doi:10.1161/CIRCULATIONAHA.110.958967
Lugenbiel P, Thomas D, Kelemen K, Trappe K, Bikou O, Schweizer PA, Voss F, Becker R, Katus HA, Bauer A (2012) Genetic suppression of Gα(s) protein provides rate control in atrial fibrillation. Basic Res Cardiol 107:1–12. doi:10.1007/s00395-012-0265-5
Maki JM, Rasanen J, Tikkanen H, Sormunen R, Makikallio K, Kivirikko KI, Soininen R (2002) Inactivation of the lysyl oxidase gene Lox leads to aortic aneurysms, cardiovascular dysfunction, and perinatal death in mice. Circulation 106:2503–2509. doi:10.1161/01.CIR.0000038109.84500.1E
Matranga C, Zamore PD (2007) Small silencing RNAs. Curr Biol 17:R789–R793. doi:10.1016/j.cub.2007.07.014
Michel MC, Li Y, Heusch G (2001) Mitogen-activated protein kinases in the heart. Naunyn Schmiedebergs Arch Pharmacol 363:245–266. doi:10.1007/s002100000363
Miller CL, Cai Y, Oikawa M, Thomas T, Dostmann WR, Zaccolo M, Fujiwara K, Yan C (2011) Cyclic nucleotide phosphodiesterase 1A: a key regulator of cardiac fibroblast activation and extracellular matrix remodeling in the heart. Basic Res Cardiol 106:1023–1039. doi:10.1007/s00395-011-0228-2
Molnar J, Fong KS, He QP, Hayashi K, Kim Y, Fong SF, Fogelgren B, Szauter KM, Mink M, Csiszar K (2003) Structural and functional diversity of lysyl oxidase and the LOX-like proteins. Biochim Biophys Acta 1647:220–224 (pii:S1570963903000530)
Nattel S (2002) New ideas about atrial fibrillation 50 years on. Nature 415:219–226. doi:10.1038/415219a
Nattel S, Burstein B, Dobrev D (2008) Atrial remodeling and atrial fibrillation: mechanisms and implications. Circ Arrhythm Electrophysiol 1:62–73. doi:10.1161/CIRCEP.107.754564
Ohtani K, Dimmeler S (2011) Control of cardiovascular differentiation by microRNAs. Basic Res Cardiol 106:5–11. doi:10.1007/s00395-010-0139-7
Reil JC, Hohl M, Oberhofer M, Kazakov A, Kaestner L, Mueller P, Adam O, Maack C, Lipp P, Mewis C, Allessie M, Laufs U, Böhm M, Neuberger HR (2010) Cardiac Rac1 overexpression in mice creates a substrate for atrial arrhythmias characterized by structural remodelling. Cardiovasc Res 87:485–493. doi:10.1093/cvr/cvq079
Reilly SN, Jayaram R, Nahar K, Antoniades C, Verheule S, Channon KM, Alp NJ, Schotten U, Casadei B (2011) Atrial sources of reactive oxygen species vary with the duration and substrate of atrial fibrillation: implications for the antiarrhythmic effect of statins. Circulation 124:1107–1117. doi:10.1161/CIRCULATIONAHA.111.029223
Ruperez M, Lorenzo O, Blanco-Colio LM, Esteban V, Egido J, Ruiz-Ortega M (2003) Connective tissue growth factor is a mediator of angiotensin II-induced fibrosis. Circulation 108:1499–1505. doi:10.1161/01.CIR.0000089129.51288.BA
Saad FA, Torres M, Wang H, Graham L (2010) Intracellular lysyl oxidase: effect of a specific inhibitor on nuclear mass in proliferating cells. Biochem Biophys Res Commun 396:944–949. doi:10.1016/j.bbrc.2010.05.028
Sivakumar P, Gupta S, Sarkar S, Sen S (2008) Upregulation of lysyl oxidase and MMPs during cardiac remodeling in human dilated cardiomyopathy. Mol Cell Biochem 307:159–167. doi:10.1007/s11010-007-9595-2
Smith-Mungo LI, Kagan HM (1998) Lysyl oxidase: properties, regulation and multiple functions in biology. Matrix Biol 16:387–398 (pii:S0945-053X(98)90012-9)
Suenari K, Chen YC, Kao YH, Cheng CC, Lin YK, Chen YJ, Chen SA (2011) Discrepant electrophysiological characteristics and calcium homeostasis of left atrial anterior and posterior myocytes. Basic Res Cardiol 106:65–74. doi:10.1007/s00395-010-0132-1
Sussman MA, Welch S, Walker A, Klevitsky R, Hewett TE, Price RL, Schaefer E, Yager K (2000) Altered focal adhesion regulation correlates with cardiomyopathy in mice expressing constitutively active rac1. J Clin Invest 105:875–886. doi:10.1172/JCI8497
Takemoto M, Node K, Nakagami H, Liao Y, Grimm M, Takemoto Y, Kitakaze M, Liao JK (2001) Statins as antioxidant therapy for preventing cardiac myocyte hypertrophy. J Clin Invest 108(10):1429–1437. doi:10.1172/JCI200113350
Thum T, Catalucci D, Bauersachs J (2008) MicroRNAs: novel regulators in cardiac development and disease. Cardiovasc Res 79:562–570. doi:10.1093/cvr/cvn137
Thum T, Gross C, Fiedler J, Fischer T, Kissler S, Bussen M, Galuppo P, Just S, Rottbauer W, Frantz S, Castoldi M, Soutschek J, Koteliansky V, Rosenwald A, Basson MA, Licht JD, Pena JT, Rouhanifard SH, Muckenthaler MU, Tuschl T, Martin GR, Bauersachs J, Engelhardt S (2008) MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature 456:980–984. doi:10.1038/nature07511
Tsai CF, Chen YC, Lin YK, Chen SA, Chen YJ (2011) Electromechanical effects of the direct renin inhibitor (aliskiren) on the pulmonary vein and atrium. Basic Res Cardiol 106:979–993. doi:10.1007/s00395-011-0206-8
Wakili R, Voigt N, Kaab S, Dobrev D, Nattel S (2011) Recent advances in the molecular pathophysiology of atrial fibrillation. J Clin Invest 121:2955–2968. doi:10.1172/JCI46315
Wang Z, Lu Y, Yang B (2011) MicroRNAs and atrial fibrillation: new fundamentals. Cardiovasc Res 89:710–721. doi:10.1093/cvr/cvq350
Wenzel S, Henning K, Habbig A, Forst S, Schreckenberg R, Heger J, Maxeiner H, Schluter KD (2010) TGF-β1 improves cardiac performance via up-regulation of laminin receptor 37/67 in adult ventricular cardiomyocytes. Basic Res Cardiol 105:621–629. doi:10.1007/s00395-010-0108-1
Yang B, Lu Y, Wang Z (2008) Control of cardiac excitability by microRNAs. Cardiovasc Res 79:571–580. doi:10.1093/cvr/cvn181
Yang SS, Han W, Cao Y, Dong G, Zhou G, Li WM, Gan RT, Chang HY, Wang Z (2011) Effects of high thoracic epidural anesthesia on atrial electrophysiological characteristics and sympathetic nerve sprouting in a canine model of atrial fibrillation. Basic Res Cardiol 106:495–506. doi:10.1007/s00395-011-0154-3
Acknowledgments
We thank Simone Jäger and Ellen Becker for their excellent technical assistance. Sources of funding: this study was supported by the Deutsche Forschungsgemeinschaft (KFO 196), the Universität des Saarlandes, the European Stroke Network (ESN) and the Ministerium für Wirtschaft und Wissenschaft des Saarlandes.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
395_2012_278_MOESM1_ESM.doc
Supplement Fig. 1 Correlation between miR-21 expression (2−ΔΔct) in left atrial myocardium of patients with atrial fibrillation (AF); n = 8 per; r = 0.9728, p < 0.0001 (DOC 256 kb)
395_2012_278_MOESM2_ESM.doc
Supplement Fig. 2 Quantification of myocyte size in the atria of mice after myocardial infarction induced by LAD occlusion with (MI + Antag-21) and without antagomir-21 treatment (MI) compared to Sham; n = 4-5 per group, *p < 0.05 vs. Sham, #p < 0.05 vs MI. (DOC 117 kb)
395_2012_278_MOESM3_ESM.doc
Supplement Table 1 Patient characteristics ACE-I = angiotensin-converting enzyme inhibitor; AT antagonist = angiotensin receptor I antagonist; FS = fractional shortening; IVSd = interventricular septum; LA = left atrium; LVEDd = left ventricular end-diastolic diameter; LVEF = left ventricular ejection fraction; LVESd = left ventricular end-systolic diameter. (DOC 77 kb)
Rights and permissions
About this article
Cite this article
Adam, O., Löhfelm, B., Thum, T. et al. Role of miR-21 in the pathogenesis of atrial fibrosis. Basic Res Cardiol 107, 278 (2012). https://doi.org/10.1007/s00395-012-0278-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-012-0278-0