Abstract
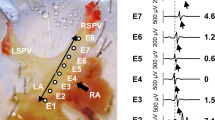
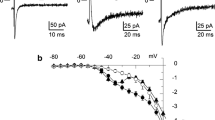
Activation of the atrial renin–angiotensin system plays an important role in the pathophysiology of atrial fibrillation (AF). The pulmonary vein (PV) and left atrium (LA) are important trigger and substrate for the genesis of AF. We investigate the effects of a direct renin inhibitor, aliskiren, on the PV and LA arrhythmogenic activity and the underlying electromechanical mechanisms. Conventional microelectrodes were used to record action potentials and contractility in isolated rabbit PVs and LA tissues before and after the administration of aliskiren (0.1, 1, 3 and 10 μM). By the whole-cell patch clamp and indo-1 fluorimetric ratio techniques, ionic currents and intracellular calcium transient were studied in isolated single PV and LA cardiomyocyte before and after the administration of aliskiren (3 μM). Aliskiren (0.1, 1, 3 and 10 μM) reduced PV firing rate in a concentration-dependent manner (6, 10, 14 and 17%) and decreased PV diastolic tension, which could be attenuated in the presence of 100 μM L-NG-Nitroarginine Methyl Ester (L-NAME). Aliskiren induced PV automatic rhythm exit block causing slow and irregular PV activity with variable pauses. Aliskiren increased PV and LA contractility, which could be abolished by pre-treating with 0.1 μM ryanodine. Aliskiren (3 μM) decreased L-type calcium currents, but increased reverse-mode of Na+/Ca2+ exchanger currents, intracellular calcium transients, and sarcoplasmic reticulum calcium content in PV and LA cardiomyocytes. Pretreatment with renin, losartan or angiotensin II did not alter the effect of aliskiren on sarcolemmal calcium flux. In conclusion, aliskiren reduces PV arrhythmogenic activity with a direct vasodilatory property and has a positive inotropic effect on cardiomyocytes. These findings may reveal the anti-arrhythmic and anti-heart failure potentials of aliskiren.









Similar content being viewed by others
References
Arora R, Verheule S, Scott L, Navarrete A, Katari V, Wilson E, Vez D, Olgin JE (2003) Arrhythmogenic substrate of the pulmonary veins assessed by high-resolution optical mapping. Circulation 107:1816–1821. doi:10.1161/01.CIR.0000058461.86339.7E
Boyett MR, Inada S, Yoo S, Li J, Liu J, Tellez JO, Greener ID, Honjo H, Billeter R, Lei M, Zhang H, Efimov IR, Dobrzynski H (2006) Connexins in the sinoatrial and atrioventricular nodes. Adv Cardiol 42:175–197. doi:10.1159/000092569
Brown MJ (2008) Aliskiren. Circulation 118:773–784. doi:10.1161/CIRCULATIONAHA.108.787630
Chang SL, Chen YC, Chen YJ, Wangcharoen W, Lee SH, Lin CI, Chen SA (2007) Mechanoelectrical feedback regulates the arrhythmogenic activity of pulmonary veins. Heart 93:82–88. doi:10.1136/hrt.2006.089359
Chang SL, Tai CT, Lin YJ, Wongcharoen W, Lo LW, Lee KT, Chang SH, Tuan TC, Chen YJ, Hsieh MH, Tsao HM, Wu MH, Sheu MH, Chang CY, Chen SA (2007) The role of left atrial muscular bundles in catheter ablation of atrial fibrillation. J Am Coll Cardiol 50:964–973. doi:10.1016/j.jacc.2007.05.026
Chen SA, Hsieh MH, Tai CT, Tsai CF, Prakash VS, Yu WC, Hsu TL, Ding YA, Chang MS (1999) Initiation of atrial fibrillation by ectopic beats originating from the pulmonary veins: electrophysiological characteristics, pharmacological responses, and effects of radiofrequency ablation. Circulation 100:1879–1886. doi:10.1161/01.CIR.100.18.1879
Chen WJ, Yeh YH, Lin KH, Chang GJ, Kuo CT (2011) Molecular characterization of thyroid hormone-inhibited atrial L-type calcium channel expression: implication for atrial fibrillation in hyperthyroidism. Basic Res Cardiol 106:163–174. doi:10.1007/s00395-010-0149-5
Chen YC, Kao YH, Huang CF, Cheng CC, Chen YJ, Chen SA (2010) Heat stress responses modulate calcium regulations and electrophysiological characteristics in atrial myocytes. J Mol Cell Cardiol 48:781–788. doi:10.1016/j.yjmcc.2009.08.006
Chen YJ, Chen SA, Chang MS, Lin CI (2000) Arrhythmogenic activity of cardiac muscle in pulmonary vein of the dog: implication for the genesis of atrial fibrillation. Cardiovasc Res 48:265–273. doi:10.1016/S0008-6363(00)00179-6
Chen YJ, Chen SA, Chen YC, Yeh HI, Chang MS, Lin CI (2002) Electrophysiology of single cardiomyocytes isolated from rabbit pulmonary veins: implication in initiation of focal atrial fibrillation. Basic Res Cardiol 97:26–34. doi:10.1007/s395-002-8384-6
Chen YJ, Chen YC, Tai CT, Yes HI, Lin CI, Chen SA (2006) Angiotensin II and angiotensin II receptor blocker modulate the arrhythmogenic activity of pulmonary veins. Br J Pharmacol 147:12–22. doi:10.1038/sj.bjp.0706445
Chen YJ, Chen YC, Wongcharoen W, Lin CI, Chen SA (2008) K201, a novel antiarrhythmic drug on calcium handling and arrhythmogenic activity of pulmonary vein cardiomyocytes. Br J Pharmacol 153:915–925. doi:10.1038/sj.bjp.0707564
Cherney DZI, Lai V, Scholey JW, Miller JA, Zinman B, Reich HN (2010) Effect of direct renin inhibition on renal hemodynamic function, arterial stiffness, and endothelial function in humans with uncomplicated type I diabetes. Diabetes Care 33:361–365. doi:10.2337/dc09-1303
Chou CC, Nihei M, Zhou S, Tan A, Kawase A, Macias ES, Fishbein MC, Lin SF, Chen PS (2005) Intracellular calcium dynamics and anisotropic reentry in isolated canine pulmonary veins and left atrium. Circulation 111:2889–2897. doi:10.1161/CIRCULATIONAHA.104.498758
De Mello WC (2006) Renin increments the inward calcium current in the failing heart. J Hypertens 24:1181–1186. doi:10.1097/01.hjh.0000226209.88312.db
Ehrlich JR, Hohnloser SH, Nattel S (2006) Role of angiotensin system and effects of its inhibition in atrial fibrillation: clinical and experimental evidence. Eur Heart J 27:512–518. doi:10.1093/eurheartj/ehi668
Fedorov VV, Schuessler RB, Hemphill M, Ambrosi CM, Chang R, Voloshina AS, Brown K, Hucker WJ, Efimov IR (2009) Structural and functional evidence for discrete exit pathways that connect the canine sinoatrial node and atria. Circ Res 104:915–923. doi:10.1161/CIRCRESAHA.108.193193
Feldt S, Batenburg WW, Mazak I, Maschke U, Wellner M, Kvakan H, Dechend R, Fiebeler A, Burckle C, Contrepas A, Danser AHJ, Bader M, Nguyen G, Luft FC, Muller DN (2008) Prorenin and renin-induced extracellular signal-regulated kinase 1/2 activation in monocytes is not blocked by aliskiren or the handle-region peptide. Hypertension 51:682–688. doi:10.1161/HYPERTENSIONAHA.107.101444
Fischer R, Dechend R, Qadri F, Markovic M, Feldt S, Herse F, Park JK, Gapelyuk A, Schwarz I, Zacharzowsky UB, Plehm R, Safak E, Heuser A, Schirdewan A, Luft FC, Schunck WH, Muller DN (2008) Dietary n-3 polyunsaturated fatty acids and direct renin inhibition improve electrical remodeling in a model of high human renin hypertension. Hypertension 51:540–546. doi:10.1161/HYPERTENSIONAHA.107.103143
Goette A, Arndt M, Rocken C (2000) Regulation of angiotensin II receptor subtypes during atrial fibrillation in humans. Circulation 101:2678–2681. doi:10.1161/01.CIR.101.23.2678
Hasin T, Elhanani O, Abassi Z, Hai T, Aronheim A (2011) Angiotensin II signaling up-regulates the immediate early transcription factor ATF3 in the left but not the right atrium. Basic Res Cardiol 106:175–187. doi:10.1007/s00395-010-0145-9
Hu YF, Chen YC, Cheng CC, Higa S, Chen YJ, Chen SA (2009) Fluvastatin reduces pulmonary vein spontaneous activity through nitric oxide pathway. J Cardiovasc Electrophysiol 20:200–206. doi:10.1111/j.1540-8167.2008.01281.x
Imanishi T, Tsujioka H, Ikejima H, Kuroi A, Takarada S, Kitabata H, Tanimoto T, Muragaki Y, Mochizuki S, Goto M, Yoshida K, Akasaka T (2008) Renin inhibitor aliskiren improves impaired nitric oxide bioavailability and protects against atherosclerotic changes. Hypertension 52:563–572. doi:10.1161/HYPERTENSIONAHA.108.111120
Kalifa J, Jalife J, Zaitsev AV, Bagwe S, Linnenbank AC, Potse M, Shah D, Jais P, Janse MJ, Haissaguerre M, de Bakker JMT, Warren M, Moreno J, Berenfeld O, Nattel S (2003) Intra-atrial pressure increases rate and organization of waves emanating from the superior pulmonary veins during atrial fibrillation. Circulation 108:668–671. doi:10.1161/01.CIR.0000086979.39843.7B
Kang M, Walker JW (2006) Endothelin-I and PKC induce positive inotropy without affecting PHi in ventricular myocytes. Exp Biol Med 231:865–870
Kohl P, Kamkin AG, Kiseleva IS, Noble D (1994) Mechanosensitive fibroblasts in the sino-atrial node region of rat heart: interaction with cardiomyocytes and possible role. Exp Physiol 79:943–956
Larbig R, Torres N, Bridge JHB, Goldhaber JI, Philipson KD (2010) Activation of reverse Na+–Ca2+ exchange by the Na+ current augments the cardiac Ca2+ transient: evidence from NCX knockout mice. J Physiol 588:3267–3276. doi:10.1113/jphysiol.2010.187708
Litwin SE, Li J, Bridge JH (1998) Na+–Ca2+ exchange and the trigger for sarcoplasmic reticulum Ca release: studies in adult rabbit ventricular myocytes. Biophys J 75:359–371
Masani F (1986) Node-like cells in the myocardial layer of the pulmonary vein of rats: an ultrastructural study. J Anat 145:133–142
McMurray JJ, Pitt B, Latini R, Maggioni AP, Solomon SD, Keefe DL, Ford J, Verma A, Lewsey J; Aliskiren Observation of Heart Failure Treatment (ALOFT) Investigators (2008) Effects of the oral direct renin inhibitor aliskiren in patients with symptomatic heart failure. Circ Heart Fail 1:17–24. doi:10.1161/CIRCHEARTFAILURE.107.740704
Pappone C, Rosanio S, Oreto G, Tocchi M, Gugliotta F, Vicedomini G, Salvati A, Dicandia C, Mazzone P, Santinelli V, Gulletta S, Chierchia S (2000) Circumferential radiofrequency ablation of pulmonary vein ostia: a new anatomic approach for curing atrial fibrillation. Circulation 102:2619–2628. doi:10.1161/01.CIR.102.21.2619
Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK, for the AVOID Study Investigators (2008) Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 358:2433–2446. doi:10.1056/NEJMoa0708379
Saito T, Waki K, Becker A (2000) Left myocardial extension onto pulmonary veins in humans: anatomic observations relevant for atrial arrhythmias. J Cardiovasc Electrophysiol 11:888–894. doi:10.1111/j.1540-8167.2000.tb00068.x
Saris JJ, ‘t Hoen PA, Garrelds IM, Dekkers DH, den Dunnen JT, Lamers JM, Danser AHJ (2006) Prorenin induces intracellular signaling in cardiomyocytes independently of angiotensin II. Hypertension 48:564–571. doi:10.1161/01.HYP.0000240064.19301.1b
Saygili E, Rana OR, Meyer C, Gemein C, Andrzejewski MG, Ludwig A, Weber C, Schotten U, Kruttgen A, Weis J, Schwinger RHG, Mischke K, Rassaf T, Kelm M, Schauerte P (2009) The angiotensin–calcineurin–NFAT pathway mediates stretch-induced up-regulation of matrix metalloproteinases-2/-9 in atrial myocytes. Basic Res Cardiol 104:435–448. doi:10.1007/s00395-008-0772-6
Schneider MP, Hua TA, Bohm M, Wachtell K, Kjeldsen SE, Schmieder RE (2010) Prevention of atrial fibrillation by renin–angiotensin system inhibition: a meta-analysis. J Am Coll Cardiol 55:2299–2307. doi:10.1016/j.jacc.2010.01.043
Sobie EA, Cannell MB, Bridge JH (2008) Allosteric activation of Na+–Ca2+ exchange by L-type Ca2+ current augments the trigger flux for SR Ca2+ release in ventricular myocytes. Biophys J 94:54–56. doi:10.1529/biophysj.107.127878
Solomon SD, Appelbaum E, Manning WJ, Verma A, Berglund T, Lukashevich V, Cherif Papst C, Smith BA, Dahlof B (2009) Effect of the direct renin inhibitor aliskiren, the angiotensin receptor blocker losartan, or both on left ventricular mass in patients with hypertension and left ventricular hypertrophy. Circulation 119:530–537. doi:10.1161/CIRCULATIONAHA.108.826214
Suenari K, Chen YC, Kao YH, Cheng CC, Lin YK, Chen YJ, Chen SA (2010) Discrepant electrophysiological characteristics and calcium homeostasis of left atrial anterior and posterior myocytes. Basic Res Cardiol 106:65–74. doi:10.1007/s00395-010-0132-1
Terracciano CMN, De Souza AI, Phillipson KD, MacLeod KT (1998) Na+–Ca2+ exchange and sarcoplasmic reticular Ca2+ regulation in ventricular myocytes from transgenic mice overexpressing the Na+–Ca2+ exchanger. J Physiol 512:651–667. doi:10.1111/j.1469-7793.1998.651bd.x
The GISSI-AF Investigators (2009) Valsartan for prevention of recurrent atrial fibrillation. N Engl J Med 360:1606–1617. doi:10.1056/NEJMoa0805710
Verheule S, Wilson EE, Arora R, Engle SK, Scott LR, Olgin JE (2002) Tissue structure and connexin expression of canine pulmonary veins. Cardiovasc Res 55:727–738. doi:10.1016/S0008-6363(02)00490-X
Wongcharoen W, Chen YC, Chen YJ, Chang CM, Yeh HI, Lin CI, Chen SA (2006) Effects of a Na+/Ca2+ exchanger inhibitor on pulmonary vein electrical activity and ouabain-induced arrhythmogenicity. Cardiovasc Res 70:497–508. doi:10.1016/j.cardiores.2006.02.026
Zima AV, Blatter LA (2004) Inositol-1,4,5-trisphosphate-dependent Ca2+ signaling in cat atrial excitation–contraction coupling and arrhythmias. J Physiol 555:607–615. doi:10.1113/jphysiol.2003.058529
Acknowledgments
This work was supported by the Center of Excellence for Clinical Trial and Research in Neuroscience (DOH100-TD-B-111-003) and grants from Chung Shan Medical University (CSMU-99-OM-A-086), Wan Fang Hospital, Taipei Medical University (100-wf-eva-01) and the National Science Council (NSC 97-2314-B-038-030-MY3, NSC 99-2314-B-016-034-MY3, NSC 99-2628-B-038-011-MY3, and NSC-99-2314-B-040-017), Taiwan, ROC.
Conflict of interest
None to be declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsai, CF., Chen, YC., Lin, YK. et al. Electromechanical effects of the direct renin inhibitor (aliskiren) on the pulmonary vein and atrium. Basic Res Cardiol 106, 979–993 (2011). https://doi.org/10.1007/s00395-011-0206-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-011-0206-8