Abstract
Objectives
To investigate whether CT slice thickness influences the performance of radiomics prognostic models in non-small-cell lung cancer (NSCLC) patients.
Methods
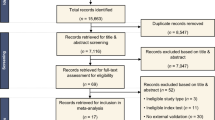
CT images including 1-, 3-, and 5-mm slice thicknesses acquired from 311 patients who underwent surgical resection for NSCLC between May 2014 and December 2015 were evaluated. Tumor segmentation was performed on CT of each slice thickness and total 94 radiomics features (shape, tumor intensity, and texture) were extracted. The study population was temporally split into development (n = 185) and validation sets (n = 126) for prediction of disease-free survival (DFS). Three radiomics models were built from three different slice thickness datasets (Rad-1, Rad-3, and Rad-5), respectively. Model performance was assessed and compared in three slice thickness datasets and mixed slice thickness dataset using C-indices.
Results
In corresponding slice thickness datasets, the C-indices of Rad-1, Rad-3, and Rad-5 for prediction of DFS were 0.68, 0.70, and 0.68 in the development set, and 0.73, 0.73, and 0.76 in the validation set (p = 0.40–0.89 and 0.27–0.90, respectively). Performance of the models was not significantly changed when they were applied to different slice thicknesses data in the validation set (C-index, 0.73–0.76, 0.72–0.73, 0.75–0.76; p = 0.07–0.92). In the mixed slice thickness dataset, performances of the models were similar to or slightly lower than their performances in the corresponding slice thickness datasets (C-index, 0.72–0.75 vs. 0.73–0.76) in the validation set.
Conclusions
The performance of radiomics models for predicting DFS in NSCLC patients was not significantly affected by CT slice thickness.
Key Points
• Three radiomics models based on 1-, 3-, and 5-mm CT datasets showed C-indices for predicting disease-free survival of 0.68–0.70 in the development set and 0.73–0.76 in the validation set, without statistical differences (p = 0.27–0.90).
• Application of the radiomics models to different slice thickness datasets showed no significant differences in performance between the values in the prediction of disease-free survival (p = 0.07–0.99).
• Three radiomics models based on 1-, 3-, and 5-mm CT datasets performed well in mixed slice thickness datasets, showing similar or slightly lower performances.



Similar content being viewed by others
Abbreviations
- CCC:
-
Concordance correlation coefficient
- CI:
-
Confidence interval
- C-index:
-
Concordance index
- DFS:
-
Disease-free survival
- NSCLC:
-
Non-small-cell lung cancer
- TNM:
-
Tumor-node-metastasis
References
Pao W, Girard N (2011) New driver mutations in non-small-cell lung cancer. Lancet Oncol 12:175–180
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Aerts HJ, Velazquez ER, Leijenaar RT et al (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006
Huang Y, Liu Z, He L et al (2016) Radiomics signature: a potential biomarker for the prediction of disease-free survival in early-stage (I or II) non-small cell lung cancer. Radiology 281:947–957
Berenguer R, Pastor-Juan MDR, Canales-Vazquez J et al (2018) Radiomics of CT features may be nonreproducible and redundant: influence of CT acquisition parameters. Radiology 288:407–415
Mackin D, Fave X, Zhang L et al (2015) Measuring computed tomography scanner variability of radiomics features. Invest Radiol 50:757–765
He L, Huang Y, Ma Z, Liang C, Liang C, Liu Z (2016) Effects of contrast-enhancement, reconstruction slice thickness and convolution kernel on the diagnostic performance of radiomics signature in solitary pulmonary nodule. Sci Rep 6:34921
Park S, Lee SM, Do KH et al (2019) Deep learning algorithm for reducing CT slice thickness: effect on reproducibility of radiomic features in lung cancer. Korean J Radiol 20:1431–1440
Choe J, Lee SM, Do KH et al (2019) Deep learning-based image conversion of CT reconstruction kernels improves radiomics reproducibility for pulmonary nodules or masses. Radiology 292:365–373
Bankier AA, MacMahon H, Goo JM, Rubin GD, Schaefer-Prokop CM, Naidich DP (2017) Recommendations for measuring pulmonary nodules at CT: a statement from the Fleischner Society. Radiology 285:584–600
Yoshizawa A, Sumiyoshi S, Sonobe M et al (2013) Validation of the IASLC/ATS/ERS lung adenocarcinoma classification for prognosis and association with EGFR and KRAS gene mutations: analysis of 440 Japanese patients. J Thorac Oncol 8:52–61
Choe J, Lee SM, Do KH et al (2020) Outcome prediction in resectable lung adenocarcinoma patients: value of CT radiomics. Eur Radiol. https://doi.org/10.1007/s00330-020-06872-z
Parmar C, Rios Velazquez E, Leijenaar R et al (2014) Robust Radiomics feature quantification using semiautomatic volumetric segmentation. PLoS One 9:e102107
Larue R, Van De Voorde L, van Timmeren JE et al (2017) 4DCT imaging to assess radiomics feature stability: an investigation for thoracic cancers. Radiother Oncol 125:147–153
Bogowicz M, Leijenaar RTH, Tanadini-Lang S et al (2017) Post-radiochemotherapy PET radiomics in head and neck cancer - the influence of radiomics implementation on the reproducibility of local control tumor models. Radiother Oncol 125:385–391
Lin LI (1989) A concordance correlation coefficient to evaluate reproducibility. Biometrics 45:255–268
Mukaka MM (2012) Statistics corner: a guide to appropriate use of correlation coefficient in medical research. Malawi Med J 24:69–71
Bender R, Lange S (2001) Adjusting for multiple testing--when and how? J Clin Epidemiol 54:343–349
Orlhac F, Frouin F, Nioche C, Ayache N, Buvat I (2019) Validation of a method to compensate multicenter effects affecting CT radiomics. Radiology 291:53–59
Zwanenburg A, Vallieres M, Abdalah MA et al (2020) The image biomarker standardization initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295:328–338
Funding
This study received funding from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (grant number: NRF-2019R1A2C1087524).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Sang Min Lee.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
The biostatistician Seonok Kim who is one of the authors of our study provided statistical advice for this manuscript.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Among the final study population of 311 patients, 214 patients were identical to the study population of our previous report (reference 12, outcome prediction in resectable lung adenocarcinoma patients: value of CT radiomics. Eur Radiol. 2020 https://doi.org/10.1007/s00330-020-06872-z).
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 62 kb)
Rights and permissions
About this article
Cite this article
Park, S., Lee, S.M., Kim, S. et al. Performance of radiomics models for survival prediction in non-small-cell lung cancer: influence of CT slice thickness. Eur Radiol 31, 2856–2865 (2021). https://doi.org/10.1007/s00330-020-07423-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07423-2