Abstract
Non-contrast cardiac computed tomography in order to obtain the coronary artery calcium score has become an established diagnostic procedure in the clinical setting, and is commonly employed in clinical and population-based research. This state-of-the-art review paper highlights the potential gain in information that can be obtained from the non-contrast coronary calcium scans without any necessary modifications to the scan protocol. This includes markers of cardio-metabolic health, such as the amount of epicardial fat and liver fat, but also markers of general health including bone density and lung density. Finally, this paper addresses the importance of incidental findings and of radiation exposure accompanying imaging with non-contrast cardiac computed tomography. Despite the fact that coronary calcium scan protocols have been optimized for the visualization of coronary calcification in terms image quality and radiation exposure, it is important for radiologists, cardiologists and medical specialists in the field of preventive medicine to acknowledge that numerous additional markers of cardio-metabolic health and general health can be readily identified on a coronary calcium scan.
Key Points
• The coronary artery calcium score substantially increased the use of cardiac CT.
• Cardio-metabolic and general health markers may be derived without changes to the scan protocol.
• Those include epicardial fat, aortic valve calcifications, liver fat, bone density, and lung density.
• Clinicians must be aware of this potential additional yield from non-contrast cardiac CT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past decade, non-contrast cardiac computed tomography (CT) has become an established diagnostic tool in clinical practice. The main purpose of these coronary calcium scans is to obtain the coronary artery calcium score (CACS) [1, 2], which is associated with a graded increased risk of future coronary events, heart failure and mortality [3,4,5], and even relates to dementia, cancer and kidney disease [6, 7]. On the other hand, a negative or zero CACS denotes a mid- to long-term risk of coronary events that is close to zero [8, 9]. As such, the current ACC/AHA guidelines on assessment of cardiovascular risk state that assessment of CACS may be considered based on a large number of observational studies: with a CACS of ≥ 300 Agatston units (or ≥ 75th percentile for age, sex and ethnicity) supporting an upward revision in risk assessment [10]. A range of alternative approaches to application of CACS for risk stratification in primary prevention has been proposed recently [11,12,13].
Most clinical radiologists and cardiologists will be aware of other cardiac imaging properties that can be obtained from coronary calcium scans, such as large myocardial scars or dimensions of the heart and the thoracic aorta [14]. These can be assessed to detect past-myocardial infarction, dilated cardiomyopathies, atrial enlargement, aneurysms and pericardial effusion. However, coronary calcium scans contain a wealth of untapped information on other cardiovascular and non-cardiovascular health parameters [15, 16]. It is important for clinicians to be aware of the potential data on cardio-metabolic and general health that can be obtained from such scans without making any modifications to the scan protocol (Table 1). Hence, the goal of this review is to provide an overview of some of the most apparent imaging markers related to cardio-metabolic and general health. Additionally, we discuss potential incidental findings and radiation exposure of coronary calcium scans.
Markers of cardio-metabolic health
With the increasing focus on preventive medicine and the accompanying demand for individual risk stratification, the ability to calculate a patient’s risk of a clinical event relies greatly on the accuracy and amount of the acquired information. The coronary calcium scan can provide us with additional information regarding the patient’s cardiovascular health beyond the CACS. In the following paragraphs we address several of these markers.
Coronary artery calcium volume and density
The Agatston-based CACS is a summary measure based on the total volume and density of epicardial coronary calcification into a single number ranging from 0 (i.e. the absence of calcifications) to scores of several thousand indicating extensive coronary atherosclerosis. However, more recent evidence suggests that calcium volume and density each separately harbour additional information with regard to the risk of subsequent clinical events [17,18,19]. Importantly, these measures of density and volume generally do not require additional processing or calculation, as these can be provided by most commercially available CACS scoring software. Moreover, the number and the regional distribution of calcifications can easily be visually assessed and provide additive predictive information regarding the future risk of major coronary events [20]. As a consequence, very recently a change in methodology to assess coronary calcium scans was proposed in order to incorporate this additional information into a new CACS [21].
Valvular calcification
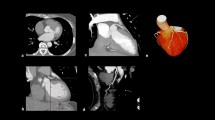
Using the same software as is used to obtain the CACS, one can quantitatively assess the burden of aortic valve calcification (Fig. 1, blue) [22, 23] or mitral annular calcification in the form of Agatston scores or volumes. The extent of aortic valvular calcification is a direct representation of degenerative aortic valve stenosis [24] and is associated with adverse cardiovascular outcomes and mortality [25, 26]. More specifically, recent evidence even highlighted that the load of aortic valve calcification measured by CT provides incremental prognostic value to predict aortic valve stenosis progression and subsequent occurrence of clinical events [27]. Similarly, mitral annular calcification, although less prevalent [28], was found to be associated with CACS [29], and to increase the risk of atrial fibrillation [30]. Additionally, progression of mitral annular calcification are an important predictor underlying left atrial abnormalities that predispose to atrial fibrillation [31].
Epicardial fat
Epicardial fat is defined as the layer of metabolically active adipose tissue that surrounds the myocardium and the coronary arteries [32, 33]. Given this close anatomical connection, changes in the amount of epicardial fat may directly influence these structures. Larger amounts of epicardial fat are associated with more extensive coronary atherosclerosis [34,35,36], but also with direct arrhythmogenic effects on the myocardium in the form of an increased risk of new-onset atrial fibrillation and greater burden of atrial fibrillation [37, 38]. Due to rapid improvements in image-processing techniques it has become possible to quantify the amount of epicardial fat on non-contrast CT scans [39, 40]. These quantification methods are robust and fully automatic, but have not yet reached the same level of usability as commercially available software packages for calcium scoring. However, given the recent insights in the clinical importance of epicardial fat, implementation of tools for epicardial fat quantification in such software packages are expected.
Liver density
In most instances, a coronary calcium scan also includes visualization of the upper part of the liver. Despite this being only a limited part of the whole liver, measurement of the mean attenuation value at two or three locations – which can readily be done using any CT-image viewer – appears to reflect the total amount of fat in the liver [41, 42]. In turn, the amount of liver fat is regarded as an important precursor of the metabolic syndrome, and is related to both subclinical and clinical cardiovascular disease [43, 44]. Liver density may also reflect subclinical hepatic fluid congestion and liver fat is associated with adverse cardiac remodelling, both of which may herald future heart failure [45].
Pulmonary artery diameter
The diameter of the pulmonary artery (Fig. 1, orange) can be measured on non-contrast scans using any CT-image viewer and may be considered as a marker of pulmonary arterial pressure [46]. When adjusted for body size by comparison to the aortic diameter in the same slice (i.e. the pulmonary-artery-to-aorta ratio), increased pulmonary artery diameters are related to risk of future adverse pulmonary events and mortality, particularly in individuals with chronic obstructive pulmonary disease [46, 47].
Beyond markers of cardio-metabolic health
In addition to aforementioned markers of cardio-metabolic health, other structures that are imaged provide additional information on for example fracture risk and the presence or risk of pulmonary events (Table 1).
Bone density
With regard to measuring the bone density (Fig.1, pink), it should be acknowledged that apart from the heart, there may be considerable variation in the imaged area, depending on patient size and position. Yet, the majority of scans will include multiple thoracic vertebrae that can be assessed for bone mineral density – a key modifiable risk factor for osteoporotic fractures [48, 49] – or the presence of vertebral osteoporotic fractures [49].
Lung density
Measuring lung density (Fig. 1, dark blue) as a direct marker of emphysema can be challenging, because in most clinical settings the field-of-view is narrowly set to visualize coronary calcium only [50]. Nonetheless, the overall lung density can generally be measured in the lower lobes of the lungs and in the areas surrounding the hila. However, it is important to mention that apart from this dedicated, narrow field-of-view, one may consider additionally reconstructing the coronary calcium scan with a wider field-of-view to also visualize all the lung tissue that was originally in the scan field. Although the tops of the lungs will still be missing, one can obtain an accurate impression of the status of the remaining part of the lungs with respect to the amount of emphysema [51, 52]. A downside of this wider field-of-view is the greater probability of detecting incidental findings.
Incidental findings
When performing imaging, both in the clinical setting as well as in the research setting, incidental findings can be expected. However, the spectrum of potential incidental findings is relatively limited for coronary calcium scans [53]. Apart from cardiovascular abnormalities, incidental findings may especially be detected in the liver and the lungs. Given that no contrast is administered during a coronary calcium scan, potential findings in the liver are largely restricted to cystic lesions. However, for the lungs a substantial number of pulmonary nodules may be expected. Especially for older individuals and smokers, clear-cut criteria on the diagnostic work-up of such pulmonary nodules have been established and refined in the past decade [54, 55]. Other less frequent incidental findings may include interstitial changes of the lung, pleural effusion, chest wall abnormalities, breast calcifications and mediastinal lymphadenopathy.
Radiation
A topic of concern accompanying the use of the coronary calcium scan is the ionizing radiation exposure to the patient or, in the research setting, to the study participant [56]. Two general key principles that should always be kept in mind when ordering a CT examination of any kind are justification in ordering the examination and optimization of the scan protocol in the way that the radiation exposure is as-low-as-reasonably-achievable (ALARA). With the newer generation CT scanners and improvements in scan protocols, radiation doses have been decreasing over the last few years and are expected to decrease further with advances in technology [57]. Specifically for prospective ECG-gated non-contrast coronary calcium scans, radiation exposure approximates 1.5 mSv (estimated using ImpactDose version 2.3, 2016, CT Imaging GmbH, Erlangen, Germany) [58, 59]. For comparison, the annual background radiation varies between 2 and 5 mSv. Nonetheless, radiation exposure should always be weighed against the information obtained from a coronary calcium scan. Following the ALARA principle in minimizing radiation exposure, it seems only reasonable to also force clinicians and researchers to transpose this principle to data acquisition once a scan is made: acquire as much as reasonably achievable relevant information from every imaging study.
Conclusion
The clinical value of the CACS in terms of individual risk assessment of future cardiac events has led to an increased use of non-contrast cardiac CT in both clinical and research settings during the past decades. Many other markers of cardio-metabolic health and general health may readily be evaluated on these examinations. Clinical cardiologists, cardiovascular radiologists and medical specialists in the field of preventive medicine should be aware of this potential diagnostic and prognostic extra-coronary yield of the coronary calcium scan, and widen their professional field-of-view to look beyond the heart.
Abbreviations
- ALARA:
-
As-low-as-reasonably-achievable
- CACS:
-
Coronary artery calcium score
- CT:
-
Computed tomography
- ECG:
-
Electrocardiography
References
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15:827–832
Becker CR, Knez A, Jakobs TF et al (1999) Detection and quantification of coronary artery calcification with electron-beam and conventional CT. Eur Radiol 9:620–624
Kavousi M, Elias-Smale S, Rutten JH et al (2012) Evaluation of newer risk markers for coronary heart disease risk classification: a cohort study. Ann Intern Med 156:438–444
Polonsky TS, McClelland RL, Jorgensen NW et al (2010) Coronary artery calcium score and risk classification for coronary heart disease prediction. JAMA 303:1610–1616
Leening MJG, Elias-Smale SE, Kavousi M et al (2012) Coronary calcification and the risk of heart failure in the elderly: the Rotterdam Study. JACC Cardiovasc Imaging 5:874–880
Handy CE, Desai CS, Dardari ZA et al (2016) The association of coronary artery calcium with noncardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc Imaging 9:568–576
Fujiyoshi A, Jacobs DR Jr., Fitzpatrick AL et al (2017) Coronary artery calcium and risk of dementia in MESA (Multi-Ethnic Study of Atherosclerosis). Circ Cardiovasc Imaging 10:e005349
Hecht HS (2010) A zero coronary artery calcium score: priceless. J Am Coll Cardiol 55:1118–1120
Blaha MJ, Cainzos-Achirica M, Greenland P et al (2016) Role of coronary artery calcium score of zero and other negative risk markers for cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 133:849–858
Goff DC Jr, Lloyd-Jones DM, Bennett G et al (2014) 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 63:2935–2959
Blaha MJ, Matsushita K (2017) Coronary artery calcium: need for more clarity in guidelines. JACC Cardiovasc Imaging 10:154–156
Leening MJG, Cook NR, Ridker PM (2017) Should we reconsider the role of age in treatment allocation for primary prevention of cardiovascular disease? Eur Heart J 38:1542–1547
Pender A, Lloyd-Jones DM, Stone NJ, Greenland P (2016) Refining statin prescribing in lower-risk individuals: informing risk/benefit decisions. J Am Coll Cardiol 68:1690–1697
Shahzad R, Bos D, Budde RP et al (2017) Automatic segmentation and quantification of the cardiac structures from non-contrast-enhanced cardiac CT scans. Phys Med Biol 62:3798–3813
Madaj P, Budoff MJ (2012) Risk stratification of non-contrast CT beyond the coronary calcium scan. J Cardiovasc Comput Tomogr 6:301–307
Mahabadi AA, Lehmann N, Mohlenkamp S et al (2016) Noncoronary measures enhance the predictive value of cardiac CT above traditional risk factors and CAC score in the general population. JACC Cardiovasc Imaging 9:1177–1185
Criqui MH, Denenberg JO, Ix JH et al (2014) Calcium density of coronary artery plaque and risk of incident cardiovascular events. JAMA 311:271–278
Callister TQ, Cooil B, Raya SP, Lippolis NJ, Russo DJ, Raggi P (1998) Coronary artery disease: improved reproducibility of calcium scoring with an electron-beam CT volumetric method. Radiology 208:807–814
Bos D, Leening MJG, Kavousi M et al (2015) Comparison of atherosclerotic calcification in major vessel beds on the risk of all-cause and cause-specific mortality: the Rotterdam Study. Circ Cardiovasc Imaging 8:e003843
Ferencik M, Pencina KM, Liu T et al (2017) Coronary artery calcium distribution is an independent predictor of incident major coronary heart disease events: results from the Framingham Heart Study. Circ Cardiovasc Imaging 10:e006592
Blaha MJ, Mortensen MB, Kianoush S, Tota-Maharaj R, Cainzos-Achirica M (2017) Coronary artery calcium scoring: is it time for a change in methodology? JACC Cardiovasc Imaging 10:923–937
Bos D, Bozorgpourniazi A, Mutlu U et al (2016) Aortic valve calcification and risk of stroke: the Rotterdam Study. Stroke 47:2859–2861
Shavelle DM, Budoff MJ, Buljubasic N et al (2003) Usefulness of aortic valve calcium scores by electron beam computed tomography as a marker for aortic stenosis. Am J Cardiol 92:349–353
Otto CM, Prendergast B (2014) Aortic-valve stenosis--from patients at risk to severe valve obstruction. N Engl J Med 371:744–756
Lindman BR, Clavel MA, Mathieu P et al (2016) Calcific aortic stenosis. Nat Rev Dis Primers 2:16006
Thanassoulis G, Campbell CY, Owens DS et al (2013) Genetic associations with valvular calcification and aortic stenosis. N Engl J Med 368:503–512
Tastet L, Enriquez-Sarano M, Capoulade R et al (2017) Impact of aortic valve calcification and sex on hemodynamic progression and clinical outcomes in AS. J Am Coll Cardiol 69:2096–2098
Kanjanauthai S, Nasir K, Katz R et al (2010) Relationships of mitral annular calcification to cardiovascular risk factors: the Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 213:558–562
Hamirani YS, Nasir K, Blumenthal RS et al (2011) Relation of mitral annular calcium and coronary calcium (from the Multi-Ethnic Study of Atherosclerosis [MESA]). Am J Cardiol 107:1291–1294
O'Neal WT, Efird JT, Nazarian S, Alonso A, Heckbert SR, Soliman EZ (2015) Mitral annular calcification and incident atrial fibrillation in the Multi-Ethnic Study of Atherosclerosis. Europace 17:358–363
O'Neal WT, Efird JT, Nazarian S et al (2017) Mitral annular calcification progression and the risk of atrial fibrillation: results from MESA. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jex093
Iacobellis G (2009) Epicardial and pericardial fat: close, but very different. Obesity (Silver Spring) 17:625 author reply 626-627
Iacobellis G, Bianco AC (2011) Epicardial adipose tissue: emerging physiological, pathophysiological and clinical features. Trends Endocrinol Metab 22:450–457
Bettencourt N, Toschke AM, Leite D et al (2012) Epicardial adipose tissue is an independent predictor of coronary atherosclerotic burden. Int J Cardiol 158:26–32
Bos D, Shahzad R, van Walsum T et al (2015) Epicardial fat volume is related to atherosclerotic calcification in multiple vessel beds. Eur Heart J Cardiovasc Imaging 16:1264–1269
Ding J, Kritchevsky SB, Hsu FC et al (2008) Association between non-subcutaneous adiposity and calcified coronary plaque: a substudy of the Multi-Ethnic Study of Atherosclerosis. Am J Clin Nutr 88:645–650
Bos D, Vernooij MW, Shahzad R et al (2017) Epicardial fat volume and the risk of atrial fibrillation in the general population free of cardiovascular disease. JACC Cardiovasc Imaging. 10:1405–1407
Wong CX, Ganesan AN, Selvanayagam JB (2017) Epicardial fat and atrial fibrillation: current evidence, potential mechanisms, clinical implications, and future directions. Eur Heart J 38:1294–1302
Shahzad R, Bos D, Metz C et al (2013) Automatic quantification of epicardial fat volume on non-enhanced cardiac CT scans using a multi-atlas segmentation approach. Med Phys 40:091910
Dey D, Wong ND, Tamarappoo B et al (2010) Computer-aided non-contrast CT-based quantification of pericardial and thoracic fat and their associations with coronary calcium and metabolic syndrome. Atherosclerosis 209:136–141
Kodama Y, Ng CS, Wu TT et al (2007) Comparison of CT methods for determining the fat content of the liver. Am J Roentgenol 188:1307–1312
Wolff L, Bos D, Murad SD et al (2016) Liver fat is related to cardiovascular risk factors and subclinical vascular disease: the Rotterdam Study. Eur Heart J Cardiovasc Imaging 17:1361–1367
Lonardo A, Ballestri S, Marchesini G, Angulo P, Loria P (2015) Nonalcoholic fatty liver disease: a precursor of the metabolic syndrome. Dig Liver Dis 47:181–190
Galassi A, Reynolds K, He J (2006) Metabolic syndrome and risk of cardiovascular disease: a meta-analysis. Am J Med 119:812–819
VanWagner LB, Wilcox JE, Colangelo LA et al (2015) Association of nonalcoholic fatty liver disease with subclinical myocardial remodeling and dysfunction: a population-based study. Hepatology 62:773–783
Terzikhan N, Bos D, Lahousse L et al (2017) Pulmonary artery to aorta ratio and risk of all-cause mortality in the general population: the Rotterdam Study. Eur Respir J 49:1602168
Wells JM, Washko GR, Han MK et al (2012) Pulmonary arterial enlargement and acute exacerbations of COPD. N Engl J Med 367:913–921
Mao SS, Luo Y, Fischer H, Buodff MJ, Li D (2016) Routine coronary calcium scan can precisely measure vertebral bone density without a quantitative calibration phantom. J Comput Assist Tomogr 40:126–130
Chou SH, LeBoff MS (2017) Vertebral imaging in the diagnosis of osteoporosis: a clinician's perspective. Curr Osteoporos Rep. 15:509–520
Halliburton SS, Abbara S, Chen MY et al (2011) SCCT guidelines on radiation dose and dose-optimization strategies in cardiovascular CT. J Cardiovasc Comput Tomogr 5:198–224
Barr RG, Bluemke DA, Ahmed FS et al (2010) Percent emphysema, airflow obstruction, and impaired left ventricular filling. N Engl J Med 362:217–227
Hoffman EA, Jiang R, Baumhauer H et al (2009) Reproducibility and validity of lung density measures from cardiac CT scans: the Multi-Ethnic Study of Atherosclerosis (MESA) Lung Study. Acad Radiol 16:689–699
Rahmani S, Budoff M (2016) Incidental findings in a population-based study using cardiac CT: experience from the Multi-Ethnic Study of Atherosclerosis. In: Kauczor HU, Hricak H, Essig M, Brady LW, Lu J (eds) Medical Radiology. Springer, Berlin Heidelberg
MacMahon H, Austin JH, Gamsu G et al (2005) Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the Fleischner Society. Radiology 237:395–400
MacMahon H, Naidich DP, Goo JM et al (2017) Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology 284:228–243
Kim KP, Einstein AJ, Berrington de Gonzalez A (2009) Coronary artery calcification screening: estimated radiation dose and cancer risk. Arch Intern Med 169:1188–1194
Paul JF, Abada HT (2007) Strategies for reduction of radiation dose in cardiac multislice CT. Eur Radiol 17:2028–2037
Kalender WA, Schmidt B, Zankl M, Schmidt M (1999) A PC program for estimating organ dose and effective dose values in computed tomography. Eur Radiol 9:555–562
Budoff MJ, Gupta M (2010) Radiation exposure from cardiac imaging procedures: do the risks outweigh the benefits? J Am Coll Cardiol 56:712–714
Acknowledgements
The authors thank Dr. Matthew J. Budoff, MD FACC FAHA FSCCT (Division of Cardiology, Los Angeles Biomedical Research Institute, Harbor-UCLA Medical Center, Torrance, CA, USA) for his comments on a draft version of the manuscript.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Daniel Bos.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Informed consent was not required because the current article is a review article.
Ethical approval
Institutional Review Board approval was not required because the current article is a review article.
Methodology
• Performed at one institution.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bos, D., Leening, M.J.G. Leveraging the coronary calcium scan beyond the coronary calcium score. Eur Radiol 28, 3082–3087 (2018). https://doi.org/10.1007/s00330-017-5264-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5264-3