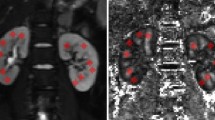
Abstract
Objective
Our objective was to evaluate pathological and functional changes in chronic kidney disease (CKD) using diffusion tensor imaging (DTI) at 3 T.
Methods
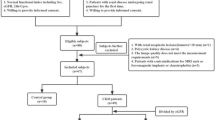
There were fifty-one patients with CKD who required biopsy and 19 healthy volunteers who were examined using DTI at 3 T. The mean values of fractional anisotropy (FA) and the apparent diffusion coefficient (ADC) were obtained from the renal parenchyma (cortex and medulla). Correlations between imaging results and the estimated glomerular filtration rate (eGFR), as well as pathological damage (glomerular lesion and tubulointerstitial injury), were evaluated.
Results
The renal cortical FA was significantly lower than the medullary in both normal and affected kidneys (p < 0.001). The parenchymal FA was significantly lower in patients than healthy controls, regardless of whether eGFR was reduced. There were positive correlations between eGFR and FA (cortex, r = 0.689, p = 0.000; and medulla, r = 0.696, p = 0.000), and between eGFR and ADC (cortex, r = 0.310, p = 0.017; and medulla, r = 0.356, p = 0.010). Negative correlations were found between FA and the glomerular lesion (cortex, r = -0.499, p = 0.000; and medulla, r = -0.530, p = 0.000), and between FA and tubulointerstitial injury (cortex, r = -0.631, p = 0.000; and medulla, r = -0.724, p = 0.000).
Conclusion
DTI is valuable for noninvasive assessment of renal function and pathology in patients with CKD. A decrease in FA could identify the glomerular lesions, tubulointerstitial injuries, and eGFR.
Key Points
• DTI can evaluate CKD regardless of whether the eGFR is reduced.
• DTI allows the assessment of renal pathology changes.
• FA appears sensitive and stable in detecting renal pathology and function.





Similar content being viewed by others
References
Levey AS, Stevens LA, Coresh J (2009) Conceptual model of CKD: applications and implications. Am J Kidney Dis 53:S4–16
Coresh J, Selvin E, Stevens LA et al (2007) Prevalence of chronic kidney disease in the United States. JAMA 298:2038–2047
Jiang SH, Karpe KM, Talaulikar GS (2011) Safety and predictors of complications of renal biopsy in the outpatient setting. Clin Nephrol 76:464–469
Eisenberger U, Thoeny HC, Binser T et al (2010) Evaluation of renal allograft function early after transplantation with diffusion-weighted MR imaging. Eur Radiol 20:1374–1383
Thoeny HC, De Keyzer F, Oyen RH, Peeters RR (2005) Diffusion-weighted MR imaging of kidneys in healthy volunteers and patients with parenchymal diseases: initial experience. Radiology 235:911–917
Namimoto T, Yamashita Y, Mitsuzaki K, Nakayama Y, Tang Y, Takahashi M (1999) Measurement of the apparent diffusion coefficient in diffuse renal disease by diffusion-weighted echo-planar MR imaging. J Magn Reson Imaging 9:832–837
Xu X, Fang W, Ling H, Chai W, Chen K (2010) Diffusion-weighted MR imaging of kidneys in patients with chronic kidney disease: initial study. Eur Radiol 20:978–983
Blondin D, Lanzman RS, Mathys C et al (2009) Functional MRI of transplanted kidneys using diffusion-weighted imaging. Röfo 181:1162–1167
Thoeny HC, De Keyzer F (2011) Diffusion-weighted MR imaging of native and transplanted kidneys. Radiology 259:25–38
Dixon WT (1988) Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging: a modest proposal with tremendous potential. Radiology 168:566–567
Hueper K, Gutberlet M, Rodt T et al (2011) Diffusion tensor imaging and tractography for assessment of renal allograft dysfunction-initial results. Eur Radiol 21:2427–2433
Gaudiano C, Clementi V, Busato F et al (2013) Diffusion tensor imaging and tractography of the kidneys: assessment of chronic parenchymal diseases. Eur Radiol 23:1678–1685
Lanzman RS, Ljimani A, Pentang G et al (2013) Kidney transplant: functional assessment with diffusion-tensor MR imaging at 3 T. Radiology 266:218–225
Mukherjee P, Berman JI, Chung SW, Hess CP, Henry RG (2008) Diffusion tensor MR imaging and fiber tractography: theoretic underpinnings. AJNR Am J Neuroradiol 29:632–641
Levey AS, Stevens LA, Schmid CH et al (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150:604–612
Notohamiprodjo M, Glaser C, Herrmann KA et al (2008) Diffusion tensor imaging of the kidney with parallel imaging: initial clinical experience. Invest Radiol 43:677–685
Li Q, Li J, Zhang L, Chen Y, Zhang M, Yan F (2014) Diffusion-weighted imaging in assessing renal pathology of chronic kidney disease: A preliminary clinical study. Eur J Radiol 83:756–762
Katafuchi R, Kiyoshi Y, Oh Y et al (1998) Glomerular score as a prognosticator in IgA nephropathy: its usefulness and limitation. Clin Nephrol 49:1–8
Chandarana H, Lee VS (2009) Renal functional MRI: Are we ready for clinical application? AJR Am J Roentgenol 192:1550–1557
Kataoka M, Kido A, Yamamoto A et al (2009) Diffusion tensor imaging of kidneys with respiratory triggering: optimization of parameters to demonstrate anisotropic structures on fraction anisotropy maps. J Magn Reson Imaging 29:736–744
Notohamiprodjo M, Dietrich O, Horger W et al (2010) Diffusion tensor imaging (DTI) of the kidney at 3 tesla-feasibility, protocol evaluation and comparison to 1.5 Tesla. Invest Radiol 45:245–254
Gaudiano C, Clementi V, Busato F et al (2011) Renal diffusion tensor imaging: is it possible to define the tubular pathway? A case report. Magn Reson Imaging 29:1030–1033
Lu L, Sedor JR, Gulani V et al (2011) Use of diffusion tensor MRI to identify early changes in diabetic nephropathy. Am J Nephrol 34:476–482
Fukuda Y, Ohashi I, Hanafusa K et al (2000) Anisotropic diffusion in kidney: apparent diffusion coefficient measurements for clinical use. J Magn Reson Imaging 11:156–160
Kido A, Kataoka M, Yamamoto A et al (2010) Diffusion tensor MRI of the kidney at 3.0 and 1.5 Tesla. Acta Radiol 51:1059–1063
Hueper K, Hartung D, Gutberlet M et al (2012) Magnetic resonance diffusion tensor imaging for evaluation of histopathological changes in a rat model of diabetic nephropathy. Invest Radiol 47:430–437
Cheung JS, Fan SJ, Gao DS, Chow AM, Man K, Wu EX (2010) Diffusion tensor imaging of liver fibrosis in an experimental model. J Magn Reson Imaging 32:1141–1148
Heusch P, Wittsack HJ, Kropil P et al (2013) Impact of blood flow on diffusion coefficients of the human kidney: a time-resolved ECG-triggered diffusion-tensor imaging (DTI) study at 3 T. J Magn Reson Imaging 37:233–236
Thoeny HC, Zumstein D, Simon-Zoula S et al (2006) Functional evaluation of transplanted kidneys with diffusion-weighted and BOLD MR imaging: initial experience. Radiology 241:812–821
Melhem ER, Itoh R, Jones L, Barker PB (2000) Diffusion tensor MR imaging of the brain: effect of diffusion weighting on trace and anisotropy measurements. AJNR Am J Neuroradiol 21:1813–1820
Wang WJ, Pui MH, Guo Y, Hu XS, Wang HJ, Yang D (2013) MR diffusion tensor imaging of normal kidneys. J Magn Reson Imaging. doi:10.1002/jmri.24450
Acknowledgements
The scientific guarantor of this publication is Qingwei Liu. The authors of this manuscript declare no relationship with any company. This study has received funding by the National Natural Science Foundation of China (61301253), Shandong Province Science and Technology Development Plan (2012GSF11820 and 2012YD18053), and the Foundation for Outstanding Young Scientist in Shandong Province (2010BSB14072). No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. These study subjects or cohorts have not been previously reported. Methodology: prospective, diagnostic or prognostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Z., Xu, Y., Zhang, J. et al. Chronic kidney disease: pathological and functional assessment with diffusion tensor imaging at 3T MR. Eur Radiol 25, 652–660 (2015). https://doi.org/10.1007/s00330-014-3461-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3461-x