Abstract
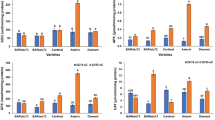
The sweetpotato cultivar Quick Sweet (QS) with a lower pasting temperature of starch is a unique breeding material, but the biochemical background of this property has been unknown. To assess the physiological impact of the reduced isoform II activity of starch synthase (SSII) on the starch properties in sweetpotato storage root, transgenic sweetpotato plants with reduced expressions of the SSII gene were generated and evaluated. All of the starches from transgenic plants showed lower pasting temperatures and breakdown measured by a Rapid Visco Analyzer. The pasting temperatures in transgenic plants were approximately 10–15°C lower than in wild-type plants. Distribution of the amylopectin chain length of the transgenic lines showed marked differences compared to that in wild-type plants: more chains with degree of polymerization (DP) 6–11 and fewer chains with DP 13–25. The starch granules from the storage root of transgenic plants showed cracking on the hilum, while those from wild-type plants appeared to be typical sweetpotato starch. In accordance with these observations, the expression of SSII in the storage roots of the sweetpotato cultivar with low pasting temperature starch (QS) was notably lower than in cultivars with normal starch. Moreover, nucleotide sequence analysis suggested that most of the SSII transcripts in the cultivar with low pasting temperature starch were inactive alleles. These results clearly indicate that the activity of SSII in sweetpotato storage roots, like those in other plants, affects the pasting properties of starch through alteration of the amylopectin structure.







Similar content being viewed by others
References
Craig J, Lloyd JR, Tomlinson K, Barber L, Edwards A, Wang TL, Martin C, Hedley CL, Smith AM (1998) Mutations in the gene encoding starch synthase II profoundly alter amylopectin structure in pea embryos. Plant Cell 10:413–426
Edwards A, Fulton DC, Hylton CM, Jobling SA, Gidley M, Roessner U, Martin C, Smith AM (1999) A combined reduction in activity of starch synthase II and III of potato has novel effect on the starch of tubers. Plant J 17:251–261
Hirose T, Terao T (2004) A comprehensive expression analysis of the starch synthase gene family in rice (Oryza sativa L.). Planta 220:9–16
Höfgen R, Willmitzer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877
Katayama K, Komae K, Kohyama K, Kato T, Tamiya S, Komaki K (2002) New sweetpotato line having low gelatinization temperature and altered starch structure. Staerke 54:51–57
Katayama K, Tamiya S, Kuranouchi T, Komaki K, Nakatani M (2003) New sweetpotato cultivar “Quick Sweet”. Bull Natl Inst Crop Sci 3:35–52
Katayama K, Tamiya S, Ishiguro K (2004) Starch properties of new sweet potato lines having low pasting temperature. Staerke 56:563–569
Kimura T, Ideta O, Saito A (2000) Identification of the gene encoding granule-bound starch synthase I in sweet potato (Ipomoea batatas (L.) Lam.). Plant Biotechnol 17:247–252
Kimura T, Otani M, Noda T, Ideta O, Shimada T, Saito A (2001) Absence of amylose in sweet potato [Ipomoea batatas (L.) Lam.] following the introduction of granule-bound starch synthase I cDNA. Plant Cell Rep 20:663–666
Kitahara K (2005) Study on organ-specific starch properties of sweetpotato. J Appl Glycosci 52:45–50
Kitahara K, Ooi Y, Mizukami S, Suganuma T, Nagahama T (1996) Physicochemical properties of starches from sweetpotato cultivars. J Appl Glycosci 43:59–66
Kitahara K, Imamura K, Omae Y, Suganuma T (1998) Characterization of molecular structure of starch granules in suspension-cultured cells from Ipomoea cordatotriloba Denn. Biosci Biotechnol Biochem 62:1962–1967
Kitahara K, Ueno J, Suganuma T, Ishiguro K, Yamakawa O (1999) Physicochemical properties of root starches from new types of sweetpotato. J Appl Glycosci 46:391–397
Kitahara K, Fukunaga S, Katayama K, Takahata Y, Nakazawa Y, Yoshinaga M, Suganuma T (2005) Physicochemical properties of sweetpotato starches with different gelatinization temperatures. Starch/Staerke 57:473–479
Kitahara K, Hamasuna K, Nozuma K, Otani M, Hamada T, Shimada T, Fujita K, Suganuma T (2007) Physicochemical properties of amylose-free and high-amylose starches from transgenic sweetpotatoes modified by RNA interference. Carbohydr Polym 69:233–240
Lin DG, Jeang CL (2005) cDNA cloning, expression, and characterization of taro SSII: a novel member of starch synthase II family. J Agric Food Chem 53:7958–7964
Lloyd JR, Landschutze V, Kossmann J (1999) Simultaneous antisence inhibition of two starch-synthase isoforms in potato tubers leads to accumulation of grossly modified amylopectin. Biochem J 338:515–521
Morell MK, Kosar-Hashemi B, Cmiel M, Samuel MS, Chandler P, Rahman S, Buleon A, Batey IL, Li Z (2003) Barley sex6 mutants lack starch synthase IIa activity and contain a starch with novel properties. Plant J 34:173–185
Murata T (1970) Enzymic mechanism of starch synthesis in sweet potato roots. Part III. The composition of carbohydrates and soluble nucleotides in the developing sweet potato roots. Nogei Kagaku Kaishi 44:412–421
Murata T, Akazawa T (1968) Enzymic mechanism of starch synthesis in sweet potato roots. I. Requirement of potassium ions for starch synthase. Arch Biochem Biophys 126:873–879
Nakamura Y, Francisco PB Jr, Hosaka Y, Sato A, Sawada T, Kubo A, Fujita N (2005) Essential amino acids of starch synthase IIa differentiate amylopectin structure and starch quality between japonica and indica rice varieties. Plant Mol Biol 58:213–227
Noda T (2001) Effect of cultivar and growth conditions on starch properties of sweet potatoes. J Appl Glycosci 48:233–238
Noda T, Takahata Y, Sato T, Suda I, Morishita T, Ishiguro K, Yamakawa O (1998) Relationships between chain length distribution of amylopectin and gelatinization properties within the same botanical origin for sweetpotato and buckwheat. Carbohydr Polym 37:153–158
Noda T, Kimura T, Otani M, Ideta O, Shimada T, Saito A, Suda I (2002) Physicochemical properties of amylose-free starch from transgenic sweet potato. Carbohydr Polym 49:253–260
Otani M, Shimada T, Kimura T, Saito A (1998) Transgenic plant production from embryogenic callus of sweet potato (Ipomoea batatas (L.) Lam.) using Agrobacterium tumefaciens. Plant Biotechnol 15:11–16
Otani M, Hamada T, Katayama K, Kitahara K, Kim SH, Takahata Y, Suganuma T, Shimada T (2007) Inhibition of the gene expression for granule-bound starch synthase I by RNA interference in sweet potato plants. Plant Cell Rep 26:1801–1807
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Shimada T, Otani M, Hamada T, Kim SH (2006) Increase of amylose content of sweetpotato starch by RNA interference of the starch branching enzyme II gene (IbSBEII). Plant Biotechnol 23:85–90
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4. Mol Biol Evol 24:1596–1599
Tanaka M, Takahata Y, Nakatani M (2005) Analysis of genes developmentally regulated during storage root formation of sweet potato. J Plant Physiol 162:91–102
Tanaka M, Takahata Y, Nakayama H, Nakatani M, Tahara M (2009) Altered carbohydrate metabolism in the storage roots of sweetpotato plants overexpressing the SRF1 gene, which encodes a Dof zinc finger transcription factor. Planta 230:737–746
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 24:4876–4882
Umemoto T, Yano M, Satoh H, Shomura A, Nakamura Y (2002) Mapping of a gene responsible for the difference in amylopectin structure between japonica-type and indica-type rice varieties. Theor Appl Genet 104:1–8
Umemoto T, Aoki N, Lin H, Nakamura Y, Inouchi N, Sato Y, Yano M, Hirabayashi H, Maruyama S (2004) Natural variation in rice starch synthase IIa affects enzyme and starch properties. Funct Plant Biol 31:671–684
Vandesompele J, Preter KD, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:research0034.1–0034.11
Wang SJ, Yeh KW, Tsai CY (1999) Molecular characterization and expression of starch granule-bound starch synthase in the sink and source tissues of sweet potato. Physiol Plant 106:253–261
Yamamori M, Fujita S, Hayakawa K, Matsuki J, Yasui T (2000) Genetic elimination of a starch granule protein, SGP-1, of wheat generates an altered starch with apparent high amylose. Theor Appl Genet 101:21–29
Zhang X, Colleoni C, Ratushna V, Sirghie-Colleoni M, James MG, Myers AM (2004) Molecular characterization demonstrates that the Zea mays gene sugary2 codes for the starch synthase isoform SSIIa. Plant Mol Biol 54:865–879
Zhang X, Szydlowski N, Delvalle D, D’Hulst C, James MG, Myers AM (2008) Overlapping functions of the starch synthases SSII and SSIII in amylopectin biosynthesis in Arbidopsis. BMC Plant Biol 8:96
Acknowledgments
The authors would like to thank Professor Kazufumi Yazaki at Kyoto University for kindly providing the hygromycin resistant gene. We also thank the staff members in the field and greenhouse management section 3 of KONARC for managing the plant materials, and Rieko Gonbori and Masako Yoshigaki, KONARC, for their laboratory work. This work was supported by grants from the Ministry of Agriculture, Forestry and Fisheries of Japan (Rural Bio-Mass Research Project, BCD-A1322, Bio-recycle Research Project, 1612).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Ebinuma.
Rights and permissions
About this article
Cite this article
Takahata, Y., Tanaka, M., Otani, M. et al. Inhibition of the expression of the starch synthase II gene leads to lower pasting temperature in sweetpotato starch. Plant Cell Rep 29, 535–543 (2010). https://doi.org/10.1007/s00299-010-0842-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-010-0842-8