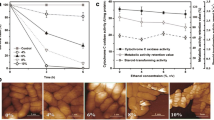
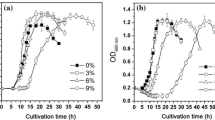
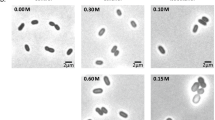
Abstract
Chemical activities of hydrophobic substances can determine the windows of environmental conditions over which microbial systems function and the metabolic inhibition of microorganisms by benzene and other hydrophobes can, paradoxically, be reduced by compounds that protect against cellular water stress (Bhaganna et al. in Microb Biotechnol 3:701–716, 2010; Cray et al. in Curr Opin Biotechnol 33:228–259, 2015a). We hypothesized that this protective effect operates at the macromolecule structure–function level and is facilitated, in part at least, by genome-mediated adaptations. Based on proteome profiling of the soil bacterium Pseudomonas putida, we present evidence that (1) benzene induces a chaotrope-stress response, whereas (2) cells cultured in media supplemented with benzene plus glycerol were protected against chaotrope stress. Chaotrope-stress response proteins, such as those involved in lipid and compatible-solute metabolism and removal of reactive oxygen species, were increased by up to 15-fold in benzene-stressed cells relative to those of control cultures (no benzene added). By contrast, cells grown in the presence of benzene + glycerol, even though the latter grew more slowly, exhibited only a weak chaotrope-stress response. These findings provide evidence to support the hypothesis that hydrophobic substances induce a chaotropicity-mediated water stress, that cells respond via genome-mediated adaptations, and that glycerol protects the cell’s macromolecular systems. We discuss the possibility of using compatible solutes to mitigate hydrocarbon-induced stresses in lignocellulosic biofuel fermentations and for industrial and environmental applications.


Similar content being viewed by others
Notes
In other words, there was an apparent cessation of biotic activity (NB stressor concentrations at which metabolic activity and cell division cease and at which death occurs can differ; see Santos et al. 2015).
References
Albano E (2006) Alcohol, oxidative stress and free radical damage. Proc Nutr Soc 67:278–290
Albertyn J, Hohmann S, Thevelein JM, Prior BA (1994) Gpd1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic-stress in Saccharomyces-cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol Cell Biol 14:4135–4144
Alexandre H, Ansanav-Galeote V, Dequin S, Blondin B (2001) Global gene expression during short-term ethanol stress in Saccharomyces cerevisiae. FEBS Lett 498:98–103
Alves FL, Stevenson A, Baxter E, Gillion JLM, Hejazi F, Hayes S, Morrison IEG, Prior BA, McGenity TJ, Rangel DEN, Magan N, Timmis KN, Hallsworth JE (2015) Concomitant osmotic and chaotropicity-induced stresses in Aspergillus wentii: compatible solutes determine the biotic window. Curr Genet 61:457–477
Ball P, Hallsworth JE (2015) Water structure and chaotropicity: their uses, abuses, and implications for biology. Phys Chem Chem Phys 17:8297–8305
Ballerstedt H, Volkers RJM, Mars AE, Hallsworth JE et al (2007) Genomotyping of Pseudomonas putida strains using P. putida KT2440-based high-density DNA microarrays: implications for transcriptomics studies. Appl Microbiol Biotechnol 75:1133–1142
Bell AN, Magill E, Hallsworth JE, Timson DJ (2013) Effects of alcohols and compatible solutes on the activity of β-galactosidase. Appl Biochem Biotechnol 169:786–794
Benndorf D, Thiersch M, Loffhagen N, Kunath C, Harms H (2006) Pseudomonas putida KT2440 responds specifically to chlorophenoxy herbicides and their initial metabolites. Proteomics 6:3319–3329
Bhaganna P, Volkers RJM, Bell ANW, Kluge K, Timson DJ, McGrath JW, Ruijssenaars HJ, Hallsworth JE (2010) Hydrophobic substances induce water stress in microbial cells. Microb Biotechnol 3:701–716
Bobadilla Fazzini RA, Preto MJ, Quintas AC, Bielecka A, dos Santos VA (2010) Consortia modulation of the stress response: proteomic analysis of single strain versus mixed culture. Environ Microbiol 12:2436–2449
Bobadilla Fazzini RA, Skindersoe ME, Bielecki P, Puchałka J, Givskov M, Martins dos Santos VA (2013) Protoanemonin: a natural quorum sensing inhibitor that selectively activates iron starvation response. Environ Microbiol 15:111–120
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brown AD (1990) Microbial water stress physiology. Principles and perspectives. Wiley, Chichester
Chin JP, Megaw J, Magill CL, Nowotarski K, Williams JP, Bhaganna P, Linton M, Patterson MF, Underwood GJC, Mswaka AY, Hallsworth JE (2010) Solutes determine the temperature windows for microbial survival and growth. Proc Natl Acad Sci USA 107:7835–7840
Chirife J, Favetto G, Fontán CF (1984) Microbial growth at reduced water activities: some—physicochemical properties of compatible solutes. J Appl Microbiol 56:259–268
Cray JA, Bell ANW, Bhaganna P, Mswaka AY, Timson DJ, Hallsworth JE (2013a) The biology of habitat dominance; can microbes behave as weeds? Microb Biotechnol 6:453–492
Cray JA, Russell JT, Timson DJ, Singhal RS, Hallsworth JE (2013b) A universal measure of chaotropicity and kosmotropicity. Environ Microbiol 15:287–296
Cray JA, Bhaganna P, Singhal RS, Patil SV, Saha D, Chakraborty R, Iwaguchi S, Timson DJ, Hallsworth JE (2014) Chaotropic and hydrophobic stress mechanisms of antifungal substances. In: Dehne HW, Deising HB, Fraaije B, Gisi U, Hermann D, Mehl A, Oerke EC, Russell PE, Stammler G, Kuck KH, Lyr H (eds) Modern fungicides and antifungal compounds, vol VII. Deutsche Phytomedizinische Gesellschaft, Braunschweig. ISBN: 978-3-941261-13-6
Cray JA, Stevenson A, Ball P, Bankar SB, Eleutherio ECA, Ezeji TC, Singhal RS, Thevelein JM, Timson DJ, Hallsworth JE (2015a) Chaotropicity: a key factor in product tolerance of biofuel-producing microorganisms. Curr Opin Biotechnol 33:228–259
Cray JA, Houghton JD, Cooke LR, Hallsworth JE (2015b) A simple inhibition coefficient for quantifying potency of biocontrol agents against plant-pathogenic fungi. Biol Control 81:93–100
Domínguez-Cuevas P, González-Pastor J-E, Marqués S, Ramos JL, de Lorenzo V (2006) Transcriptional tradeoff between metabolic and stress-response programs in Pseudomonas putida KT2440 cells exposed to toluene. J Biol Chem 281:11981–11991
Dos Santos VAP, Heim S, Moore ERB, Strätz M, Timmis KN (2004) Insights into the genomic basis of niche specificity of Pseudomonas putida KT2440. Environ Microbiol 6:1264–1286
Eleutherio ECA, Dearaujo PS, Panek AD (1993) Role of the trehalose carrier in dehydration resistance of Saccharomyces cerevisiae. Biochim Biophys Acta 1156:263–266
Escapa IF, García JL, Bühler B, Blank LM, Prieto MA (2012) The polyhydroxyalkanoate metabolism controls carbon and energy spillage in Pseudomonas putida. Environ Microbiol 14:1049–1063
Görg A (1991) Two-dimensional electrophoresis. Nature 349:545–546
Hallsworth JE (1998) Ethanol-induced water stress in yeast. J Ferment Bioeng 85:125–137
Hallsworth JE, Magan N (1994) Improved biological control by changing polyols/trehalose in conidia of entomopathogens. Brighton Crop Protection Conference 1994—Pests and Diseases, vol 3. BCPC Publications, Farnham, pp 1091–1096
Hallsworth JE, Magan N (1995) Manipulation of intracellular glycerol and erythritol enhances germination of conidia at low water availability. Microbiol SGM 29:7–13
Hallsworth JE, Nomura Y (1999) A simple method to determine the water activity of ethanol-containing samples. Biotechnol Bioeng 62:242–245
Hallsworth JE, Nomura Y, Iwahara M (1998) Ethanol-induced water stress and fungal growth. J Ferment Bioeng 86:451–456
Hallsworth JE, Prior BA, Nomura Y, Iwahara M, Timmis KN (2003a) Compatible solutes protect against chaotrope (ethanol)-induced, nonosmotic water stress. Appl Environ Microb 69:7032–7034
Hallsworth JE, Heim S, Timmis KN (2003b) Chaotropic solutes cause water stress in Pseudomonas putida. Environ Microbiol 5:1270–1280
Hallsworth JE, Yakimov MM, Golyshin PN, Gillion JLM, D’Auria G, Alves FL, La Cono V, Genovese M, McKew BA, Harris G, Guiliano L, Timmis KN, McGenity TJ (2007) Limits of life in MgCl2-containing environments: chaotropicity defines the window. Environ Microbiol 9:803–813
Hamada T, Maeda Y, Matsuda H, Sameshima Y, Sameshima Y, Honda K, Omasa T, Kato J, Ohtake H (2009) Effect of cell-surface hydrophobicity on bacterial conversion of water-immiscible chemicals in two-liquid-phase culture systems. J Biosci Bioeng 108:116–120
Harrison JP, Gheeraert N, Tsigelnitskiy D, Cockell CS (2013) The limits for life under multiple extremes. Trends Microbiol 21:204–212
Harrison JP, Hallsworth JE, Cockell CS (2015) Reduction of the temperature sensitivity of Halomonas hydrothermalis by iron starvation combined with microaerobic conditions. Appl Environ Microbiol 81:2156–2162
Hartmans S, Smits JP, van der Werf MJ, Volkering F, de Bont JAM (1989) Metabolism of styrene oxide and 2-phenylethanol in the styrene-degrading Xanthobacter strain 124X. Appl Environ Microbiol 55:2850–2855
Hassan MN, Afghan S, Hafeez FY (2011) Biological control of red rot in sugarcane by native pyoluteorin-producing Pseudomonas putida strain NH-50 under field conditions and its potential modes of action. Pest Manag Sci 67:1147–1154
Hathout Y, Riordan K, Gehrmann M, Fenselau C (2002) Differential protein expression in the cytosol fraction of an MCF-7 breast cancer cell line selected for resistance towards Melphalan. J Proteom Res 1:435–442
Heim S, Ferrer M, Heuer H, Regenhardt D, Nimtz M, Timmis KN (2003) Proteome reference map of Pseudomonas putida strain KT2440 for genome expression profiling: distinct responses of KT2440 and Pseudomonas aeruginosa strain PAO1 to iron deprivation and a new form of superoxide dismutase. Environ Microbiol 5:1257–1269
Hocking AD (1993) Responses of xerophilic fungi to changes in water activity. In: Jennings DH (ed) Stress Tolerance of Fungi. Marcel Decker, New York, pp 233–256
Hubalek Z (2003) Protectants used in the cryopreservation of microorganisms. Cryobiol 46:205–229
Joo WH, Shin YS, Lee Y, Park SM, Jeong KY, Seo JY, Park JU (2000) Intracellular changes of trehalose content in toluene tolerant Pseudomonas sp. BCNU 171 after exposure to toluene. Biotechnol Lett 22:1021–1024
Kar JR, Hallsworth JE, Singhal RS (2015) Glycine betaine-mediated protection of peas (Pisum sativum L.) during blanching and frozen storage. J Sci Food Agric. doi:10.1080/10942912.2015.1099043
Kar JR, Hallsworth JE, Singhal RS (2015) Fermentative production of glycine betaine and trehalose from acid whey using Actinopolyspora halophila (MTCC 263). Environ Technol Innov 3:68–76
Kashangura C, Hallsworth JE, Mswaka, AY (2006) Phenotypic diversity amongst strains of Pleurotus sajor-caju: implications for cultivation in arid environments. Mycol Res 110: 312–317
Kavazis AN, Talbert EE, Smuder AJ, Hudson MB, Nelson WB, Powers SK (2009) Mechanical ventilation induces diaphragmatic mitochondrial dysfunction and increased oxidant production. Free Rad Biol Med 46:842–850
Keane A, Lau PCK, Ghoshal S (2008) Use of a whole-cell biosensor to assess the bioavailability enhancement of aromatic hydrocarbon compounds by nonionic surfactants. Biotechnol Bioeng 99:86–98
Kelly BC, Ikonomou MG, Blair JD, Morin AE, Gobas FAPC (2007) Food web-specific biomagnification of persistent organic pollutants. Science 317:236–239
Kets EPW, de Bont JAM, Heipieper HJ (1996a) Physiological response of Pseudomonas putida S12 subjected to reduced water activity. FEMS Microbiol Lett 139:133–137
Kets EPW, Galinski EA, de Wit M, de Bont JAM, Heipieper HJ (1996b) Mannitol, a novel bacterial compatible solute in Pseudomonas putida S12. J Bacteriol 178:6665–6670
Kim K, Lee S, Lee K, Lim D (1998) Isolation and characterization of toluene-sensitive mutants from the toluene-resistant bacterium Pseudomonas putida GM73. J Bacteriol 180(14):3692–3696
Koopman F, Wierckx N, de Winde JH, Ruijssenaars HJ (2010) Identification and characterization of the furfural and 5-(hydroxymethyl)furfural-degradation pathways of Cupriavidus basilensis HMF14. Proc Natl Acad Sci USA 107:4919–4924
Kurbatov L, Albrecht D, Herrmann H, Petruschka L (2006) Analysis of the proteome of Pseudomonas putida KT2440 grown on different sources of carbon and energy. Environ Microbiol 8:466–478
Lamm SH, Grunwald HW (2006) Benzene exposure and hematotoxicity. Science 312:998
Len ACL, Harty DWS, Jacques NA (2004) Stress-responsive proteins are upregulated in Streptococcus mutans during acid tolerance. Microbiology 150:1339–1351
Lievens B, Hallsworth JE, Pozo MI, Belgacem ZB, Stevenson A, Willems KA, Jacquemyn H (2015) Microbiology of sugar-rich environments: diversity, ecology, and system constraints. Environ Microbiol 17:278–298
Littlejohns JV, McAuley KB, Daugulis AJ (2010) Model for a solid-liquid stirred tank two-phase partitioning bioscrubber for the treatment of BTEX. J Hazard Mat 175:872–882
McCammick EM, Gomase VS, Timson DJ, McGenity TJ, Hallsworth JE (2010) Water–hydrophobic compound interactions with the microbial cell. In: Timmis KN (ed) Handbook of hydrocarbon and lipid microbiology—hydrocarbons, oils and lipids: diversity, properties and formation, vol 2. Springer, New York, pp 1451–1466
Nelson KE, Paulsen IT, Weinel C, Dodson RJ, Hilbert H et al (2002) Complete genome sequence and comparative analysis of the metabolically versatile Pseudomonas putida KT2440. Environ Microbiol 4:799–808
Oren A, Hallsworth JE (2014) Microbial weeds in saline habitats: the enigma of the weed-like Haloferax mediterranei. FEMS Microbiol Lett 359:134–142
Park SH, Oh KH, Kim CK (2001) Adaptive and cross-protective responses of Pseudomonas sp. DJ-12 to several aromatics and other stress shocks. Curr Microbiol 43:176–181
Radniecki TS, Dolan ME, Semprini L (2008) Physiological and transcriptional responses of Nitrosomonas europaea to toluene and benzene inhibition. Environ Sci Technol 42:4093–4098
Rangel DEN, Braga GUL, Fernandes ÉKK, Keyser CA, Hallsworth JE, Roberts DW (2015a) Stress tolerance and virulence of insect-pathogenic fungi are determined by environmental conditions during conidial formation. Curr Genet 61:383–404
Rangel DEN, Alder-Rangel A, Dadachova E, Finlay RD, Kupiec M, Dijksterhuis J, Braga GUL, Corrochano LM, Hallsworth JE (2015b) Fungal stress biology: a preface to the Fungal Stress Responses special edition. Curr Genet 61:231–238
Rummel JD, Beaty DW, Jones MA, Bakermans C, Barlow NG, Boston PJ, Chevrier VF, Clark BC, de Vera JPP, Gough RV, Hallsworth JE, Head JW, Hipkin VJ, Kieft TL, McEwen AS, Mellon MT, Mikucki JA, Nicholson WL, Omelon CR, Peterson R, Roden EE, Sherwood Lollar B, Tanaka KL, Viola D, Wray JJ (2014) A new analysis of Mars “Special Regions”: findings of the second MEPAG Special Regions Science Analysis Group (SR-SAG2). Astrobiology 14:887–968
Russo A, Palumbo M, Scifo C, Cardile V, Barcellona ML, Renis M (2001) Ethanol-induced oxidative stress in rat astrocytes: role of HSP70. Cell Biol Toxicol 17:153–168
Santos PM, Benndorf D, Sá-Correia I (2004) Insights into Pseudomonas putida KT2440 response to phenol-induced stress by quantitative proteomics. Proteomics 4:2640–2652
Santos R, Stevenson A, de Carvalho CCCR, Grant IR, Hallsworth JE (2015) Extraordinary solute stress tolerance contributes to the environmental tenacity of mycobacteria. Environ Microbiol Rep 7:746–764
Sauer T, Galinski EA (1998) Bacterial milking: a novel bioprocess for production of compatible solutes. Biotechnol Bioeng 57:306–313
Segura A, Godoy P, Van Dillewijn P, Hurtado A, Arroyo N, Santacruz S, Ramos JL (2005) Proteomic analysis reveals the participation of energy- and stress-related proteins in the response of Pseudomonas putida DOT-T1E to toluene. J Bacteriol 187:5937–5945
Sikkema J, de Bont JAM, Poolman B (1995) Mechanisms of membrane toxicity of hydrocarbons. Microbiol Rev 59:201–222
Smirnoff N, Cumbes QJ (1989) Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 28:1057–1060
Stevenson A, Hallsworth JE (2014) Water and temperature relations of soil Actinobacteria. Environ Microbiol Rep 6:744–755
Stevenson A, Cray JA, Williams JP, Santos R, Sahay R, Neuenkirchen N, McClure CD, Grant IR, Houghton JDR, Quinn JP, Timson DJ, Patil SV, Singhal RS, Anton J, Dijksterhuis J, Hocking AD, Lievens B, Rangel DEN, Voytek MA, Gunde-Cimerman N, Oren A, Timmis KN, McGenity TJ, Hallsworth JE (2015a) Is there a common water-activity limit for the three domains of life? ISME J 9:1333–1351
Stevenson A, Burkhardt J, Cockell CS, Cray JA, Dijksterhuis J, Fox-Powell M, Kee TP, Kminek G, McGenity TJ, Timmis KN, Timson DJ, Voytek MA, Westall F, Yakimov MM, Hallsworth JE (2015b) Multiplication of microbes below 0.690 water activity: implications for terrestrial and extraterrestrial life. Environ Microbiol 17:257–277
Suryawanshi RK, Patil CD, Borase HP, Narkhede CP, Stevenson A, Hallsworth JE, Patil SV (2015) Towards an understanding of bacterial metabolites prodigiosin and violacein and their potential for use in commercial sunscreens Internat. J Cosmet Sci 37:98–107
Tam LT, Eymann C, Albrecht D, Sietmann R, Schauer F, Hecker M, Antelmann H (2006) Differential gene expression in response to phenol and catechol reveals different metabolic activities for the degradation of aromatic compounds in Bacillus subtilis. Environ Microbiol 8:1408–1427
Timmis KN (ed) (2010) Handbook of hydrocarbon and lipid microbiology—hydrocarbons, oils and lipids: diversity, properties and formation, vol 2. Springer, New York
Trevisol ET, Panek AD, Mannarino SC, Eleutherio EC (2011) The effect of trehalose on the fermentation performance of aged cells of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 90:697–704
Velázquez F, Parro V, de Lorenzo V (2005) Inferring the genetic network of m-xylene metabolism through expression profiling of the xyl genes of Pseudomonas putida mt-2. Mol Microbiol 57:1557–1569
Volkers RJM, de Jong AL, Hulst AG, van Baar BLM, de Bont JS, Wery J (2006) Chemostat-based proteomic analysis of toluene-affected Pseudomonas putida S12. Environ Microbiol 8:1674–1679
Wierckx NJP, Ballerstedt H, de Bont JAM, de Winde JH, Ruijssernaars HJ, Wery J (2008) Transcriptome analysis of a phenol-producing Pseudomonas putida S12 construct: genetic and physiological basis for improved production. J Bacteriol 190:2822–2830
Williams JP, Hallsworth JE (2009) Limits of life in hostile environments; no limits to biosphere function? Environ Microbiol 11:3292–3308
Womack AM, Bohannan BJM, Green JL (2010) Biodiversity and biogeography of the atmosphere. Phil Trans R Soc B 365:3645–3653
Wyatt TT, Golovina EA, van Leeuwen MR, Hallsworth JE, Wösten HAB, Dijksterhuis J (2015a) A decrease in bulk water and mannitol and accumulation of trehalose and trehalose-based oligosaccharides define a two-stage maturation process towards extreme stress resistance in ascospores of Neosartorya fischeri (Aspergillus fischeri). Environ Microbiol 17:283–294
Wyatt TT, van Leeuwen MR, Gerwig GJ, Golovina EA, Hoekstra FA, Kuenstner EJ, Palumbo EA, Snyder NL, Visagie C, Verkennis A, Hallsworth JE, Kamerling JP, Wösten HAB, Dijksterhuis J (2015b) Functionality and prevalence of trehalose-based oligosaccharides as novel compatible solutes in ascospores of Neosartorya fischeri (Aspergillus fischeri) and other fungi. Environ Microbiol 17:395–411
Yakimov MM, Lo Cono V, La Spada G, Bortoluzzi G, Messina E, Smedile F, Werner J, Teeling H, Borghini M, Ferrer M, Cray JA, Hallsworth JE, Golyshin PN, Giuliano L (2015) Microbial community of seawater-brine interface of the deep-sea brine Lake Kryos as revealed by recovery of mRNA are active below the chaotropicity limit of life. Environ Microbiol 17:364–382
Yun SH, Kim YH, Joo EJ, Choi JS, Sohn JH, Kim SI (2006) Proteome analysis of cellular response of Pseudomonas putida KT2440 to tetracycline stress. Curr Microbiol 53:95–101
Yun SH, Park GW, Kim JY, Kwon SO, Choi CW, Leem SH, Kwon KH, Yoo JS, Lee C, Kim S, Kim SI (2011) Proteomic characterization of the Pseudomonas putida KT2440 global response to a monocyclic aromatic compound by iTRAQ analysis and 1DE-MudPIT. J Proteomics 74:620–628
Zager RA (1996) Mitochondrial free radical production induces lipid peroxidation during myohemoglobinuria. Kidney Internat 49:741–751
Acknowledgments
This project was carried out within the research program of the Kluyver Centre for Genomics of Industrial Fermentation which is part of the Netherlands Genomics Initiative/Netherlands Organization for Scientific Research, the EU-funded MIFRIEND and LINDANE projects, a Beaufort Marine Research Award for Marine Biodiscovery (Marine Institute, Ireland), and a Biotechnology and Biological Sciences Research Council-funded project (UK; Grant No. BBF0034711) which is part of the Pseudomonas putida Systems Biology of Microorganisms project (PSYSMO). We are grateful for useful discussions with Giuseppe Albano (Edinburgh University, UK), Philip G. Hamill, Luke Morton, Allen Y. Mswaka, Andrew Stevenson, and Dave J. Timson, (Queen’s University Belfast), Victor de Lorenzo (Centro Nacional de Biotecnología, CSIC, Spain), Terry J. McGenity and Jen Waring (University of Essex), Mary Palfreyman (Outwood Grange College, UK), and Kenneth N. Timmis (Helmholtz Centre for Infection Research, Germany).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial/commercial conflicts of interest.
Additional information
Communicated by M. Kupiec.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhaganna, P., Bielecka, A., Molinari, G. et al. Protective role of glycerol against benzene stress: insights from the Pseudomonas putida proteome. Curr Genet 62, 419–429 (2016). https://doi.org/10.1007/s00294-015-0539-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-015-0539-1