Abstract
The discovery and characterization of bacterial carbohydrate-active enzymes is a fundamental component of biotechnology innovation, particularly for renewable fuels and chemicals; however, these studies have increasingly transitioned to exploring the complex regulation required for recalcitrant polysaccharide utilization. This pivot is largely due to the current need to engineer and optimize enzymes for maximal degradation in industrial or biomedical applications. Given the structural simplicity of a single cellulose polymer, and the relatively few enzyme classes required for complete bioconversion, the regulation of cellulases in bacteria has been thoroughly discussed in the literature. However, the diversity of hemicelluloses found in plant biomass and the multitude of carbohydrate-active enzymes required for their deconstruction has resulted in a less comprehensive understanding of bacterial hemicellulase-encoding gene regulation. Here we review the mechanisms of this process and common themes found in the transcriptomic response during plant biomass utilization. By comparing regulatory systems from both Gram-negative and Gram-positive bacteria, as well as drawing parallels to cellulase regulation, our goals are to highlight the shared and distinct features of bacterial hemicellulase-encoding gene regulation and provide a set of guiding questions to improve our understanding of bacterial lignocellulose utilization.
Key points
• Canonical regulatory mechanisms for bacterial hemicellulase-encoding gene expression include hybrid two-component systems (HTCS), extracytoplasmic function (ECF)-σ/anti-σ systems, and carbon catabolite repression (CCR).
• Current transcriptomic approaches are increasingly being used to identify hemicellulase-encoding gene regulatory patterns coupled with computational predictions for transcriptional regulators.
• Future work should emphasize genetic approaches to improve systems biology tools available for model bacterial systems and emerging microbes with biotechnology potential. Specifically, optimization of Gram-positive systems will require integration of degradative and fermentative capabilities, while optimization of Gram-negative systems will require bolstering the potency of lignocellulolytic capabilities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The decomposition of plant biomass plays a significant role in environmental and biotechnological settings (Zhang et al. 2020). As the largest source of renewable carbon on the planet, the deconstruction of its polysaccharide components is heavily studied (Von Freiesleben et al. 2018; Michalak et al. 2020; Mhatre et al. 2022). Plant cell wall polysaccharides are broadly classified as either cellulose or hemicellulose. Cellulose polymers are exclusively comprised of glucose with a single linkage type (Gardner and Blackwell 1974). Alternatively, hemicelluloses possess greater linkage and sugar varieties which can include xyloglucans, xylans, mannans, arabinans, and pectins (Hoch 2007). This diversity in linkage and sugar type contributes to the insolubility and recalcitrance of plant cell walls, making them difficult to degrade (Holland et al. 2020).
Environmental bacteria and fungi are the central decomposers of this material (Pascoal et al. 2021), and produce carbohydrate-active enzymes (CAZymes) for its deconstruction (Henrissat et al. 2022). Considerable biochemical and bioinformatic research has organized CAZymes into classes and families based on amino acid sequences and are documented in the CAZy database (Drula et al. 2022). This resource has facilitated efforts to predict and sort novel CAZymes for evolutionary phylogeny studies of lignocellulose degradation (Aspeborg et al. 2012; Wu et al. 2023), as well as enzyme engineering for industrial applications (Chettri and Verma 2023; Jayachandran et al. 2023).
As bacterial lignocellulose degradation systems become more fully described, work has branched out to several new areas to include the regulation of CAZyme-encoding genes. While cellulase systems in both Gram-negative and Gram-positive bacteria have been reviewed (Liu et al. 2021; Ziles Domingues et al. 2022), there have been much fewer for hemicellulase systems because of the large number of substrates and enzymes required, as well as the assertion that carbon catabolite repression (CCR) is the dominant modulator of gene expression (Stülke and Hillen 1999). Despite these challenges, recent hemicellulase-encoding gene regulation studies have characterized novel systems that were leveraged to engineer a single bacterium capable of fully degrading and fermenting lignocellulose (Mhatre et al. 2022; Singhania et al. 2022).
The goal of this review is to consolidate previously summarized work for a single phyla (Grondin et al. 2017; Lee et al. 2020) and provide commentary on the current direction of regulation-based studies for genes encoding hemicellulases like xyloglucanases, xylanases, mannanases, arabinanases, and pectinases in both Gram-negative and Gram-positive bacteria. Furthermore, this review discusses the breadth of knowledge regarding CAZyme-encoding gene regulatory systems to include the recent influx of transcriptomic and computational studies that predict regulons specific to hemicellulase-encoding genes. We conclude with a few open questions and offer suggestions on promising future directions for studying the regulation of hemicellulase-encoding genes that may be of environmental or industrial interest.
Canonical regulatory mechanisms for bacterial hemicellulase-encoding gene expression
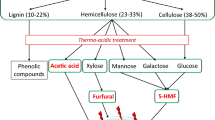
Expression of CAZyme-encoding genes requires precise regulation to ensure efficient energy expenditure under specific nutrient conditions. Despite the multitude of mechanisms that bacteria employ to regulate gene expression, there are only three systems commonly used for CAZyme-encoding genes, specifically hybrid two-component systems, extracytoplasmic function-σ/anti-σ systems, and carbon catabolite repression (Fig. 1). Given that these regulatory systems have been comprehensively reviewed previously (Liu et al. 2019; Pinto et al. 2019; Franzino et al. 2022), we will only briefly summarize each of their general functions and the current knowledge on these systems that is relevant for the expression of hemicellulase-encoding genes.
Common regulatory systems for Carbohydrate-Active Enzyme (CAZyme) encoding genes in Gram-positive and Gram-negative bacteria. A Hybrid two-component system in Bacteroides thetaiotaomicron. Upon sensing of arabinoxylan from the transmembrane domain, the intracellular histidine kinase (HK) phosphorylates the associated response regulator (RR) which recruits RNA polymerase for gene transcription. B ECF-σ/anti-σ factor system in Bacteroides xylanisolvens. Binding of arabinoxylan to the carbohydrate domain of the transmembrane ECF protein releases the intracellular σ factor from the membrane-attached anti-σ factor which aids RNA polymerase in gene transcription. C Carbon catabolite repression in Gram-negative Escherichia coli. In the absence of glucose, phosphorylated EIIA accumulates and activates adenylate cyclase (AC) via phosphorylation, which generates high cAMP levels. The cAMP subsequently binds to the cAMP receptor protein (CRP) and initiates transcription of hemicellulase-encoding genes. D Carbon catabolite repression in Gram-positive Bacillus subtilis. In the absence of glucose, fructose 1,6-biphosphate is not generated because glycolysis does not occur. Without fructose 1,6-biphosphate, histidine protein (HPr) does not get phosphorylated and therefore cannot dimerize with the carbon catabolite control protein (CcpA). Without this dimerization, the coupled protein cannot inhibit transcription. For all panels, phosphate is shown as a gold circle with a “P”; arabinoxylan is shown with orange stars for xylose and the green stars for arabinose” fructose is shown as a green pentagon. Model generated with BioRender.com
Hybrid two-component systems
Hybrid two-component systems (HTCS) in bacteria use a sensing/phosphorylation relay mechanism to up-regulate genes involved in antibiotic resistance, virulence, biofilm formation, quorum sensing, and carbohydrate metabolism (Gutu et al. 2013; Cui et al. 2018; Gellatly et al. 2018; Kampik et al. 2020). This system, which is found in both Gram-negative and Gram-positive bacteria, recognizes an external stimulus with a cytoplasmic-membrane protein that initiates a phosphorylation cascade to modulate gene expression (Howell et al. 2003). As shown in Fig. 1A, a substrate binds the sensor domain of a transmembrane histidine kinase. Substrate binding initiates a phosphate transfer from ATP to a histidine residue on the cytoplasmic domain. The phosphorylated histidine kinase then transfers the phosphate to a response regulator which binds the transcriptional start site of interest to modulate transcription (Buschiazzo and Trajtenberg 2019; Francis and Porter 2019). It should be noted that there are examples of much lengthier phospho-relays with additional histidine kinases and response regulators before RNA polymerase recruitment. Two specific examples can be found in Bacteroides thetaiotaomicron and Bacillus cereus for glycan utilization and stress response, respectively (Been et al. 2006; Sonnenburg et al. 2006).
Previous research on hybrid two-component systems characterized the regulation of genes encoding xylanases, glucanases, arabinanases, and esterases from a diverse set of Gram-negative and Gram-positive bacteria (Emami et al. 2009; Martens et al. 2011; Shulami et al. 2014; Kampik et al. 2020). For example, in Gram-negative Cellvibrio japonicus, Bacteroides thetaiotaomicron, and Gram-positive Ruminiclostridium cellulolyticum, it was noted that HTCS regulators induced expression for biochemically or physiologically important xylanase-, arabinosidase-, and esterase-encoding genes (Emami et al. 2009; Martens et al. 2011; Kampik et al. 2020). The characterized HTCSs associated with xylanase and arabinanase-encoding gene expression are now cataloged as response regulators belonging to the AraC/XylS family of transcriptional activators (Emami et al. 2009; Celik et al. 2013). This family has recently been reviewed and is categorized based on two characteristic helix-turn-helix DNA-binding motifs (Cortes-Avalos et al. 2021). Regulation predominantly occurs via activation when the phosphorylated regulator binds to a recognized -10 and -35 consensus sequence upstream of the promoter for RNA polymerase recruitment (Celik et al. 2013). The sensing domains of these HTCS bind branched xylo-oligosaccharides or arabino-oligosaccharides in the periplasmic space for Gram-negative bacteria (Emami et al. 2009; Schwalm et al. 2017) and extracellularly for Gram-positive bacteria (Lansky et al. 2020). For the former, species like C. japonicus and B. thetaiotaomicron require an efficient mechanism to degrade extracellular hemicellulose into oligosaccharides and transport them to the periplasm where they can be sensed by the corresponding HTCS. It is therefore unsurprising that these two species possess a disproportionally high number of outer membrane transporters that can bring large complex oligosaccharides into the periplasm (Emami et al. 2009; Larsbrink et al. 2014; Blake et al. 2018; Pollet et al. 2021).
Extracytoplasmic function (ECF)-σ/anti-σ systems
Similar to HTCS, extracytoplasmic function (ECF)-σ/anti-σ systems are also comprised of a membrane-spanning sensory protein with a cytoplasmic regulatory protein partner that controls gene expression, with specific roles in bacterial virulence, stress response, and carbohydrate catabolism (Sun et al. 2004; Alvarez-Martinez et al. 2007; Wang et al. 2019a). ECF-σ/anti-σ systems are found in both Gram-negative and Gram-positive bacteria, but have been most well-characterized in Actinobacteria and human gut symbionts belonging to the Bacteroides phylum (Martens et al. 2009; Bahari et al. 2011; Huang et al. 2015; Despres et al. 2016a; Wang et al. 2019a). The anti-σ element of the system is a protein in the cytoplasmic membrane that binds a cytoplasmic ECF-σ protein (Helmann 2002) (Fig. 1B). Release of the ECF-σ protein occurs upon substrate binding, which can be a glycan, metal, or chemical stressor like limonene (Pudio et al. 2015; Marcos-torres et al. 2016; Goutam et al. 2017). The freed σ-factor then binds to RNA polymerase, forming a holoenzyme, and initiates transcription after binding a recognized consensus mRNA sequence (Bae et al. 2015).
In the context of carbohydrate catabolism, ECF-σ/anti-σ systems are prominent regulators in human gut symbionts, especially for the expression of genes encoding O-glycan-degrading enzymes (Martens et al. 2008). ECF-σ/anti-σ systems in Bacteroides species also frequently regulate genes encoding TonB-dependent transporters (e.g., SusC/D) (Martens et al. 2009). Furthermore, Gram-negative Cytophaga hutchinsonii and Gram-positive Clostridium thermocellum also have well-characterized ECF-σ/anti-σ systems that regulate cellulase-encoding gene expression (Nataf et al. 2010; Sand et al. 2015; Wang et al. 2019a). In C. thermocellum, cellulosomes are assembled using at least six ECF-σ/anti-σ systems that are specific for distinct cellulolytic regulons (Ortiz de Ora et al. 2018; Ichikawa et al. 2022).
In contrast to what is known about ECF-σ/anti-σ system to control cellulase-encoding genes, the regulatory involvement of ECF-σ/anti-σ systems for hemicellulase-encoding genes is less understood. Using the best-described examples from Actinobacteria, ECF-σ/anti-σ systems have been placed into families based on the regulons they control (Huang et al. 2015). For example, ECF families 52 and 53 have been computationally predicted to possess a C-terminal fusion domain comprised of the anti-sigma factor sequence coupled with a transmembrane portion of the protein (Marcos-Torres et al. 2022). More interestingly, some ECF52 and ECF53 proteins also have computationally predicted glycosyl hydrolase catalytic domains and carbohydrate-binding domains (Huang et al. 2015; Pinto et al. 2019); however, experimental validation has yet to be performed. In C. thermocellum, xylanase-encoding genes are regulated by alternative sigma factors σI6 and σI7 and the vegetative promoter σA (Sand et al. 2015; Ichikawa et al. 2022). It was demonstrated that the vegetative σA provided basal expression of xylanase-encoding genes, while σI6 and σI7 were employed for stronger expression in the presence of xylans (Bahari et al. 2011; Sand et al. 2015). Furthermore, the characterization of C. thermocellum ECF-σ/anti-σ systems aided in the prediction of homologous regulators in related species like Psuedobacteroides cellulosolvens, specifically for a pectin-degrading regulon (Ortiz de Ora et al. 2018).
Carbon catabolite repression
The final canonical system, carbon catabolite repression (CCR), is widely known for controlling the preferential utilization of specific carbon sources (typically glucose) over others (Ammar et al. 2018). In contrast to HTCS and ECF systems, which work similarly in Gram-negative and Gram-positive bacteria, the CCR mechanism in Gram-negative is markedly different compared to Gram-positive bacteria (Kundig et al. 1964; Deutscher and Saier 1983). In Gram-negative bacteria, a phosphotransferase system is utilized wherein glucose is imported intracellularly and simultaneously phosphorylated by a component of the transport protein (EIIA). Expression of non-glucose metabolizing genes has very low basal expression and requires activation (Fig. 1C). A phosphorylated EI protein transfers a phosphate group to the HPr protein, which in turn phosphorylates EIIA. In the absence of glucose, there is an abundance of phosphorylated EIIA (EIIA ~ P), which activates adenylate cyclase (AC) via phosphorylation (Magasanik 1961; Feucht and Saier 1980). The resulting accumulation of cAMP activates the cAMP receptor protein (CRP) and increases the transcription of genes that encode the proteins responsible for the metabolism of non-preferred carbon sources.
In Gram-positive bacteria, expression of genes important to the metabolism of non-glucose sugars requires inactivation of the repressor catabolite control protein (CcpA) (Fig. 1D). This occurs in the absence of glucose wherein fructose 1,6-bisphosphate (FBP) is not generated because glycolysis is not occurring. Without FBP, histidine protein (HPr) cannot be phosphorylated and dimerized with CcpA to repress the transcription of genes involved in metabolizing non-preferred carbon sources (Deutscher and Saier 1983). It should be noted that CcpA can also act as a transcriptional activator for quorum sensing (trpA), stress response (cidAB), and export of excess carbon (ackA) in Streptococcus pneumoniae, Streptococcus mutans, and Bacillus subtilis respectively (Henkin 1996; Kim et al. 2019a). Additionally, other counter-examples of CCR in Pseudomonas sp. found preferential utilization of succinate, citrate, or aromatic compounds over glucose (Liu 1952; Basu et al. 2006).
One example of CCR-based regulation for hemicellulase-encoding genes is found in Bacillus subtilis and uses both CcpA and the repressor GmuR (Sadaie et al. 2008). Mannanase-encoding genes in B. subtilis are in an operon that also contains genes encoding substrate-specific transporters and metabolic enzymes. In the presence of cellobiose or mannobiose (and in the absence of glucose), expression of the mannan utilization operon occurs due to a lack of fructose 1,6-bisphosphate. This results in limited amounts of phosphorylated HPr, which is necessary for CcpA binding to the promoter region. Consequently, the mannanase-encoding genes are de-repressed. Mannanase-encoding genes are further regulated by the repressor GmuR, which requires phosphorylation via GmuA, a component protein of the phosphotransferase system (PTS) and a structural homolog to EIIA (Sadaie et al. 2008). Briefly, glucomannan disaccharides are imported and phosphorylated via the PTS (comprised of transport proteins GmuABC). Inverse to the processes described for carbon catabolite repression, the presence of glucomannan oligosaccharides results in an abundance of unphosphorylated GmuA. Consequently, GmuR cannot be phosphorylated, which results in the transcription of mannanase-encoding genes.
Co-regulation of arabinanase and xylanase-encoding genes are found in Gram-negative and Gram-positive bacteria, with two characterized repressors being AraR and XylR (Laikova et al. 2001; Rodionov et al. 2001). Both belong to the LacI family of transcriptional regulators and work in conjunction with CCR (Book et al. 2016; Ohashi et al. 2021; Rodionov et al. 2021). Co-regulation of xylanase and arabinanase genes provides an efficient means of streamlining gene expression given the monosaccharide composition of lignocellulose, namely hexoses coming from cellulose and pentoses coming from hemicellulose (Jamander et al. 2014; Kim et al. 2015). Not surprisingly, CCR has been widely studied to characterize the regulation of lignocellulose-derived sugar metabolism in Clostridium, Caldicellulosiruptor, Pseudomonas, and Escherichia species (Gosset 2005; Vanfossen et al. 2009; Bruder et al. 2015; Liu et al. 2015).
Current applications of canonical systems
The use of bacteria as lignocellulose bioprocessors has renewed interest in the three canonical regulatory mechanisms for biotechnologically relevant bacteria (Mearls et al. 2015; Taylor et al. 2018; Elmore et al. 2020; Ling et al. 2022). Using HTCS and ECF-σ/anti-σ systems, recent studies have focused on the regulation of polysaccharide utilization loci (PULs) containing hemicellulase-encoding genes, especially in Bacteroides sp. (Luis et al. 2018; Mackie and Cann 2018; Pereira et al. 2021; Beidler et al. 2023). Similarly, C. thermocellum is commonly used to study ECF-σ/anti-σ systems due to it possessing unique σI factors that can be studied heterologously in B. subtilis (Munoz-Gutierrez et al. 2016). Comparative studies of C. thermocellum σI factors were also important to the discovery that transcriptional initiation of cellulosomal genes relied on an auto-proteolysis system for ECF upon binding to the glycan of interest (Chen et al. 2023a). Likewise, dismantling CCR-related mechanisms in biotechnologically relevant bacteria (e.g., E. coli, C. thermocellum, and P. putida) found that co-utilization of xylose and glucose is more easily achieved with intracellular cellobiose hydrolysis (Xiong et al. 2018; Wang et al. 2019b; Cabulong et al. 2021). Intracellular cellobiose hydrolysis and phosphorylation bypassed some of the inhibitory effects caused by bacterial sensing/detection of extracellular glucose. Moreover, Pseudomonas putida KT2440 has undergone extensive engineering to co-metabolize glucose with cellobiose, galactose, xylose, and arabinose (Dvorak and de Lorenzo 2018; Peabody et al. 2019; Elmore et al. 2020).
Transcriptomic approaches to identify hemicellulase-encoding gene regulatory patterns
The use of transcriptomic data to assess global changes in CAZyme-encoding gene regulation has rapidly become a standard approach to identify critical components of polysaccharide degradation (Gruninger et al. 2018; Lillington et al. 2020; Chen et al. 2023b). This method is particularly useful for non-model bacterial systems whose regulatory mechanisms are less characterized compared to E. coli or B. subtilis. While it should be noted that CAZyme-encoding gene expression was previously known to be regulated by growth rate and bacterial life cycle for Bacteroides succinogenes and Clostridium thermocellum (Russell 1987; Rydzak et al. 2012), more recent reports have uncovered unique differences in hemicellulase-encoding gene regulation for both Gram-positive and Gram-negative bacteria. Below is a summarization of the recent developments using transcriptomics to elucidate regulatory features in lignocellulose-degrading bacteria.
Hemicellulase gene expression in Gram-positive species
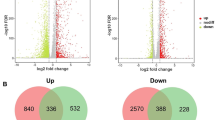
Current RNAseq analyses using Gram-positive bacteria grown on hemicelluloses have often revealed highly specific gene expression responses (Blumer-schuette et al. 2017; La Rosa et al. 2019; Rodionov et al. 2021). For example, the human gut symbiont Roseburia intestinalis has a substrate-specific response during growth on glucomannan and galactomannan (Fig. 2A) (La Rosa et al. 2019). Notably, 16 up-regulated genes were from two distinct mannan utilization loci that differ from PULs described in Bacteroides by the absence of genes that encode TonB-dependent transporters. Additionally, R. intestinalis growth on galactose (a component of galactomannan) did not result in up-regulation of any of these genes, suggesting that mannose or manno-oligosaccharides were the sole nutritional signal for mannan deconstruction (La Rosa et al. 2019).
Differences in up-regulation of CAZyme-encoding genes from selected Gram-positive and Gram-negative bacteria when grown using hemicelluloses. A CAZyme-encoding gene expression of Gram-positive Roseburia intestinalis and Gram-negative Cellvibrio japonicus on glucomannan. B CAZyme-encoding gene expression response of Gram-positive Caldicellulosiruptor bescii and Gram-negative Roseithermus sacchariphilus on xylan. C CAZyme-encoding gene expression response of Gram-positive Caldicellulosiruptor saccharolyticus and Gram-negative Bacteroides xylanisolvens on pectin
Highly specific CAZyme-encoding gene regulation has been observed in Bacillus sp. N16-5, where up-regulation of β-mannanase and α-galactosidase-encoding genes was only observed when the bacterium was grown using galactomannan, but not on xylan, pectin, CMC, or any tested monosaccharide (glucose, fructose, mannose, galactose, arabinose, or xylose) (Song et al. 2013). Furthermore, Bacillus sp. N16-5 grown using xylan only up-regulated β-xylanase-encoding genes, but growth on xylan or xylose up-regulated xylulokinase and xylose-related transporter-encoding genes.
As a third example, in Caldicellulosirupter species like C. bescii and C. saccharolyticus, xylanase-encoding genes were strongly up-regulated during growth on xylan (Fig. 2B) but repressed on either xylose or cellulose (Blumer-schuette et al. 2017; Rodionov et al. 2021). Expression data of C. bescii when grown using xylan also identified a putative key xylanase for extracellular xylan degradation (Xyn11A-2) (Crosby et al. 2022); however, a comparison of enzymatic activity between the C. bescii xylanases showed relatively mediocre activity for Xyn11A-2. The authors suspect this observed difference in gene expression could be a compensatory mechanism to overcome the modest activity of Xyn11A-2. The use of transcriptomic data from C. bescii when grown on xylan has also proven useful for pairing the important degradative loci to their likely regulators, which included XynR, XylR, AraR, BxgRS, and AxuRS (Rodionov et al. 2021). Interestingly, transcriptomic analysis of C. saccharolyticus grown using pectin found a much broader gene expression response than that observed on other hemicelluloses (Blumer-schuette et al. 2017). Growth of C. saccharolyticus using pectin elicited up-regulation of various CAZyme-encoding genes, including cellulases, mannanases, xylanases, arabinanases, pectinases, and chitinases (Fig. 2C).
As a final example, Clostridium sp. exhibited some divergence in their regulatory circuits for xylanase-encoding genes (Petit et al. 2015; Munir et al. 2016). The expression of xylanase-encoding genes possessed by C. termitidis was dependent on xylan, but not xylose, cellobiose, or cellulose, while those belonging to C. phytofermentans were up-regulated when grown on both xylan and cellulose. Alongside the differences in hemicellulase gene expression observed between growth media, growth rate is also a critical mediator of CAZyme gene expression in Clostridium sp., with several studies reporting C. thermocellum transcription of cellulase-encoding genes dependent upon growth phase (Dror et al. 2003; Riederer et al. 2011). One interesting exception was for a xylanase-encoding gene (xynC), which exhibited high expression irrespective of growth rate (Dror et al. 2005).
Hemicellulase gene expression in Gram-negative species
For Gram-negative bacterial species, transcriptomic studies have revealed much broader gene expression responses than those observed in Gram-positive bacteria (Blake et al. 2018; Chen et al. 2018; Novak and Gardner 2023). For example, in Leeuwenhoekiella sp. MAR_2009_132 and Salegentibacter sp. Hel_I_6, up-regulated α- and β-mannanase-encoding genes were identified when these bacteria were grown on both α- or β-mannan despite the selective activity of these CAZymes for each substrate (Chen et al. 2018). This suggested that these species regulate mannanase gene expression with less specificity, possibly at the level of the mannose monosaccharide given that these bacteria cannot differentiate between α- versus β-mannan.
A broad gene expression response was revealed in the saprophyte Cellvibrio japonicus when grown on glucomannan (Fig. 2A) (Novak and Gardner 2023). Eight of the ten predicted mannanase-encoding genes were up-regulated, as well as an additional 46 CAZyme-encoding genes. Strong up-regulation of non-substrate specific CAZyme-encoding genes in C. japonicus suggests that it is likely the presence of complex polysaccharides that induce gene expression. Additionally, a previous study of the C. japonicus transcriptomic response on cellobiose also resulted in broader up-regulation of cellulases and hemicellulases (Nelson et al. 2017). Interestingly, a much more specific response was elicited when C. japonicus was grown on oat-spelt xylan (Blake et al. 2018). This report concluded that C. japonicus only up-regulated xylanase genes during mid-exponential growth, though a comparison of the RNAseq from the stationary phase showed up-regulation of genes encoding xylanases, arabinanases, mannanases, and cellulases. In terms of the growth rate affecting CAZyme-encoding gene expression in C. japonicus, it was observed that expression was more prominent during active growth compared to the stationary phase (Blake et al. 2018; Novak and Gardner 2023).
Roseithermus sacchariphilus exhibited a transcriptomic response quite dissimilar to C. japonicus when it was grown on beechwood xylan (Liew et al. 2020). This bacterium had up-regulation of genes encoding cellulases, mannanases, xylanases, arabinanases, pectinases, and other glycosidases (Fig. 2B). Surprisingly, pectinase-encoding genes were the most prominently up-regulated CAZyme-encoding genes when R. sacchariphilus was grown on xylan. The authors hypothesize that the broad response was due to co-expression of genes encoding various glycosidic activities by the same promoter. However, they also suggested that a multi-timepoint transcriptomic analysis could reveal more about the patterns of hemicellulase gene expression.
Finally, expression of CAZyme-encoding genes in Bacteroides xylanisolvens also elicited a broad gene expression response on oat-spelt xylan, with up-regulation of 150 carbohydrate utilization-encoding genes that included all identified PULs for xylan utilization and 15 PULs for starch and pectic metabolism (Despres et al. 2016a). The authors hypothesized that the broad response was from the detection of shared oligosaccharides present in both oat-spelt xylan and pectins (i.e., arabinoside side-chains). However, this response was very different when B. xylanisolvens was grown on citrus pectin and resulted in a much more specific result (Fig. 2C) (Despres et al. 2016b). Here, researchers were able to compare the gene expression response on two different types of pectins and discern the PULs that were most likely to be involved in the degradation of different pectic linkages. Specifically, PUL 2 was suspected to be important to degrading type II rhamnogalacturonan, PUL 13 was likely involved in de-branching arabinose sidechains, and PULs 49 and 50 were the most up-regulated on both pectins and were suspected to be involved in degrading homogalacturonan and type I rhamnogalacturonan, respectively. Additionally, B. xylanisolvens shared the traits observed in other bacterial species with high expression of CAZyme-encoding genes during active growth compared to the stationary phase (Despres et al. 2016b).
Hemicellulase gene expression in bacterial co-culture
There has been increasing interest in the metatranscriptomic of co-cultured bacteria using complex polysaccharide-rich substrates given that environmental lignocellulose degradation is performed by a microbial community. For example, a study of the Gram-positive Butyrivibrio hungatei MB2003 transcriptome during mono- and co-culture with rumen gut symbiont Butyrivibrio proteoclasticus B316 found that in monoculture, B. hungatei was unable to grow on xylan or pectin despite the presence and expression of several hemicellulase-encoding genes (Palevich et al. 2019). Strikingly, when in co-culture with B. proteoclasticus, B. hungatei had a substantial increase in its growth capabilities at the expense of B. proteoclasticus final cell density. Since B. hungatei acts more as a sugar scavenger than a hemicellulose-degrader, its RNAseq results in monoculture unsurprisingly showed marked increases in the expression of many genes important to translation, signal transduction, defensive mechanisms, lipid/amino acid metabolism, and cell wall biogenesis compared to its co-cultured counterpart. During co-culture with B. proteoclasticus, B. hungatei expressed fewer genes overall but exhibited more specificity in the expression of genes encoding for carbohydrate metabolism (e.g., ABC sugar transporters). Interestingly, B. proteoclasticus gene expression was relatively unchanged between mono- and co-culture (excluding a few CAZyme-encoding genes which were up-regulated during co-culture) despite the increase in competition provided by culturing with B. hungatei.
As another example, the Gram-negative gut symbionts P. intestinalis, P. muris, and P. rodentium underwent comparative metatranscriptomic analysis, and the study concluded that P. intestinalis was the most competitive strain due to its distinct up-regulation of PULs encoding xylanase and pectinase-encoding genes when the rat host was given a diet heavy in arabinoxylans (Galvez et al. 2020). The three most up-regulated glycoside hydrolase families in all three species were from GH43, GH2, and GH28. These families contain members able to hydrolyze β-glucan, β-xylan, α-arabinan, and pectic linkages (Lombard et al. 2014).
Co-cultures containing both Gram-positive and Gram-negative species have been used to investigate the bottlenecks of complete lignocellulose bioconversion in the guts of rumen or humans (Leth et al. 2018; Badhan et al. 2022). A recent metatranscriptomic study examined a complex consortium of Gram-positive and Gram-negative gut symbionts in ruminant animals grown in ex vivo batch culture on total tract indigestible residue (TTIR). The primary goal of the study was to assess the bottlenecks in ruminant digestion to uncover mechanisms to enhance the system. Transcripts encoding xylanases were abundant when the micro-community was grown on TTIR, which indicated that heteroxylans and xyloglucans were the primary remaining polysaccharide in the TTIR. It was hypothesized that the sheer quantity of inter- and intramolecular bonds act as a hindrance to enzyme accessibility to the substrate.
Overall, there appears to be a distinguishing difference between the hemicellulose-encoding gene expression patterns in Gram-positive versus Gram-negative bacteria. Specifically, the narrowed specificity of gene expression observed in Gram-positive compared to Gram-negative species. Additionally, investigations of co-culture transcriptomics containing Gram-positive and/or Gram-negative communities on lignocellulose have focused on the interspecies relationships and competition for carbon acquisition (Palevich et al. 2019; Galvez et al. 2020; Badhan et al. 2022). The knowledge obtained from these analyses has subsequently been applied in studies on gut microbiomes and biotechnology applications, specifically for studies that successfully predicted the impact of synthetic gut microbiota on host immune response (Afrizal et al. 2022) and identified patterns in microbe abundance based on diet (Corbin et al. 2023).
Computational prediction of transcriptional regulators using compilations of transcriptomic data
In addition to the plethora of information provided by RNAseq data from a singular dataset, compilations of such data can extrapolate more information on transcriptional regulatory systems using computational methods. For example, transcriptomic compilations with DNA-binding motif studies have predicted extensive transcriptional regulatory networks of several different bacteria (Poudel et al. 2020; Rychel et al. 2020). The known computationally predicted regulons of Gram-negative plant bioprocessors are relatively exclusive to the fermentative bioprocessing bacteria that possess few hemicellulases (Sastry et al. 2019; Lim et al. 2022). However, this approach has yielded interesting results for Gram-positive species. For example in C. thermocellum, a LacI transcriptional regulator (GlyR2) was computationally predicted as important for genes encoding two mannanases (man5A and man26A), a xylanase (clo1313_2530), and two cellulases (clo1313_0413 and clo1313_1425) (Wilson et al. 2017; Hebdon et al. 2021). Previous experimental research on GlyR2 had identified it as a mannobiose-responsive transcriptional repressor with only confirmed regulatory activity on a mannanase-encoding gene (man5A) (Wilson et al. 2017). GlyR2 was hypothesized to have indirect effects on the transcriptional regulation of certain hemicellulose-encoding genes that may require different conditions to de-repress other genes with the recognized binding motif (Hebdon et al. 2021). Additionally, a C. bescii genome analysis and comparison to other Caldicellulosiruptor species improved the organism-specific bioprocessing model through the discovery of 16 key regulators important to the degradation and metabolism of hemicellulose and pectin (Rodionov et al. 2021). It was noted that most of these regulators were involved in the expression of xylanase or pectinase-encoding genes, while genes that encoded cellulases, mannanases, and amylases generally only had one regulator for each CAZyme type. Additionally, the mechanistic regulatory actions of the predicted regulators were overwhelmingly repressive in function with the few activators belonging to the AraC family. Interestingly, the study found that most of these activators were involved in the regulation of pectinase-encoding genes.
Future directions
A thorough understanding of how hemicellulase-encoding genes are regulated is essential to optimize lignocellulose bioprocessing (Chettri et al. 2020). Consequently, detailed studies that include hemicellulase-encoding gene regulation are generally conducted exclusively on well-characterized model bacteria and those already being used as chassis in biotechnology applications (Xiong et al. 2018; Rodionov et al. 2021).
Given that metagenomic and metatranscriptomic data for less characterized lignocellulolytic bacteria with unoptimized systems are available (Dai et al. 2015; Kougias et al. 2018; Lopez-Mondejar et al. 2020) but beyond the scope of this review, we have endeavored to summarize and highlight the current state of hemicellulose-encoding gene regulation patterns between Gram-positive and Gram-negative bacteria.
Overall, we argue there are two critical features of hemicellulase-encoding gene regulation that must be considered for optimization, which are (1) identifying the specific metabolic inducer (often an oligosaccharide), and (2) mitigating the impacts of carbon catabolite repression. Current lignocellulose bioconversion systems typically use Gram-positive species for saccharification and Gram-negative species for fermentation (Dai et al. 2015; Thapa et al. 2019). While it has been previously argued that co-culture or consortia-based bioconversion processes will improve the efficiency and completeness of lignocellulose degradation (Chin et al. 2020; Kumar et al. 2023), the amount of strain engineering and optimization significantly increases for each strain added to the process, especially given the current trend of focusing only on improving either degradative or metabolic/fermentative capabilities. Therefore, the following commentary will focus exclusively on the optimization of single bacterium bioprocessing systems for the complete deconstruction and utilization of lignocellulose (Table 1).
Optimizing Gram-positive systems will require integration of degradative and fermentative capabilities
Clostridia and Caldicellulosiruptor species are highly studied genera for their prolific degradation of plant polymers (Artzi et al. 2018; Brunecky et al. 2018; Williams-Rhaesa et al. 2018). However, neither system has been successfully engineered to fully metabolize and ferment all components of lignocellulose. In the case of Clostridia systems, this is due to an inherent inability to ferment pentoses. A previous attempt to engineer a pathway for xylose fermentation in C. thermocellum found that while xylose and Avicel could be co-utilized, xylan and Avicel could not (Xiong et al. 2018). It was argued that this is likely due to an inhibitory effect posed by cello-oligosaccharides on xylanases or unknown regulators that repress xylanase gene expression in the presence of cello-oligosaccharides. More recently, efforts have transitioned to develop CRISPR/Cas systems or riboswitches (Marcano-Velazquez et al. 2019; Walker et al. 2020) to mediate the observed repression of xylanase gene transcription in the presence of cellodextrins or cellobiose.
In Caldicellulosiruptor systems, the limiting factor is that the expression and degradative efficiency of heterologously expressed CAZymes is low. C. bescii has been extensively manipulated to improve its saccharifying proficiency via heterologous expression of xylanases (Kim et al. 2018; Crosby et al. 2022); however, it has been observed that degraded oligosaccharides repress the expression of secreted enzymes. Additionally, many heterologously expressed genes in C. bescii employ a highly active constitutive promoter, which is unoptimized for lignocellulose bioprocessing due to the energetic output required to constitutively and highly express heterologous CAZyme-encoding genes (Conway et al. 2017; Kim et al. 2017; Lee et al. 2020). Therefore, control over the expression of the heterologously expressed genes could spare the metabolic burden of their high expression levels and improve this limitation.
Optimizing Gram-negative systems will require bolstering the potency of lignocellulolytic capabilities
Gram-negative species elicit a much broader hemicellulase-encoding gene regulatory response than Gram-positive bacteria. We argue that this diversification of CAZyme gene expression is an underutilized resource to optimize lignocellulose bioconversion in single bacterium systems. Biotechnology-relevant model systems like E. coli and P. putida have been largely focused on improving co-utilization of hexoses and pentoses by overcoming the effects of CCR (Kim et al. 2019b; Peabody et al. 2019; Elmore et al. 2020; Cabulong et al. 2021). However, these systems are limited as they are unable to innately degrade lignocellulose. The necessary step needed to drive either model into a fully self-sufficient system is the inclusion of lignocellulolytic machinery. This approach has several obstacles, most pressingly, identifying the minimally sufficient set of CAZymes that can completely depolymerize plant biomass and engineering an efficient export system for these CAZymes from the heterologous host.
In contrast, the genes/proteins needed to ferment plant sugars or produce other bioproducts are known and could be integrated into lignocellulolytic Gram-negative species. One example of a system not yet tapped for industrial use but has to potential to do so is Cellvibrio japonicus, a Gram-negative saprophyte that can fully degrade lignocellulose (Deboy et al. 2008; Gardner et al. 2014; Larsbrink et al. 2014; Blake et al. 2018). C. japonicus has also been shown to make ethanol and rhamnolipids as targeted products from lignocellulose bioconversion on a proof-of-concept scale (Gardner and Keating 2010; Horlamus et al. 2018). Another Gram-negative model is Saccharophagus degradans which also possesses a large number of CAZymes capable of degrading polysaccharides including cellulose, xylan, and pectin (Ensor et al. 1999). Engineering efforts using S. degradans have successfully generated strains capable of producing polyhydroxyalkanoate (PHAs) from cellulose, xylan, and agarose (Esteban Alva Munoz and Riley 2008; Sawant et al. 2017). However, S. degradans cannot generate ethanol and still relies on co-culture with other microbes for its production (Takagi et al. 2016). While both C. japonicus and S. degradans show promise with their degradative ability, improvements to their genetic systems are still needed to heterologously express the necessary metabolic pathways to produce high-value products.
Concluding statement
This review discussed mechanisms that regulate hemicellulase-encoding gene expression in Gram-positive versus Gram-negative bacteria. Experimental studies that characterize the molecular mechanisms of hemicellulase gene expression are useful to identify relevant activators or repressors for each regulon, and we argue that such research is essential for the field to significantly advance. Given the discussed limitations of the reviewed models, the field should prioritize efforts that predict transcriptional regulatory networks and engineer the requisite enzymes for plant sugar bioconversion in species innately capable of prolific lignocellulose degradation.
Data Availability
N/A.
References
Afrizal A, Jennings SAV, Hitch TCA, Riedel T, Basic M, Panyot A, Treichel N, Hager FT, Wong EO-Y, Wolter B, Viehof A, von Strempel A, Eberl C, Buhl EM, Abt B, Bleich A, Tolba R, Blank LM, Navarre WW, Kiessling F, Horz H-P, Torow N, Cerovic V, Stecher B, Strowig T, Overmann J, Clavel T (2022) Enhanced cultured diversity of the mouse gut microbiota enables custom-made synthetic communities. Cell Host Microbe 30:1630–1645. https://doi.org/10.1016/j.chom.2022.09.011
Alvarez-Martinez CE, Lourenço RF, Baldini RL, Laub MT, Gomes SL (2007) The ECF sigma factor σT is involved in osmotic and oxidative stress responses in Caulobacter crescentus. Mol Biol 66:1240–1255. https://doi.org/10.1111/j.1365-2958.2007.06005.x
Ammar EM, Wang X, Rao CV (2018) Regulation of metabolism in Escherichia coli during growth on mixtures of the non-glucose sugars: arabinose, lactose, and xylose. Nat Sci Reports 8:1–11. https://doi.org/10.1038/s41598-017-18704-0
Artzi L, Dadosh T, Milrot E, Levin-zaidman S, Morag E, Bayer EA (2018) Colocalization and disposition of cellulosomes in Clostridium clariflavum as revealed by correlative superresolution imaging. Mbio 9:e00012-18
Aspeborg H, Coutinho PM, Wang Y, Brumer H, Henrissat B (2012) Evolution, substrate specificity and subfamily classification of glycoside hydrolase family 5 (GH5). BMC Evol Biol 12. https://doi.org/10.1186/1471-2148-12-186
Badhan A, Low KE, Jones DR, Xing X, Milani MRM, Polo RO, Klassen L, Venketachalam S, Hahn MG, Abbott DW, McAllister TA (2022) Mechanistic insights into the digestion of complex dietary fibre by the rumen microbiota using combinatorial high-resolution glycomics and transcriptomic analyses. Comput Struct Biotechnol J 20:148–164
Bae B, Feklistov A, Lass-Napiorkowska A, Landick R, Darst SA (2015) Structure of a bacterial RNA polymerase holoenzyme open promoter complex. Elife 4:1–23. https://doi.org/10.7554/eLife.08504
Bahari L, Gilad Y, Borovok I, Bareket HK, Yakir D, Shoham Y, Lamed R, Bayer EA (2011) Glycoside hydrolases as components of putative carbohydrate biosensor proteins in Clostridium thermocellum. J Ind Microbiol Biotechnol 38:825–832. https://doi.org/10.1007/s10295-010-0848-9
Basu A, Apte SK, Phale PS (2006) Preferential utilization of aromatic compounds over glucose by Pseudomonas putida CSV86. Appl Environ Microbiol 72:2226–2230. https://doi.org/10.1128/AEM.72.3.2226
Been M De, Francke C, Moezelaar R, Abee T, Siezen RJ (2006) Comparative analysis of two-component signal transduction systems of Bacillus cereus, Bacillus thuringiensis and Bacillus anthracis. Microbiology 152:3035–3048
Beidler I, Robb CS, Vidal-Melgosa S, Zühlke M-K, Bartosik D, Solanki V, Markert S, Becher D, Schweder T, Hehemann J-H (2023) Marine Bacteroidetes use a conserved enzymatic cascade to digest diatom β-mannan. ISME 17:276–285. https://doi.org/10.1038/s41396-022-01342-4
Blake AD, Beri NR, Guttman HS, Cheng R, Gardner JG (2018) The complex physiology of Cellvibrio japonicus xylan degradation relies on a single cytoplasmic β-xylosidase for xylo-oligosaccharide utilization. Mol Microbiol 107:610–622. https://doi.org/10.1111/mmi.13903
Blumer-schuette SE, Zurawski JV, Conway JM, Khatibi P, Lewis DL, Li Q, Chiang VL, Kelly RM (2017) Caldicellulosiruptor saccharolyticus transcriptomes reveal consequences of chemical pretreatment and genetic modification of lignocellulose. Microb Biotechnol 10:1546–1557. https://doi.org/10.1111/1751-7915.12494
Book AJ, Lewin GR, Mcdonald BR, Takasuka TE, Fox G, Currie CR (2016) Evolution of high cellulolytic activity in symbiotic Streptomyces through selection of expanded gene content and coordinated gene expression. PLoS Biol 1–21. https://doi.org/10.1371/journal.pbio.1002475
Bruder M, Moo-young M, Chung DA, Chou CP (2015) Elimination of carbon catabolite repression in Clostridium acetobutylicum — a journey toward simultaneous use of xylose and glucose. Appl Microbiol Biotechnol 99:7579–7588. https://doi.org/10.1007/s00253-015-6611-4
Brunecky R, Chung D, Sarai NS, Hengge N, Russell JF, Young J, Mittal A, Pason P, Vander WT, Michener W, Shollenberger T, Westpheling J, Himmel ME, Bomble YJ (2018) Biotechnology for biofuels high activity CAZyme cassette for improving biomass degradation in thermophiles. Biotechnol Biofuels 11:1–12. https://doi.org/10.1186/s13068-018-1014-2
Buschiazzo A, Trajtenberg F (2019) Two-component sensing and regulation: how do histidine kinases talk with response regulators at the molecular level? Annu Rev Microbiol 73:507–528. https://doi.org/10.1146/annurev-micro-091018-054627
Cabulong RB, Bañares AB, Nisola GM, Lee W-K, Chung W-J (2021) Enhanced glycolic acid yield through xylose and cellobiose utilization by metabolically engineered Escherichia coli. Bioprocess Biosyst Eng 44:1081–1091. https://doi.org/10.1007/s00449-020-02502-6
Celik H, Blouzard J-C, Voigt B, Becher D, Trotter V, Fierobe H-P, Tardif C, Pages S, De PP (2013) A Two-Component System (XydS/R) Controls the expression of genes encoding CBM6-containing proteins in response to straw in Clostridium cellulolyticum. PLoS ONE 8:e56063. https://doi.org/10.1371/journal.pone.0056063
Chen J, Robb CS, Unfried F, Kappelmann L, Markert S, Song T, Harder J, Avc B, Becher D, Xie P, Amann RI, Hehemann J, Schweder T (2018) Alpha- and beta-mannan utilization by marine Bacteroidetes. Environ Microbiol 20:4127–4140. https://doi.org/10.1111/1462-2920.14414
Chen L, Qu Z, Yu W, Zheng L, Qiao H, Wang D, Wei B, Zhao Z (2023b) Comparative genomic and transcriptome analysis of Bacillus velezensis CL-4 fermented corn germ meal. AMB Express 13:1–12. https://doi.org/10.1186/s13568-023-01510-5
Chen C, Dong S, Yu Z, Qiao Y, Li J, Ding X, Li R, Lin J, Bayer EA, Liu Y, Cui Q, Feng Y (2023a) Essential autoproteolysis of bacterial anti-σ factor RsgI for transmembrane signal transduction. Sci Adv 14. https://doi.org/10.1126/sciadv.adg4846
Chettri D, Verma AK (2023) Biological significance of carbohydrate active enzymes and searching their inhibitors for therapeutic applications. Carbohydr Res 529:108853. https://doi.org/10.1016/j.carres.2023.108853
Chettri D, Verma AK, Verma AK (2020) Innovations in CAZyme gene diversity and its modification for biorefinery applications. Biotechnol Reports 28:1–17. https://doi.org/10.1016/j.btre.2020.e00525
Chin DWK, Lim S, Pang YL, Lam MK (2020) Fundamental review of organosolv pretreatment and its challenges in emerging consolidated bioprocessing. Biofuels Bioprod Biorefining 14:808–829. https://doi.org/10.1002/bbb.2096
Conway JM, McKinley BS, Seals NL, Hernandez D, Khatibi PA, Poudel S, Giannone RJ, Hettich RL, Williams-Rhaesa AM, Lipscomb GL, Adams MWW, Kelly RM (2017) Functional analysis of the glucan degradation locus in Caldicellulosiruptor bescii reveals essential roles of component glycoside hydrolases in plant biomass deconstruction. Appl Environ Microbiol 83:e01828-e1917. https://doi.org/10.1128/AEM.01828-17
Corbin KD, Carnero EA, Dirks B, Igudesman D, Yi F, Marcus A, Davis TL, Pratley RE, Rittmann BE, Krajmalnik-Brown R, Smith SR (2023) Host-diet-gut microbiome interactions in fluence human energy balance: a randomized clinical trial. Nat Commun 14:3161. https://doi.org/10.1038/s41467-023-38778-x
Cortes-Avalos D, Martinez-Perez N, Ortiz-Moncada MA, Juarez-Gonzalez A, Vanos-Vargas AA, Estrada-de los Santos P, Perez-Rueda E, Ibarra JA (2021) An update of the unceasingly growing and diverse AraC/XylS family of transcriptional activators. FEMS Microbiol Lett 45:1–13
Crosby JR, Laemthong T, Bing RG, Zhang K, Tanwee TNN, Lipscomb GL, Rodionov DA, Zhang Y, Adams MWW, Kelly RM (2022) Biochemical and regulatory analyses of xylanolytic regulons in Caldicellulosiruptor bescii reveal genus-wide features of hemicellulose utilization. Appl Environ Microbiol 88:e01302-e1322
Cui C, Yang C, Song S, Fu S, Sun X, Yang L, He F, Zhang L-H, Zhang Y, Deng Y (2018) A novel two-component system modulates quorum sensing and pathogenicity in Burkholderia cenocepacia. Mol Microbiol 108:32–44. https://doi.org/10.1111/mmi.13915
Dai X, Tian Y, Li J, Su X, Wang X, Zhao S, Liu L, Luo Y, Liu D, Zheng H, Wang J, Dong Z, Hu S, Huang L (2015) Metatranscriptomic analyses of plant cell wall polysaccharide degradation by microorganisms in the cow rumen. Appl Environ Microbiol 81:1375–1386. https://doi.org/10.1128/AEM.03682-14
De BM, Francke C, Moezelaar R, Abee T, Siezen RJ (2006) Comparative analysis of two-component signal transduction systems of Bacillus cereus, Bacillus thuringiensis and Bacillus anthracis. Microbiol 152:3035–3048. https://doi.org/10.1099/mic.0.29137-0
Deboy RT, Mongodin EF, Fouts DE, Tailford LE, Khouri H, Emerson JB, Mohamoud Y, Watkins K, Henrissat B, Gilbert HJ, Nelson KE (2008) Insights into plant cell wall degradation from the genome sequence of the soil bacterium Cellvibrio japonicus. J Bacteriol 190:5455–5463. https://doi.org/10.1128/JB.01701-07
Despres J, Forano E, Lepercq P, Comtet-marre S, Jubelin G, Chambon C, Yeoman CJ, Miller MEB, Fields CJ, Martens E, Terrapon N, Henrissat B, White BA, Mosoni P (2016a) Xylan degradation by the human gut Bacteroides xylanisolvens XB1A involves two distinct gene clusters that are linked at the transcriptional level. BMC Genomics 17:1–14. https://doi.org/10.1186/s12864-016-2680-8
Despres J, Forano E, Lepercq P, Comtet-Marre S, Jubelin G, Yeoman CJ, Miller MEB, Fields CJ, Terrapon N, Le BC, Renard CMGC, Henrissat B, White BA, Mosoni P (2016b) Unraveling the pectinolytic function of Bacteroides xylanisolvens using a RNA-seq approach and mutagenesis. BMC Genomics 17:1–14. https://doi.org/10.1186/s12864-016-2472-1
Deutscher J, Saier MH Jr (1983) ATP-dependent protein kinase-catalyzed phosphorylation of a seryl residue in HPr, a phosphate carrier protein of the phosphotransferase system in Streptococcus pyogenes. Proc Natl Acad Sci U S A 80:6790–6794. https://doi.org/10.1073/pnas.80.22.6790
Dror TW, Morag E, Rolider A, Bayer EA, Lamed R, Shoham Y (2003) Regulation of the cellulosomal celS (cel48A) gene of Clostridium thermocellum is growth rate dependent. J Bacteriol 185:3042–3048. https://doi.org/10.1128/JB.185.10.3042
Dror TW, Rolider A, Bayer EA, Lamed R, Shoham Y (2005) Regulation of major cellulosomal endoglucanases of Clostridium thermocellum differs from that of a prominent cellulosomal xylanase. J Bacteriol 187:2261–2266. https://doi.org/10.1128/JB.187.7.2261
Drula E, Garron M, Dogan S, Lombard V, Henrissat B, Terrapon N (2022) The carbohydrate-active enzyme database: functions and literature. Nucleic Acids Res 50:571–577
Dvorak P, de Lorenzo V (2018) Refactoring the upper sugar metabolism of Pseudomonas putida for co-utilization of cellobiose, xylose, and glucose. Metab Eng 48:94–108
Elmore JR, Dexter GN, Salvachúa D, Brien MO, Klingeman DM, Gorday K, Michener JK, Peterson DJ, Beckham GT, Guss AM (2020) Engineered Pseudomonas putida simultaneously catabolizes five major components of corn stover lignocellulose : glucose, xylose, arabinose, p-coumaric acid, and acetic acid. Metab Eng 62:62–71. https://doi.org/10.1016/j.ymben.2020.08.001
Emami K, Topakas E, Nagy T, Henshaw J, Jackson KA, Nelson KE, Mongodin EF, Murray JW, Lewis RJ, Gilbert HJ (2009) Regulation of the xylan-degrading apparatus of Cellvibrio japonicus by a novel two-component system. J Biol Chem 284:1086–1096. https://doi.org/10.1074/jbc.M805100200
Ensor LA, Stosz SK, Weiner RM (1999) Expression of multiple complex polysaccharide-degrading enzyme systems by marine bacterium strain 2–40. J Ind Microbiol Biotechnol 23:123–126
Esteban Alva Munoz L, Riley MR (2008) Utilization of cellulosic waste from tequila bagasse and production of polyhydroxyalkanoate (PHA) bioplastics by Saccharophagus degradans. Agric Biosyst Eng 100:882–888. https://doi.org/10.1002/bit.21854
Feucht BU, Saier MH (1980) Fine control of adenylate cyclase by the phosphoenolpyruvate: sugar phosphotransferase systems in Escherichia coli and Salmonella typhimurium. J Bacteriol 141:603–610. https://doi.org/10.1128/jb.141.2.603-610.1980
Francis VI, Porter SL (2019) Multikinase networks : two-component signaling networks integrating multiple stimuli. Annu Rev Microbiol 13:1–25
Franzino T, Boubakri H, Cernava T, Abrouk D, Achouak W, Reverchon S, Nasser W, Haichar FZ (2022) Implications of carbon catabolite repression for plant-microbe interactions. Plant Commun 3:1–21. https://doi.org/10.1016/j.xplc.2021.100272
Galvez EJ, Iljazovic A, Amend L, Lesker TR, Renault T, Thiemann S, Hao L, Roy U, Gronow A, Charpentier E, Strowig T (2020) Distinct polysaccharide utliization determines interspeccies competition between intestinal Prevotella spp. Cell Host Microbe 28:838–852
Gardner KH, Blackwell J (1974) The structure of native cellulose. Biopolymers 13:1975–2001
Gardner J, Keating DH (2010) Requirement of the type II secretion system for utilization of cellulosic substrates by Cellvibrio japonicus. Appl Environ Microbiol 76:5079–5087. https://doi.org/10.1128/AEM.00454-10
Gardner JG, Crouch L, Labourel A, Forsberg Z, Bukhman YV, Vaaje-kolstad G, Gilbert HJ, Keating DH (2014) Systems Biology Defines the Biological Significance of Redox-Active Proteins during Cellulose Degradation in an Aerobic Bacterium 94:1121–1133. https://doi.org/10.1111/mmi.12821
Gellatly SL, Bains M, Breidenstein EBM, Strehmel J, Reffuveille F, Taylor PK, Yeung ATY, Overhage J, Hancock REW (2018) Novel roles for two-component regulatory systems in cytotoxicity and virulence-related properties in Pseudomonas aeruginosa. AIMS Microbiol 4:173–191. https://doi.org/10.3934/microbiol.2018.1.173
Gosset G (2005) Improvement of Escherichia coli production strains by modification of the phosphoenolpyruvate: sugar phosphotransferase system. Microb Cell Fact 4:1–11. https://doi.org/10.1186/1475-2859-4-14
Goutam K, Gupta AK, Gopal B (2017) The fused SnoaL 2 domain in the Mycobacterium tuberculosis sigma factor oJ modulates promoter recognition. Nucleic Acids Res 45:9760–9772. https://doi.org/10.1093/nar/gkx609
Grondin JM, Tamura K, Déjean G, Abbott DW, Brumer H (2017) Polysaccharide utilization loci: fueling microbial communities. J Bacteriol 199:1–15
Gruninger RJ, Nguyen TT, Reid ID, Yanke JL, Wang P, Abbott DW, Tsang A, Mcallister T (2018) Application of transcriptomics to compare the carbohydrate active enzymes that are expressed by diverse genera of anaerobic fungi to degrade plant cell wall carbohydrates. Front Microbiol 9:1–15. https://doi.org/10.3389/fmicb.2018.01581
Gutu AD, Sgambati N, Strasbourger P, Brannon MK, Jacobs MA, Haugen E, Kaul RK, Johansen HK, Hoiby N, Moskowitz SM (2013) Polymyxin resistance of Pseudomonas aeruginosa phoQ mutants is dependent on additional two-component regulatory systems. Antimicrob Agents Chemother 57:2204–2215. https://doi.org/10.1128/AAC.02353-12
Hebdon SD, Gerritsen AT, Chen Y, Marcano JG, Chou KJ (2021) Genome-wide transcription factor DNA binding sites and gene regulatory networks in Clostridium thermocellum. Front Microbiol 12:1–21. https://doi.org/10.3389/fmicb.2021.695517
Helmann JD (2002) The extracytoplasmic function (ECF) sigma factors. Adv Microb Physiol 46:47–110
Henkin TM (1996) The role of the CcpA transcriptional regulator in carbon metabolism in Bacillus subtilis. FEMS Microbiol Lett 135:9–15
Henrissat B, Terrapon N, Coutinho PM, Lombard V, Drula E, Garron M-L, Hornung B (2022) Carbonydrate-active enZYmes database. http://www.cazy.org/. Accessed Oct 2023
Hoch G (2007) Cell wall hemicelluloses as mobile carbon stores in non-reproductive plant tissues. Funct Ecol 21:823–834. https://doi.org/10.1111/j.1365-2435.2007.01305.x
Holland C, Ryden P, Edwards CH, Grundy MML (2020) Plant cell walls : impact on nutrient bioaccessibility. MDPI Foods 9:1–16
Horlamus F, Wittgens A, Noll P, Michler J, Muller I, Weggenmann F, Oellig C, Rosenau F, Henkel M, Hausmann R (2018) One - step bioconversion of hemicellulose polymers to rhamnolipids with Cellvibrio japonicus: a proof-of-concept for a potential host strain in future bioeconomy. Glob Chang Biol Bioenergy 11:260–268. https://doi.org/10.1111/gcbb.12542
Howell A, Dubrac S, Krogh K, Noone D, Fert J, Msadek T, Devine K (2003) Genes controlled by the essential YycG / YycF two- component system of Bacillus subtilis revealed through a novel hybrid regulator approach. Mol Microbiol 49:1639–1655. https://doi.org/10.1046/j.1365-2958.2003.03661.x
Huang X, Pinto D, Fritz G, Mascher T (2015) Environmental sensing in actinobacteria: a comprehensive survey on the signaling capacity of this phylum. J Bacteriol 197:2517–2535. https://doi.org/10.1128/JB.00176-15
Ichikawa S, Ito D, Asaoka S, Abe R, Katsuo N, Ito T, Ito D, Karita S (2022) The expression of alternative sigma-I7 factor induces the transcription of cellulosomal genes in the cellulolytic bacterium Clostridium thermocellum. Enzyme Microb Technol 156:110002. https://doi.org/10.1016/j.enzmictec.2022.110002
Jamander J, Hallstrom BM, Larsson G (2014) Simultaneous uptake of lignocellulose-based monosaccharides by Escherichia coli. Biotechnol Bioeng 11:1108–1115. https://doi.org/10.1002/bit.25182
Jayachandran D, Smith P, Irfan M, Sun J, Yarborough JM, Bomble YJ, Lam E, Chundawat SPS (2023) Engineering and characterization of carbohydrate-binding modules for imaging cellulose fibrils biosynthesis in plant protoplasts. Biotechnol Bioeng 120:2253–2268. https://doi.org/10.1002/bit.28484
Kampik C, Denis Y, Pages S, Perret S, Tardif C, Fierobe H-P, de Philip P (2020) A novel two-component system, XygS/XygR, positively regulates xyloglucan degradation, import, and catabolism in Ruminiclostridium cellulolyticum. Appl Environ Microbiol 86:e01357-e1420
Kim SM, Choi BY, Ryu YS, Jung SH, Park JM, Kim G-H, Lee SK (2015) Simultaneous utilization of glucose and xylose via novel mechanisms in engineered Escherichia coli. Metab Eng 30:141–148. https://doi.org/10.1016/j.ymben.2015.05.002
Kim SK, Chung D, Himmel ME, Bomble YJ, Westpheling J (2017) Heterologous expression of a β-d-glucosidase in Caldicellulosiruptor bescii has a surprisingly modest effect on the activity of the exoproteome and growth on crystalline cellulose. J Ind Microbiol Biotechnol 44:1643–1651. https://doi.org/10.1007/s10295-017-1982-4
Kim S-K, Himmel ME, Bomble YJ, Westpheling J (2018) Expression of a cellobiose phosphorylase from Thermotoga maritima in Caldicellulosiruptor bescii improves the phosphorolytic pathway and results in a dramatic increase in cellulolytic activity. Appl Environ Microbiol 84:e02348-e2417. https://doi.org/10.1128/AEM.02348-17
Kim H, Waters A, Turner ME, Rice KC, Ahn S (2019a) Regulation of cid and lrg expression by CcpA in Streptococcus mutans. Microbiology 165:113–123. https://doi.org/10.1099/mic.0.000744
Kim J, Tremaine M, Grass JA, Purdy HM, Landick R, Kiley PJ, Reed JL (2019b) Systems metabolic engineering of Escherichia coli improves co-conversion of lignocellulose-derived sugars. Biotechnol J 14. https://doi.org/10.1002/biot.201800441
Kougias PG, Campanaro S, Treu L, Tsapekos P, Armani A, Angelidaki I (2018) Spatial distribution and diverse metabolic functions of lignocellulose-degrading uncultured bacteria as revealed by genomic-centric metagenomics. Appl Environ Microbiol 84:e01244-e1318
Kumar V, Fox BG, Takasuka TE (2023) Consolidated bioprocessing of plant biomass to polyhydroxyalkanoate by co-culture of Streptomyces sp. SirexAA-E and Priestia Megaterium Bioresour Technol 376:128934. https://doi.org/10.1016/j.biortech.2023.128934
Kundig W, Ghosh S, Roseman S (1964) Phosphate bound to histidine in a protein as an intermediate in a novel phospho-transferase system. Proc Natl Acad Sci U S A 52:1067–1074. https://doi.org/10.1073/pnas.52.4.1067
La Rosa SL, Leth ML, Michalak L, Hansen ME, Pudlo NA, Glowacki R, Pereira G, Workman CT, Arntzen MØ, Pope PB, Martens EC, Hachem MA, Westereng B (2019) The human gut Firmicute Roseburia intestinalis is a primary degrader of dietary β -mannans. Nat Commun 10:1–14. https://doi.org/10.1038/s41467-019-08812-y
Laikova ON, Mironov AA, Gelfand MS (2001) Computational analysis of the transcriptional regulation of pentose utilization systems in the gamma subdivision of Proteobacteria. FEMS Microbiol Lett 205:315–322
Lansky S, Salama R, Shulami S, Lavid N, Sen S, Schapiro I, Shoham Y, Shoham G (2020) Carbohydrate-binding capability and functional conformational changes of AbnE, an Arabino-oligosaccharide binding protein. J Mol Biol 432:2099–2120. https://doi.org/10.1016/j.jmb.2020.01.041
Larsbrink J, Rogers TE, Hemsworth GR, Mckee LS, Tauzin AS, Spadiut O, Klinter S, Pudlo NA, Urs K, Koropatkin NM, Creagh AL, Haynes CA, Kelly AG, Cederholm SN, Davies GJ, Martens EC, Brumer H (2014) A discrete genetic locus confers xyloglucan metabolism in select human gut Bacteroidetes. Nature 506:498–502. https://doi.org/10.1038/nature12907
Lee LL, Crosby JR, Rubinstein GM, Laemthong T, Bing RG, Straub CT, Adams MWW, Kelly RM (2020) The biology and biotechnology of the genus Caldicellulosiruptor: recent developments in ‘Caldi World.’ Extremophiles 24:1–15. https://doi.org/10.1007/s00792-019-01116-5
Leth ML, Ejby M, Workman C, Ewald DA, Pedersen SS, Sternberg C, Bahl MI, Licht TR, Aachmann FL, Westereng B, Hachem MA (2018) Differential bacterial capture and transport preferences facilitate co-growth on dietary xylan in the human gut. Nat Microbiol 3:570–580. https://doi.org/10.1038/s41564-018-0132-8
Liew KJ, Bruce NC, Sani RK, Chong CS, Yaakop AS, Shamsir MS, Goh KM (2020) Global transcriptomic responses of Roseithermus sacchariphilus strain RA in media supplemented with beechwood xylan. Microorganisms 8:1–22. https://doi.org/10.3390/microorganisms8070976
Lillington SP, Leggieri PA, Heom KA, Malley MAO (2020) ScienceDirect Nature’s recyclers : anaerobic microbial communities drive crude biomass deconstruction. Curr Opin Biotechnol 62:38–47. https://doi.org/10.1016/j.copbio.2019.08.015
Lim HG, Rychel K, Sastry AV, Bentley GJ, Mueller J, Schindel HS, Larsen PE, Laible PD, Guss AM, Niu W, Johnson CW, Beckham GT, Feist AM, Palsson BO (2022) Machine-learning from Pseudomonas putida KT2440 transcriptomes reveals its transcriptional regulatory network. Metab Eng 72:297–310. https://doi.org/10.1016/j.ymben.2022.04.004
Ling C, Peabody GL, Salvachúa D, Kim Y, Kneucker CM, Calvey CH, Monninger MA, Munoz NM, Poirier BC, Ramirez KJ, John PCS, Woodworth SP, Magnuson JK, Burnum-johnson KE, Guss AM, Johnson CW, Beckham GT (2022) Muconic acid production from glucose and xylose in Pseudomonas putida via evolution and metabolic engineering. Nat Commun 13:1–14. https://doi.org/10.1038/s41467-022-32296-y
Liu P (1952) Utilization of carbohydrates by Pseudomonas aeruginosa. J Bacteriol 64:773–781
Liu Y, Rainey PB, Zhang X-X (2015) Molecular mechanisms of xylose utilization by Pseudomonas fluorescens: overlapping genetic responses to xylose, xylulose, ribose and mannitol. Mol Microbiol 98:553–570. https://doi.org/10.1111/mmi.13142
Liu L, Huang W-C, Liu Y, Li M (2021) Diversity of cellulolytic microorganisms and microbial cellulases. Int Biodeterior Biodegradation 163:105277. https://doi.org/10.1016/j.ibiod.2021.105277
Liu C, Sun D, Zhu J, Liu W (2019) Two-component signal transduction systems : a major strategy for connecting input stimuli to biofilm formation. Front Microbiol 9. https://doi.org/10.3389/fmicb.2018.03279
Lombard V, Ramulu HG, Drula E, Coutinho PM, Henrissat B (2014) The carbohydrate-active enzymes database ( CAZy ) in 2013. Nucleic Acids Res 42:490–495. https://doi.org/10.1093/nar/gkt1178
Lopez-Mondejar R, Tlaskal V, Vetrovsy T, Stursova M, Toscan R, Nunes da Rocha U, Baldrian P (2020) Metagenomics and stable isotope probing reveal the complementary contribution of fungal and bacterial communities in the recycling of dead biomass in forest soil. Soil Biol Biochem 148:1–11. https://doi.org/10.1016/j.soilbio.2020.107875
Luis AS, Briggs J, Zhang X, Farnell B, Ndeh D, Labourel A, Baslé A, Cartmell A, Terrapon N, Stott K, Lowe EC, Mclean R, Shearer K, Schückel J, Venditto I, Ralet M-C, Henrissat B, Martens EC, Mosimann SC, Abbott DW, Gilbert HJ (2018) Dietary pectic glycans are degraded by coordinated enzyme pathways in human colonic Bacteroides. Nat Microbiol 3:210–219. https://doi.org/10.1038/s41564-017-0079-1
Mackie RI, Cann I (2018) Let Them Eat Fruit Nat Microbiol 3:127–129. https://doi.org/10.1038/s41564-018-0108-8
Magasanik B (1961) Catabolite repression. Cold Spring Harb Symp Quant Biol 26:249–256. https://doi.org/10.1101/SQB.1961.026.01.031
Marcano-Velazquez JG, Lo J, Nag A, Maness PC, Chou KJ (2019) Developing riboswitch-mediated gene regulatory controls in thermophilic bacteria. ACS Synth Biol 8:633–640. https://doi.org/10.1021/acssynbio.8b00487
Marcos-torres FJ, Perez J, Gomez-Santos N, Moraleda-Munoz A, Munoz-Dorado J (2016) In depth analysis of the mechanism of action of metal-dependent sigma factors: characterization of CorE2 from Myxococcus xanthus. Nucleic Acids Res 44:5571–5584. https://doi.org/10.1093/nar/gkw150
Marcos-Torres FJ, Moraleda-Muñoz A, Contreras-Moreno FJ, Muñoz-Dorado J, Pérez J (2022) Mechanisms of action of non-canonical ECF sigma factors. Int J Mol Sci 23:3601. https://doi.org/10.3390/ijms23073601
Martens EC, Chiang HC, Gordon JI (2008) Mucosal glycan foraging enhances fitness and transmission of a saccharolytic human gut bacterial symbiont. Cell Host Microbe 4:447–457. https://doi.org/10.1016/j.chom.2008.09.007
Martens EC, Roth R, Heuser JE, Gordon JI (2009) Coordinate regulation of glycan degradation and polysaccharide capsule biosynthesis by a prominent human gut symbiont. J Biol Chem 284:18445–18457. https://doi.org/10.1074/jbc.M109.008094
Martens EC, Lowe EC, Chiang H, Pudlo NA, Wu M, Nathan P, Abbott DW, Henrissat B, Gilbert HJ, Bolam DN, Gordon JI (2011) Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol 9:e1001221. https://doi.org/10.1371/journal.pbio.1001221
Mearls EB, Olson DG, Herring CD, Lynd LR (2015) Development of a regulatable plasmid-based gene expression system for Clostridium thermocellum. Appl Microbiol Biotechnol 99:7589–7599. https://doi.org/10.1007/s00253-015-6610-5
Mhatre A, Kalscheur B, Mckeown H, Bhakta K, Sarnaik AP, Flores A, Nielsen DR, Wang X, Soundappan T, Varman AM (2022) Consolidated bioprocessing of hemicellulose to fuels and chemicals through an engineered Bacillus subtilis-Escherichia coli consortium. Renew Energy 193:288–298. https://doi.org/10.1016/j.renene.2022.04.124
Michalak L, La Rosa SL, Leivers S, Lindstad LJ, Røhr ÅK, Aachmann FL, Westereng B (2020) A pair of esterases from a commensal gut bacterium remove acetylations from all positions on complex β-mannans. Proc Natl Acad Sci U S A 117:7122–7130. https://doi.org/10.1073/pnas.1915376117
Munir RI, Spicer V, Krokhin OV, Shamshurin D, Zhang X, Taillefer M, Blunt W, Cicek N, Sparling R, Levin DB (2016) Transcriptomic and proteomic analyses of core metabolism in Clostridium termitidis CT1112 during growth on α-cellulose, xylan, cellobiose and xylose. BMC Microbiol 16:1–21. https://doi.org/10.1186/s12866-016-0711-x
Munoz-Gutierrez I, Ortiz de Ora L, Grinberg IR, Garty Y, Bayer EA, Shoham Y, Lamed R, Borovok I (2016) Decoding biomass-sensing regulons of Clostridium thermocellum alternative sigma-I factors in a heterologous Bacillus subtilis host system. PLoS ONE 11:e0146316. https://doi.org/10.1371/journal.pone.0146316
Nataf Y, Bahari L, Kahel-Raifer H, Borovok I, Lamed R, Bayer EA, Sonenshein AL, Shoham Y (2010) Clostridium thermocellum cellulosomal genes are regulated by extracytoplasmic polysaccharides via alternative sigma factors. Proc Natl Acad Sci U S A 107:18646–18651. https://doi.org/10.1073/pnas.1012175107
Nelson CE, Rogowski A, Morland C, Wilhide JA, Gilbert HJ, Gardner JG (2017) Systems analysis in Cellvibrio japonicus resolves predicted redundancy of b -glucosidases and determines essential physiological functions. Mol Microbiol 104:294–305. https://doi.org/10.1111/mmi.13625
Novak JK, Gardner JG (2023) Galactomannan utilization by Cellvibrio japonicus relies on a single essential α-galactosidase encoded by the aga27A gene. Mol Microbiol 119:312–325. https://doi.org/10.1111/mmi.15024
Ohashi K, Hataya S, Nakata A, Matsumoto K, Kato N, Sato W, Carlos-Shanley C, Beebe ET, Currie CR, Fox BG, Takasuka TE (2021) Mannose- and mannobiose-specific responses of the insect-associated cellulolytic bacterium Streptomyces sp. Strain SirexAA-e Appl Environ Microbiol 87:e02719-e2720
Ortiz de Ora L, Lamed R, Liu YJ, Xu J, Cui Q, Feng Y, Shoham Y, Bayer EA, Muñoz-Gutiérrez I (2018) Regulation of biomass degradation by alternative σ factors in cellulolytic clostridia. Sci Rep 8:1–11. https://doi.org/10.1038/s41598-018-29245-5
Palevich N, Kelly WJ, Ganesh S, Rakonjac J, Attwood GT (2019) Butyrivibrio hungatei MB2003 competes effectively for soluble sugars released by Butyrivibrio proteoclasticus B316 during growth on xylan or pectin. Appl Environ Microbiol 85:e02056-e2118
Pascoal C, Fernandes I, Seena S, Danger M, Ferreira V, Cássio F (2021) Linking microbial decomposer diversity to plant litter decomposition and associated processes in streams. In: Swan CM, Boyero L, Canhoto C (eds) The ecology of plant litter decomposition in stream ecosystems. Springer International Publishing, Cham, pp 163–192
Peabody VGL, Elmore JR, Martinez-Baird J, Guss AM (2019) Engineered Pseudomonas putida KT2440 co-utilizes galactose and glucose. Biotechnol Biofuels 12:1–7. https://doi.org/10.1186/s13068-019-1627-0
Pereira GV, Abdel-Hamid AM, Dutta S, D’Alessandro-Gabazza CN, Wefers D, Farris JA, Bajaj S, Wawrzak Z, Atomi H, Mackie RI, Gabazza EC, Shukla D, Koropatkin NM, Cann I (2021) Degradation of complex arabinoxylans by human colonic Bacteroidetes. Nat Commun 12:1–21. https://doi.org/10.1038/s41467-020-20737-5
Petit E, Coppi MV, Hayes JC, Tolonen AC, Warnick T, Latouf WG, Amisano D, Biddle A, Mukherjee S, Ivanova N, Lykidis A, Land M, Hauser L, Kyrpides N, Henrissat B, Lau J, Schnell DJ, Church GM, Leschine SB, Blanchard JL (2015) Genome and transcriptome of Clostridium phytofermentans, catalyst for the direct conversion of plant feedstocks to fuels. PLoS ONE 10:1–18. https://doi.org/10.1371/journal.pone.0118285
Pinto D, Liu Q, Mascher T (2019) ECF σ factors with regulatory extensions: the one-component systems of the σ universe. Mol Microbiol 112:399–409. https://doi.org/10.1111/mmi.14323
Pollet RM, Martin LM, Koropatkin NM (2021) TonB-dependent transporters in the Bacteroidetes: unique domain structures and potential functions. Mol Microbiol 115:490–501. https://doi.org/10.1111/mmi.14683
Poudel S, Tsunemoto H, Seif Y, Sastry AV, Szubin R, Xu S, Machado H, Olson CA, Anand A, Pogliano J, Nizet V, Palsson BO (2020) Revealing 29 sets of independently modulated genes in Staphylococcus aureus, their regulators, and role in key physiological response. Proc Natl Acad Sci U S A 117:17228–17239. https://doi.org/10.1073/pnas.2008413117
Pudio NA, Urs K, Kumar SS, German JB, Mills DA, Martens EC (2015) Symbiotic human gut bacteria with variable metabolic priorities for host mucosal glycans. Mbio 6:e01282-e1315. https://doi.org/10.1128/mBio.01282-15.Editor
Riederer A, Takasuka TE, Makino S, Stevenson DM, Bukhman YV, Elsen NL, Fox BG (2011) Global gene expression patterns in Clostridium thermocellum as determined by microarray analysis of chemostat cultures on cellulose or cellobiose. Appl Environ Microbiol 77:1243–1253. https://doi.org/10.1128/AEM.02008-10
Rodionov DA, Mironov AA, Gelfand MS (2001) Transcriptional regulation of pentose utilisation systems in the Bacillus / Clostridium group of bacteria. FEMS Microbiol Lett 205:305–314
Rodionov DA, Rodionova IA, Rodionov VA, Arzamasov AA, Zhang K, Rubinstein GM, Tanwee TNN, Bing RG, Crosby JR, Nookaew I, Basen M, Brown SD, Wilson CM, Klingeman DM, Poole II FL, Zhang Y, Kelly RM, Adams MWW (2021) Transcriptional regulation of plant biomass degradation and carbohydrate utilization genes in the extreme thermophile Caldicellulosiruptor bescii. mSystems 6 https://doi.org/10.1128/mSystems.01345-20
Russell JB (1987) Effect of extracellular ph on growth and proton motive force of Bacteroides succinogenes, a cellulolytic ruminal bacterium. Appl Environ Microbiol 53:2379–2383
Rychel K, Sastry AV, Palsson BO (2020) Machine learning uncovers independently regulated modules in the Bacillus subtilis transcriptome. Nat Commun 11:1–10. https://doi.org/10.1038/s41467-020-20153-9
Rydzak T, McQueen PD, Krokhin OV, Spicer V, Ezzati P, Dwivedi RC, Shamshurin D, Levin DB, Wilkins JA, Sparling R (2012) Proteomic analysis of Clostridium thermocellum core metabolism: relative protein expression profiles and growth phase-dependent changes in protein expression. BMC Microbiol 12:1–18. https://doi.org/10.1186/1471-2180-12-214
Sadaie Y, Nakadate H, Fukui R, Yee LM, Asai K (2008) Glucomannan utilization operon of Bacillus subtilis. FEMS Microbiol Lett 279:103–109. https://doi.org/10.1111/j.1574-6968.2007.01018.x
Sand A, Holwerda EK, Ruppertsberger NM, Maloney M, Olson DG, Nataf Y, Borovok I, Sonenshein AL, Bayer EA, Lamed R, Lynd LR, Shoham Y (2015) Three cellulosomal xylanase genes in Clostridium thermocellum are regulated by both vegetative SigA and alternative SigI6 factors. FEBS Lett 589:3133–3140. https://doi.org/10.1016/j.febslet.2015.08.026
Sastry AV, Gao Y, Szubin R, Hefner Y, Xu S, Kim D, Choudhary KS, Yang L, King ZA, Palsson BO (2019) The Escherichia coli transcriptome mostly consists of independently regulated modules. Nat Commun 10:1–14. https://doi.org/10.1038/s41467-019-13483-w
Sawant SS, Salunke BK, Taylor LE, Kim BS (2017) Enhanced agarose and xylan degradation for production of polyhydroxyalkanoates by co-culture of marine bacterium, Saccharophagus degradans and its contaminant. Bacillus Cereus MDPI Appl Sci 7:225. https://doi.org/10.3390/app7030225
Schwalm ND III, Townsed GE II, Groisman EA (2017) Prioritization of polysaccharide utilization and control of regulator activation in Bacteroides thetaiotaomicron. Mol Microbiol 104:32–45. https://doi.org/10.1111/mmi.13609
Shulami S, Shenker O, Langut Y, Lavid N, Gat O, Zaide G, Zehavi A, Sonenshein AL, Shoham Y (2014) Multiple regulatory mechanisms control the expression of the Geobacillus stearothermophilus gene for extracellular xylanase. J Biol Chem 289:25957–25975. https://doi.org/10.1074/jbc.M114.592873
Singhania RR, Patel AK, Singh A, Haldar D, Soam S, Chen C-W, Tsai M-L, Dong C-D (2022) Consolidated bioprocessing of lignocellulosic biomass: technological advances and challenges. Bioresour Technol 354:1–9. https://doi.org/10.1016/j.biortech.2022.127153
Song Y, Xue Y, Ma Y (2013) Global microarray analysis of carbohydrate use in alkaliphilic hemicellulolytic bacterium Bacillus sp. N16–5. PLoS One 8:e54090. https://doi.org/10.1371/journal.pone.0054090
Sonnenburg ED, Sonnenburg JL, Manchester JK, Hansen EE, Chiang HC, Gordon JI (2006) A hybrid two-component system protein of a prominent human gut symbiont couples glycan sensing in vivo to carbohydrate metabolism. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.0603249103
Stülke J, Hillen W (1999) Carbon catabolite repression in bacteria. Curr Opin Microbiol 2:195–201. https://doi.org/10.1016/S1369-5274(99)80034-4
Sun R, Converse PJ, Ko C, Tyagi S, Morrison NE, Bishai WR (2004) Mycobacterium tuberculosis ECF sigma factor sigC is required for lethality in mice and for the conditional expression of a defined gene set. Mol Microbiol 52:25–38. https://doi.org/10.1111/j.1365-2958.2003.03958.x
Takagi T, Morisaka H, Aburaya S, Tatsukami Y, Kuroda K, Ueda M (2016) Putative alginate assimilation process of the marine bacterium Saccharophagus degradans 2–40 based on quantitative proteomic analysis. Mar Biotechnol 18:15–23. https://doi.org/10.1007/s10126-015-9667-3
Taylor LE II, Knott BC, Baker JO, Alahuhta PM, Hobdey SE, Linger JG, Lunin VV, Amore A, Subramanian V, Podkaminer K, Xu Q, VanderWall TA, Schuster LA, Chaudhari YB, Adney WS, Crowley MF, Himmel ME, Decker SR, Beckham GT (2018) Engineering Enhanced Cellobiohydrolase Activity Nat Commun 9:1–10. https://doi.org/10.1038/s41467-018-03501-8
Thapa SP, Ii EWD, Lyu Q, Weisberg AJ, Stevens DM, Clarke CR, Coaker G, Chang JH (2019) The evolution, ecology, and mechanisms of infection by Gram-positive, plant-associated bacteria. Annu Rev Phytopathol 57:341–365
Vanfossen AL, Verhaart MRA, Kengen SMW, Kelly RM (2009) Carbohydrate utilization patterns for the extremely thermophilic bacterium Caldicellulosiruptor saccharolyticus reveal broad growth substrate preferences. Appl Environ Microbiol 75:7718–7724. https://doi.org/10.1128/AEM.01959-09
Von Freiesleben P, Spodsberg N, Stenbæk A, Stålbrand H, Krogh KBRM, Meyer AS (2018) Boosting of enzymatic softwood saccharification by fungal GH5 and GH26 endomannanases. Biotechnol Biofuels 11:1–15. https://doi.org/10.1186/s13068-018-1184-y
Walker JE, Lanahan AA, Zheng T, Toruno C, Lynd LR, Cameron JC, Olson DG, Eckert CA (2020) Development of both type I-B and type II CRISPR/Cas genome editing systems in the cellulolytic bacterium Clostridium thermocellum. Metab Eng Commun 10:e00116. https://doi.org/10.1016/j.mec.2019.e00116
Wang X, Zhang W, Zhou H, Chen G, Liu W (2019a) An extracytoplasmic function sigma factor controls cellulose membrane protein in Cytophaga hutchinsonii. Appl Environ Microbiol 85:e02606-e2618
Wang Y, Horlamus F, Henkel M, Kovacic F, Schläfle S, Hausmann R, Wittgens A, Rosenau F (2019b) Growth of engineered Pseudomonas putida KT2440 on glucose, xylose, and arabinose: hemicellulose hydrolysates and their major sugars as sustainable carbon sources. Glob Chang Biol Bioenergy 11:249–259. https://doi.org/10.1111/gcbb.12590
Williams-Rhaesa AM, Awuku NK, Lipscomb GL, Poole FL, Rubinstein GM, Conway JM, Kelly RM, Adams MW (2018) Native xylose-inducible promoter expands the genetic tools for the biomass-degrading, extremely thermophilic bacterium Caldicellulosiruptor bescii. Extremophiles 22:629–638. https://doi.org/10.1007/s00792-018-1023-x
Wilson CM, Klingeman DM, Schlachter C, Syed MH, Wu C, Guss AM, Brown SD (2017) LacI transcriptional regulatory networks in Clostridium thermocellum DSM1313. Appl Environ Microbiol 83:e02751-e2816
Wu X, Bei S, Zhou X, Luo Y, He Z, Song C, Yuan H, Pivato B (2023) Metagenomic insights into genetic factors driving bacterial niche differentiation between bulk and rhizosphere soils. Sci Total Environ 891:164221
Xiong W, Reyes LH, Michener WE, Maness P-C, Chou KJ (2018) Engineering cellulolytic bacterium Clostridium thermocellum to co-ferment cellulose- and hemicellulose-derived sugars simultaneously. Biotechnol Bioeng 115:1755–1763. https://doi.org/10.1002/bit.26590
Zhang B, Gao Y, Zhang L, Zhou Y (2020) The plant cell wall: biosynthesis, construction, and functions. J Integr Plant Biol 63:251–272. https://doi.org/10.1111/jipb.13055
Ziles Domingues S, Timmers LFSM, Eichelberger Granada C (2022) Cellulase production by bacteria is a strain-specific characteristic with a high biotechnological potential. A review of cellulosome of highly studied strains. Cellulose 29:8065–8083. https://doi.org/10.1007/s10570-022-04790-5
Acknowledgements
The authors wish to thank Gardner laboratory members for helpful discussions during the writing of this review.
Funding
This work was supported by the National Science Foundation, Division of Environmental Biology under Award Number 2038304.
Author information
Authors and Affiliations
Contributions
J. K. N. led the manuscript writing.
J. G. G. contributed to writing the manuscript. All authors read and approved the final submitted version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
N/A
Consent for publication
N/A.
Competing interests
This report was prepared as an account of work sponsored by an agency of the US Government. Neither the US Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the US Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the US Government or any agency thereof. The authors also declare that they have no conflict of interest for this publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Novak, J.K., Gardner, J.G. Current models in bacterial hemicellulase-encoding gene regulation. Appl Microbiol Biotechnol 108, 39 (2024). https://doi.org/10.1007/s00253-023-12977-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00253-023-12977-4