Abstract
Background
Preterm children may have cognitive deficits and behavioural disorders suggestive of grey matter (GM) injury. The prevalence is higher in preterm children with diffuse periventricular leukomalacia (dPVL).
Objective
Evaluate changes in the volume of 116 GM areas in preterm children with dPVL.
Methods and materials
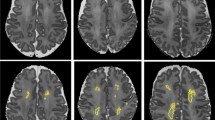
Eleven preterm children with dPVL, gestational age 32.8 ± 2.6 weeks, examined at corrected age 22.0 ± 18.2 months and 33 matched preterm controls with normal brain MRI were studied. Volumes of 116 individual GM areas, and white matter/cerebrospinal fluid (WM/CSF) ratio were calculated on T1-weighted high-resolution images after segmentation.
Results
Relative to controls, children with dPVL had decreased GM volume of the hippocampus, amygdala, and frontal lobes and temporal middle gyrus (P < 0.05); increased GM volume of the putamen, thalamus, globus pallidum, superior temporal gyrus and of the parietal and occipital lobes (P < 0.05) and lower WM volume/higher CSF volume (P < 0.05). WM/CSF ratios also differed (P < 0.05).
Conclusions
Preterm children with dPVL have increased regional GM volume in some areas probably related with a process of brain plasticity-regeneration and reduced GM volume in areas associated with cognition and memory.



Similar content being viewed by others
References
Volpe JJ (2009) Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol 8:110–124
Blumenthal I (2004) Periventricular leucomalacia: a review. Eur J Pediatr 163:435–442
Folkerth RD (2006) Periventricular leukomalacia: overview and recent findings. Pediatr Dev Pathol 9:3–13
Volpe JJ (2003) Cerebral white matter injury of the premature infant-more common than you think. Pediatrics 112:176–180
Anderson P, Doyle LW (2003) Neurobehavioral outcomes of school-age children born extremely low birth weight or very preterm in the 1990s. JAMA 289:3264–3272
Anderson PJ, Doyle LW (2008) Cognitive and educational deficits in children born extremely preterm. Semin Perinatol 32:51–58
Hack M, Fanaroff AA (1999) Outcomes of children of extremely low birthweight and gestational age in the 1990’s. Early Hum Dev 53:193–218
Briscoe J, Gathercole SE, Marlow N (1998) Short-term memory and language outcomes after extreme prematurity at birth. J Speech Lang Hear Res 41:654–666
Nosarti C, Giouroukou E, Healy E et al (2008) Grey and white matter distribution in very preterm adolescents mediates neurodevelopmental outcome. Brain 131:205–217
Nosarti C, Al-Asady MH, Frangou S et al (2002) Adolescents who were born very preterm have decreased brain volumes. Brain 125:1616–1623
Peterson BS, Anderson AW, Ehrenkranz R et al (2003) Regional brain volumes and their later neurodevelopmental correlates in term and preterm infants. Pediatrics 111:939–948
Peterson BS, Vohr B, Staib LH et al (2000) Regional brain volume abnormalities and long-term cognitive outcome in preterm infants. JAMA 284:1939–1947
Inder TE, Warfield SK, Wang H et al (2005) Abnormal cerebral structure is present at term in premature infants. Pediatrics 115:286–294
Lin Y, Okumura A, Hayakawa F et al (2001) Quantitative evaluation of thalami and basal ganglia in infants with periventricular leukomalacia. Dev Med Child Neurol 43:481–485
Tzarouchi LC, Astrakas LG, Zikou A et al (2009) Periventricular leukomalacia in preterm children: assessment of grey and white matter and cerebrospinal fluid changes by MRI. Pediatr Radiol 39:1327–1332
Alemán-Gómez YM-GL, Valdés-Hernandez P (2006) IBASPM: toolbox for automatic parcellation of brain structures. Twelfth Annual Meeting of the Organization for Human Brain Mapping, Florence, Italy
Inder TE, Anderson NJ, Spencer C et al (2003) White matter injury in the premature infant: a comparison between serial cranial sonographic and MR findings at term. AJNR 24:805–809
Falip C, Blanc N, Maes E et al (2007) Postnatal clinical and imaging follow-up of infants with prenatal isolated mild ventriculomegaly: a series of 101 cases. Pediatr Radiol 37:981–989
Altaye M, Holland SK, Wilke M et al (2008) Infant brain probability templates for MRI segmentation and normalization. NeuroImage 43:721–730
Abernethy LJ, Palaniappan M, Cooke RW (2002) Quantitative magnetic resonance imaging of the brain in survivors of very low birth weight. Arch Dis Child 87:279–283
Schmidt-Kastner R, Freund TF (1991) Selective vulnerability of the hippocampus in brain ischemia. Neuroscience 40:599–636
Kuchna I (1994) Quantitative studies of human newborns’ hippocampal pyramidal cells after perinatal hypoxia. Folia Neuropathol 32:9–16
Thompson DK, Wood SJ, Doyle LW et al (2008) Neonate hippocampal volumes: prematurity, perinatal predictors, and 2-year outcome. Ann Neurol 63:642–651
Abraham H, Vincze A, Jewgenow I, et al. Myelination in the human hippocampal formation from midgestation to adulthood. Int J Dev Neurosci 28:401–410
Sim FJ, Windrem MS, Goldman SA (2009) Fate determination of adult human glial progenitor cells. Neuron Glia Biol 5:45–55
Roy NS, Wang S, Harrison-Restelli C et al (1999) Identification, isolation, and promoter-defined separation of mitotic oligodendrocyte progenitor cells from the adult human subcortical white matter. J Neurosci 19:9986–9995
Lodygensky GA, Rademaker K, Zimine S et al (2005) Structural and functional brain development after hydrocortisone treatment for neonatal chronic lung disease. Pediatrics 116:1–7
Gimenez M, Junque C, Narberhaus A et al (2004) Hippocampal gray matter reduction associates with memory deficits in adolescents with history of prematurity. NeuroImage 23:869–877
Isaacs EB, Lucas A, Chong WK et al (2000) Hippocampal volume and everyday memory in children of very low birth weight. Pediatr Res 47:713–720
Beauchamp MH, Thompson DK, Howard K et al (2008) Preterm infant hippocampal volumes correlate with later working memory deficits. Brain 131:2986–2994
Tzarouchi LC, Astrakas LG, Xydis V et al (2009) Age-related grey matter changes in preterm infants: an MRI study. NeuroImage 47:1148–1153
Burgel U, Amunts K, Hoemke L et al (2006) White matter fiber tracts of the human brain: three-dimensional mapping at microscopic resolution, topography and intersubject variability. NeuroImage 29:1092–1105
Bracht T, Tuscher O, Schnell S et al (2009) Extraction of prefronto-amygdalar pathways by combining probability maps. Psychiatry Res 174:217–222
Heinz A, Braus DF, Smolka MN et al (2005) Amygdala-prefrontal coupling depends on a genetic variation of the serotonin transporter. Nat Neurosci 8:20–21
Thierry AM, Gioanni Y, Degenetais E et al (2000) Hippocampo-prefrontal cortex pathway: anatomical and electrophysiological characteristics. Hippocampus 10:411–419
Nagae LM, Hoon AH Jr, Stashinko E et al (2007) Diffusion tensor imaging in children with periventricular leukomalacia: variability of injuries to white matter tracts. AJNR 28:1213–1222
Srinivasan L, Dutta R, Counsell SJ et al (2007) Quantification of deep gray matter in preterm infants at term-equivalent age using manual volumetry of 3-tesla magnetic resonance images. Pediatrics 119:759–765
Ricci D, Anker S, Cowan F et al (2006) Thalamic atrophy in infants with PVL and cerebral visual impairment. Early Hum Dev 82:591–595
Haynes RL, Xu G, Folkerth RD et al (2011) Potential neuronal repair in cerebral white matter injury in the human neonate. Pediatr Res 69:62–67
Okoshi Y, Itoh M, Takashima S (2001) Characteristic neuropathology and plasticity in periventricular leukomalacia. Pediatr Neurol 25:221–226
Okoshi Y, Mizuguchi M, Itoh M et al (2007) Altered nestin expression in the cerebrum with periventricular leukomalacia. Pediatr Neurol 36:170–174
Johnston MV (2003) Brain plasticity in paediatric neurology. Eur J Paediatr Neurol 7:105–113
Staudt M (2007) (Re-)organization of the developing human brain following periventricular white matter lesions. Neurosci Biobehav Rev 31:1150–1156
Taupin P (2006) Adult neurogenesis and neuroplasticity. Restor Neurol Neurosci 24:9–15
Johnston MV (2009) Plasticity in the developing brain: implications for rehabilitation. Dev Disabil Res Rev 15:94–101
Huttenlocher PR, Dabholkar AS (1997) Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol 387:167–178
Yang Z, Levison SW (2006) Hypoxia/ischemia expands the regenerative capacity of progenitors in the perinatal subventricular zone. Neuroscience 139:555–564
Ong J, Plane JM, Parent JM et al (2005) Hypoxic-ischemic injury stimulates subventricular zone proliferation and neurogenesis in the neonatal rat. Pediatr Res 58:600–606
Felling RJ, Snyder MJ, Romanko MJ et al (2006) Neural stem/progenitor cells participate in the regenerative response to perinatal hypoxia/ischemia. J Neurosci 26:4359–4369
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tzarouchi, L.C., Xydis, V., Zikou, A.K. et al. Diffuse periventricular leukomalacia in preterm children: assessment of grey matter changes by MRI. Pediatr Radiol 41, 1545–1551 (2011). https://doi.org/10.1007/s00247-011-2223-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-011-2223-z