Abstract
In birds the major histocompatibility complex (MHC) organization differs both among and within orders; chickens Gallus gallus of the order Galliformes have a simple arrangement, while many songbirds of the order Passeriformes have a more complex arrangement with larger numbers of MHC class I and II genes. Chicken MHC genes are found at two independent loci, classical MHC-B and non-classical MHC-Y, whereas non-classical MHC genes are yet to be verified in passerines. Here we characterize MHC class I transcripts (α1 to α3 domain) and perform amplicon sequencing using a next-generation sequencing technique on exon 3 from house sparrow Passer domesticus (a passerine) families. Then we use phylogenetic, selection, and segregation analyses to gain a better understanding of the MHC class I organization. Trees based on the α1 and α2 domain revealed a distinct cluster with short terminal branches for transcripts with a 6-bp deletion. Interestingly, this cluster was not seen in the tree based on the α3 domain. 21 exon 3 sequences were verified in a single individual and the average numbers within an individual were nine and five for sequences with and without a 6-bp deletion, respectively. All individuals had exon 3 sequences with and without a 6-bp deletion. The sequences with a 6-bp deletion have many characteristics in common with non-classical MHC, e.g., highly conserved amino acid positions were substituted compared with the other alleles, low nucleotide diversity and just a single site was subject to positive selection. However, these alleles also have characteristics that suggest they could be classical, e.g., complete linkage and absence of a distinct cluster in a tree based on the α3 domain. Thus, we cannot determine for certain whether or not the alleles with a 6-bp deletion are non-classical based on our present data. Further analyses on segregation patterns of these alleles in combination with dating the 6-bp deletion through MHC characterization across the genus Passer may solve this matter in the future.




Similar content being viewed by others
References
Alcaide M, Edwards SV, Cadahía L, Negro JJ (2009) MHC class I genes of birds of prey: isolation, polymorphism and diversifying selection. Conserv Genet 10:1349–1355
Babik W, Taberlet P, Ejsmond MJ, Radwan J (2009) New generation sequencers as a tool for genotyping of highly polymorphic multilocus MHC system. Mol Ecol Resour 9:713–719. doi:10.1111/j.1755-0998.2009.02622.x
Balakrishnan CN, Ekblom R, Volker M, Westerdahl H, Godinez R, Kotkiewicz H, Burt DW, Graves T, Griffin DK, Warren WC, Edwards SV (2010) Gene duplication and fragmentation in the zebra finch major histocompatibility complex. BMC Biol 8:29
Bjorkman PJ, Saper MA, Samraoui B, Bennett WS, Strominger JL, Wiley DC (1987) The foreign antigen binding site and T cell recognition regions of class I histocompatibility antigens. Nature 329:512–518
Bollmer JL, Dunn PO, Whittingham LA, Wimpee C (2010) Extensive MHC class II B gene duplication in a Passerine, the Common Yellowthroat (Geothlypis trichas). J Hered 101:448–460
Bonneaud C, Sorci G, Morin V, Westerdahl H, Zoorob R, Wittzell H (2004) Diversity of Mhc class I and IIB genes in house sparrows (Passer domesticus). Immunogenetics 55:855–865
Borg AA, Pedersen SA, Jensen H, Westerdahl H (2011) Variation in MHC genotypes in two populations of house sparrow (Passer domesticus) with different population histories. Ecol Evol 1(2):145–159. doi:10.1002/ece3.13
Borghans JAM, Beltman JB, De Boer RJ (2004) MHC polymorphism under host-pathogen coevolution. Immunogenetics 55:732–739
Bos DH, Waldman B (2006) Evolution by recombination and transspecies polymorphism in the MHC class I gene of Xenopus laevis. Mol Biol Evol 23:137–143
Burri R, Salamin N, Studer RA, Roulin A, Fumagalli L (2010) Adaptive divergence of ancient gene duplicates in the avian MHC class II beta. Mol Biol Evol 27:2360–2374
Cleasby IR, Nakagawa S, Gillespie DOS, Burke T (2010) The influence of sex and body size on nestling survival and recruitment in the house sparrow. Biol J Linn Soc 101:680–688
Delany ME, Robinson CM, Goto RM, Miller MM (2009) Architecture and organization of chicken microchromosome 16: order of the NOR, MHC-Y, and MHC-B Subregions. Heredity 100:507–514
Delport W, Poon AF, Frost SDW, Kosakovsky Pond SL (2010) Datamonkey 2010 a suite of phylogenetic analysis tools for evolutionary biology. Bioinformatics 26(19):2455–2457. doi:10.1093/bioinformatics/btq429
Doherty PC, Zinkernagel RM (1975) Enhanced immunological surveillance in mice heterozygous at the H-2 gene complex. Nature 256:50–52
Ekblom R, French L, Slate J, Burke Terry (2010) Evolutionary analysis and expression profiling of zebra finch immune genes. Genome Biol Evol 2:781–790. doi:10.1093/gbe/evq061
Ekblom R, Stapley J, Ball AD, Birkhead T, Burke T, Slate J (2011) Genetic mapping of the major histocompatibility complex in the zebra finch (Taeniopygia guttata). Immunogenetics 63:523–530
Follin E, Karlsson M, Lundegaard C, Nielsen M, Wallin S, Paulsson K, Westerdahl H (2013) In silico peptide-binding predictions of passerine MHC class I reveal similarities across distantly related species, suggesting convergence on the level of protein function. Immunogenetics 65:299–311. doi:10.1007/s00251-012-0676-3
Freeman-Gallant CR, Johnson EM, Saponara F, Stanger M (2002) Variation at the major histocompatibility complex in Savannah sparrows. Mol Ecol 11:1125–1130
Galan M, Guivier E, Caraux G, Charbonnel N, Cosson JF (2010) A 454 multiplex sequencing method for rapid and reliable genotyping of highly variable genes. BMC Genom 2010(11):296
Glaberman S, Du Pasquier L, Caccone A (2008) Characterization of a nonclassical class I MHC gene in a reptile, the Galapagos Marine Iguana (Amblyrhynchus cristatus). PLoS ONE 3(8):e2859. doi:10.1371/journal.pone.0002859
Goyos A, Sowa J, Ohta Y, Robert J (2011) Remarkable conservation of distinct nonclassical MHC class I lineages in divergent amphibian species. J Immunol 186:372–381. doi:10.4049/jimmunol.1001467
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucl Acid Symp Ser 41:95–98
Hedrick PW (1994) Evolutionary genetics of the major histocompatibility complex. Am Nat 143:945–964
Hedrick PW (2002) Pathogen resistance and genetic variation at MHC loci. Evolution 56:1902–1908
Hess CM, Gasper J, Hoekstra HE, Hill CE, Edwards SV (2000) MHC class II pseudogene and genomic signature of a 32-kb cosmid in the house finch (Carpodacus mexicanus). Genome Res 10:613–623
Hosomichi K, Shiina T, Suzuki S, Tanaka M, Shimizu S, Iwamoto S, Hara H, Yoshida Y, Kulski J, Inoko H, Hanzawa K (2006) The major histocompatibility complex (Mhc) class IIB region has greater genomic structural flexibility and diversity in the quail than the chicken. BMC Genom 7:322. doi:10.1186/1471-2164-7-322
Hosomichi K, Miller MM, Goto RM, Wang Y, Suzuki S, Kulski JK, Nishibori M, Inoko H, Hanzawa K, Shiina T (2008) Contribution of mutation, recombination, and gene conversion to chicken MHC-B haplotype diversity. J Immunol 181:3393–3399
Hughes AL, Nei M (1988) Patterns of nucleotide substitution at major histocompatibility complex class I loci reveals overdominant selection. Nature 335:167–170
Hughes AL, Yeager M (1998) Natural selection at major histocompatibility complex loci of vertebrates. Annu Rev Genet 32:415–435
Hughes CR, Miles S, Walbroehl JM (2008) Support for the minimal essential MHC hypothesis: a parrot with a single, highly polymorphic MHC class II B gene. Immunogenetics 60:219–231
Kaufman J, Salomonsen J, Flajnik M (1994) Evolutionary conservation of MHC class I and class II molecules—different yet the same. Semin Immunol 6:411–424
Kaufman J, Volk H, Wallny HJ (1995) A “minimal essential Mhc” and an “unrecognized Mhc”: two extremes in selection for polymorphism. Immunol Rev 143:63–88
Kaufman J, Milne S, Gobel TW, Walker BA, Jacob JP, Auffray C, Zoorob R, Beck S (1999) The chicken B locus is a minimal essential major histocompatibility complex. Nature 401:923–925
Koch M, Camp S, Collen T, Avila D, Salomonsen J, Wallny HJ, van Hateren A, Hunt L, Jacob JP, Johnston F, Marston DA, Shaw I, Dunbar PR, Cerundolo V, Jones EY, Kaufman J (2007) Structures of an MHC class I molecule from B21 chickens illustrate promiscuous peptide binding. Immunity 27:885–899
Koskakovsky Pond SL, Frost SDW (2005) Datamonkey: rapid detection of selective pressure on individual sites of codon alignments. Bioinformatics 21(10):2531–2533
Miller MM, Goto RM, Taylor RL Jr, Zoorob R, Auffray C, Briles RW, Briles WE, Bloom SE (1996) Assignment of Rfp-Y to the chicken major histocompatibility complex/NOR microchromosome and evidence for high-frequency recombination associated with the nucleolar organizer region. Proc Natl Acad Sci USA 93:3958–3962
Murphy K, Travers P, Walport M (2008) Janeway’s immunobiology, 7th edn. Garland Science, New York
Nakagawa S, Burke T (2008) The mask of seniority? A neglected age indicator in house sparrows Passer domesticus. Avian Biol 39:222–225
Nakagawa S, Lee JW, Woodward BK, Hatchwell BJ, Burke T (2008) Differential selection according to the degree of cheating in a status signal. Biol Lett 4:667–669
Nei M, Gojobori T (1986) Simple methods for estimating the numbers of synonymous and nonsynonymous nucleotide substitutions. Mol Biol Evol 3:418–426
Ockendon N, Griffith SC, Burke T (2009) Extrapair paternity in an insular population of house sparrows after the experimental introduction of individuals from the mainland. Behav Ecol 20:305–312
Potts WK, Wakeland EK (1990) Evolution of diversity at the major histocompatibility complex. Trends Ecol Evol 5:181–187
Promerova M, Albrecht T, Bryja J (2009) Extremely high MHC class I variation in a population of a long-distance migrant, the Scarlet Rosefinch (Carpodacus erythrinus). Immunogenetics 61:451–461
Reed KM, Bauer MM, Monson MS, Benoit B, Chaves LD, O’Hare TH, Delany ME (2011) Defining the Turkey MHC: identification of expressed class I- and class IIB-like genes independent of the MHC-B. Immunogenetics 63:753–771
Rodgers JR, Cook RG (2005) MHC class IB molecules bridge innate and acquired immunity. Nat Rev Immunol 5:459–471
Salter RD, Norment AM, Chen BP, Clayberger C, Krensky AM, Littman DR, Parham P (1989) Polymorphism in the α3 domain of HLA-A molecules affects binding to CD8. Nature 338:345–347
Salter RD, Benjamin RJ, Wesley PK, Buxton SE, Garrett TPJ, Clayberger C, Krensky AM, Norment AM, Littman DR, Parham P (1990) A binding-site for the T-cell co-receptor Cd8 on the alpha-3 domain of Hla-A2. Nature 345:41–46
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Habour Laboratory Press, Cold Spring Harbour
Schroeder J, Burke T, Owens I, Nakagawa S (2012) Maternal effects and heritability of annual productivity. J Evol Biol 25:149–156
Schut E, Aguilar JR, Merino S, Magrath MJ, Komdeur J, Westerdahl H (2011) Characterization of MHC-I in the blue tit (Cyanistes caeruleus) reveals low levels of genetic diversity and trans-population evolution across European populations. Immunogenetics 63:531–542
Sepil I, Moghadam HK, Huchard E, Sheldon BC (2012) Characterization and 454 pyrosequencing of Major Histocompatibility Complex class I genes in the great tit reveal complexity in a passerine system. BMC Evol Biol 12:68. doi:10.1186/1471-2148-12-68
Shawar S, Vayas J, Rodgers J, Rich R (1994) Antigen presentation by major histocompatibility class I-B molecules. Annu Rev Immunol 12:839–880
Shiina T, Shimizu S, Hosomichi K, Kohara S, Watanabe S, Hanzawa K, Beck S, Kulski JK, Inoko H (2004) Comparative genomic analysis of two avian (quail and chicken) MHC regions. J Immunol 172:6751–6763
Sibly RM, Witt CC, Wright NA, Venditti C, Jetz W, Brown JH (2012) Energetics, lifestyle, and reproduction in birds. Proc Natl Acad Sci USA 109:10937–10941
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML Web servers. Syst Biol 57:758–771
Strand T, Westerdahl H, Hoeglund J, Alatalo RV, Siitari H (2007) The Mhc class II of the Black grouse (Tetrao tetrix) consists of low numbers of B and Y genes with variable diversity and expression. Immunogenetics 59:725–734
Strandh M, Lannefors M, Bonadonna F, Westerdahl H (2011) Characterization of MHC class I and II genes in a subantarctic seabird, the Blue Petrel, Halobaena caerulea (Procellariiformes). Immunogenetics 63:653–666. doi:10.1007/s00251-011-0534-8
Stuglik MT, Radwan J, Babik W (2011) jMHC: software assistant for multilocus genotyping of gene families using next-generation amplicon sequencing. Mol Ecol Resour 11(4):739–742. doi:10.1111/j.1755-0998.2011.02997.x
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. doi:10.1093/molbev/msr121. http://www.kumarlab.net/publications
Westerdahl H (2007) Passerine MHC: genetic variation and disease resistance in the wild. J Ornithol 148:S469–S477
Westerdahl H, Wittzell H, von Schantz T (1999) Polymorphism and transcription of Mhc class I genes in a passerine bird, the great reed warbler. Immunogenetics 49:158–170
Westerdahl H, Wittzell H, von Schantz T, Bensch S (2004) MHC class I typing in a songbird with numerous loci and high polymorphism using motif-specific PCR and DGGE. Heredity 92:534–542
Wittzell H, Madsen T, Westerdahl H, Shine R, von Schantz T (1998) MHC variation in birds and reptiles. Genetica 104:301–309
Wittzell H, Bernot A, Auffray C, Zoorob R (1999) Concerted evolution of two Mhc class II B loci in pheasants and domestic chickens. Mol Biol Evol 16:479–490
Yeager M, Hughes AL (1996) Trans-species polymorphism vs independent evolution of class II MHC sequence motifs. Hum Immunol 47:O689
Zagalska-Neubauer M, Babik W, Stuglik M, Gustafsson L, Cichon M, Radwan J (2010) 454 sequencing reveals extreme complexity of the class II Major Histocompatibility Complex in the collared flycatcher. BMC Evol Biol 10:395. doi:10.1186/1471-2148-10-395
Acknowledgments
We would like to thank Terry Burke for letting us use DNA from the sparrows on Lundy and the Landmark trust and the Lundy Company for letting us work on Lundy. We are grateful to all the staff on Lundy especially Kevin Welsh, Patricia, and Roger Fursdon. Thanks to Ian Cleasby for assistance in the field. We are grateful to Mimi Lannefors and Anna Drews for help in the laboratory and to Emily O’Connor, Kristin Scherman, and Maria Strandh for valuable comments on earlier versions of the manuscript. The project was funded by the Swedish Research Council, VR (621-2206-2876) to Helena Westerdahl and Stiftelsen Lunds Djurskyddsfond to Maria Karlsson.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
239_2013_9575_MOESM1_ESM.pdf
Supplementary Fig. 1. Alignment of house sparrow MHC-I amino acid sequences (Pado-UA), covering the α2 region, retrieved from 454-amplicon sequencing, species specific nomenclature and GenBank accession numbers are used for all sequences (Pado-UA*200-260 (novel from the present study), Pado-UA*324 JN609623, Pado-UA*326 JN609626, Pado-UA*312 JN609636, Pado-UA*309 JN609635, Pado-UA*322 JN609643, Pado-UA*317 JN609640 (found previously and also in the present study) in comparison with the house sparrow transcripts (Pado-UA*230-233, *240-243, this study), great reed warbler (Acar cN3, AJ005503; cN15, AJ005505) and chicken (MHC-B, Gaga, HQ141386; MHC-Y NM_001030675). Amino acid sequences are numbered according to full-length chicken MHC-I. Pado-UA*240-242 have no deletions, Pado-UA*243 has a 3 bp deletion (site 149) and Pado-UA*230-233 have 6 bp deletions (site 246-247). Identity with sequence Pado-UA*240 is indicated with dots, codons corresponding to the PBR according to human HLA (Björkman et al. 1987) with (P); sites subject to negative selection (N) and to positive selection (+) according to HyPhy. HyPhy was run for 38 454-sequences with a 6 bp deletion (with 6 bp deletion), for 20 454-sequences without a 6 bp deletion (without 6 bp deletion) and for the 58 sequences combined (all 454-sequences) (PDF 21 kb)
239_2013_9575_MOESM2_ESM.pdf
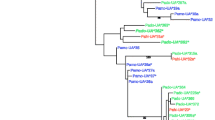
Supplementary Fig. 2. Phylogenetic reconstruction of house sparrow (Pado-UA*230-233 and Pado-UA*240-243) and great reed warbler (Acar cN3, AJ005503; cN15, AJ005505) exon 2, 3 and 4 MHC-I nucleotide sequences, with chicken sequences (MHC-B, Gaga, HQ141386; MHC-Y, NM_001030675) as outgroups, using Neighbor-joining (Jukes-Cantor, bootstrap (bt) values based on 1000 replicates). (a) Phylogenetic reconstruction based on nucleotides in exon 2, (b) phylogenetic reconstruction based on nucleotides in exon 3, (c) phylogenetic reconstruction based on nucleotides in exon 4, (d) phylogenetic reconstruction based on synonymous substitutions in exon 2, (e) phylogenetic reconstruction based on synonymous substitutions in exon 3, (f) phylogenetic reconstruction based on synonymous substitutions in exon 4, (g) phylogenetic reconstruction based on nonsynonymous substitutions in exon 2, (h) phylogenetic reconstruction based on nonsynonymous substitutions in exon 3 and (i) phylogenetic reconstruction based on nonsynonymous substitutions in exon 4. The house sparrow sequences with a 6 bp deletion form a significantly supported monophyletic cluster in all trees based on exons 2 and 3 (a bt = 99,b bt = 100,d bt = 77,e bt = 82,g bt = 99,and h bt = 99), but not in trees based on exon 4 (c, f, i) (PDF 18 kb)
239_2013_9575_MOESM3_ESM.jpg
Supplementary Fig. 3. Inheritance patterns of house sparrow exon 3 sequences (in this paragraph called alleles, with (‘short’) and without (‘long’) the 6bp deletion) in three house sparrow families with 2-4 chicks. Alleles are retrieved from 454-amplicon sequencing, species specific nomenclature and GenBank accession numbers are used for all sequences (Pado-UA*200-260 (novel from the present study), Pado-UA*324 JN609623, Pado-UA*326 JN609626, Pado-UA*312 JN609636, Pado-UA*309 JN609635, Pado-UA*322 JN609643, Pado-UA*317 JN609640 (found previously and also in the present study). Male alleles are indicated with blue, and the two male haplotypes are indicated light and dark blue. Female alleles are indicated with red and the two female haplotypes are pink and red. When alleles are found in both the male and female in-pair haplotypes cannot be inferred and these alleles are non-informative in considering inheritance (indicated green). In clutch 3 the female has three ‘short’ alleles, one allele is red and is inherited in all chicks except in Chick2, Chick2 inherits two pink alleles. This female furthermore has three ‘long’ alleles, one is red and is inherited in all four chicks except in Chick2, Chick2 again inherits two pink alleles. In clutch 3 the male has eight ‘short’ alleles, four are dark blue and they are inherited in all four chicks. This male has six ‘long’ alleles, three are dark blue and are inherited in all four chicks. This male also has four ‘short’ light blue alleles and three ‘long’ light blue alleles, these alleles are not found in any of the four chicks. We find the same pattern of inheritance independent of whether we study inheritance of ‘long’ or ‘short’ alleles, this finding indicates that the ‘long’ and ‘short’ alleles are linked, hence found in the same chromosomal region. Three individuals with 454-read numbers lower than 120 for the ‘short’ alleles were added in this analysis (number of reads from amplification with the ‘short’ primer combination; 64 in Chick2 Clutch3, 65 inChick2 Clutch2 and 92 in the male in Clutch1) (JPG 248 kb)
239_2013_9575_MOESM4_ESM.docx
Supplementary Table 1. Verified MHC-I exon 2-4 cDNA sequences, covering the α1, α2 and α3 regions, in individual 1 and 2 (Ind1 and Ind2), respectively (with a 6 bp deletion, Pado-UA*230-233; without a 6 bp deletion, Pado-UA*240-343) (DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Karlsson, M., Westerdahl, H. Characteristics of MHC Class I Genes in House Sparrows Passer domesticus as Revealed by Long cDNA Transcripts and Amplicon Sequencing. J Mol Evol 77, 8–21 (2013). https://doi.org/10.1007/s00239-013-9575-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-013-9575-y