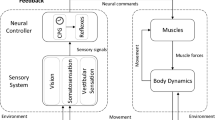
Abstract
The foot is a critical interface between the body and supporting surface during walking, but there is no coherent framework on which to model the dynamics of the stance and swing phases. To establish this framework, we studied the rotational and translational dynamics of foot movement in three dimensions with a motion detection system (OPTOTRAK), while subjects walked on a treadmill. Positions, velocities, and durations were normalized to leg-length and gravity. Foot position and rotation at toe-off were closely related to walking velocity. Foot pitch at toe clearance increased with walking velocity, but the medial–lateral and vertical toe positions were unaltered. Phase–plane trajectories along the fore-aft direction, i.e., plots of toe velocity versus position, were circular during the swing phases, with radii proportional to walking velocity. Peak forward, lateral, and upward velocities were linearly related to corresponding excursions, forming main sequences. A second order model predicted the changes in toe position and velocity, and the approximately hyperbolic decrements in duration as a function of walking velocity. The model indicates that the foot is controlled in an overdamped manner during the stance phase and as a feedback-controlled undamped pendulum during the swing. The data and model suggest that the state of the foot at toe-off, set by walking velocity during the stance phase, determines the dynamics of the swing phase. Thus, in addition to determining locomotion kinematics, walking velocity plays a critical role in determining the phase–plane trajectories and main sequence relationships of foot movements during the swing phases.










Similar content being viewed by others
References
Austin GP, Garrett GE, Bohannon RW (1999) Kinematic analysis of obstacle clearance during locomotion. Gait Posture 10:109–120
Ayyappa E (1997a) Normal human locomotion, Part 1: Basic concepts and terminology. J Prosthet Orthot 9:10–17
Ayyappa E (1997b) Normal human locomotion, Part 2: Motion, ground reaction force and muscle activity. J Prosthet Orthot 9:42–57
Bahill AT, Clark MG, Stark L (1975) The main sequence, a tool for studying human eye movements. Math Biosci 24:191–204
Basmajian JV (1976) The human bicycle. In: Komi PV (eds) Biomechanics, vol 5-A. University Park Press, Baltimore
Bauby CE, Kuo AD (2000) Active control of lateral balance in human walking. J Biomech 33:1433–1440
Berthoz A, Viaud-Delmon I (1999) Multisensory integration in spatial orientation. Curr Opin Neurobiol 9:708–712
Borghese NA, Bianchi L, Lacquaniti F (1996) Kinematic determinants of human locomotion. J Physiol 494(Pt 3):863–879
Boyd IA (1980) The isolated mammalian muscle spindle. Trends Neurosci 3:258–265
Brown TG (1914) On the nature of the fundamental activity of the nervous centers. J Physiol 48:18–46
Cavagna GA, Margaria R (1966) Mechanics of walking. J Appl Physiol 21:271–278
Cavagna GA, Thys H, Zamboni A (1976) The sources of external work in level walking and running. J Physiol 262:639–657
Chandler RF, Caluser CE, McConville JT, Reynolds HM, Young JW (1975) Investigation of inertial properties of the human body. In: AMRL Technical Report, vol 13. Wright-Patterson Air Force Base, pp 74–137
Chevallereau C, Abba G, Aoustin Y, Plestan F, Westervelt ER, Canudas-de-Wit C, Grizzle JW (2003) RABBIT: a testbed for advanced control theory. IEEE Control Syst Mag 23:57–79
Cohen B, Matsuo V, Raphan T (1977) Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic after-nystagmus. J Physiol 270:321–344
Cohen B, Xiang Y, John P, Yakushin SB, Kunin M, Raphan T (2005) Three dimensional kinematics of quadrupedal locomotion in monkeys. Soc. Neurosc. Abstr. 168.11
Courtine G, Roy RR, Raven J, Hodgson J, McKay H, Yang H, Zhong H, Tuszynski MH, Edgerton VR (2005) Performance of locomotion and foot grasping following a unilateral thoracic corticospinal tract lesion in monkeys (Macaca mulatta). Brain 128:2338–2358
Della Croce U, Riley PO, Lelas JL, Kerrigan DC (2001) A refined view of the determinants of gait. Gait Posture 14:79–84
Desoer CA (1970) Notes for a second course on linear systems. Van Nostrand Reinhold, Cincinnati
Doke J, Donelan JM, Kuo AD (2005) Mechanics and energetics of swinging the human leg. J Exp Biol 208:439–445
Donelan JM, Kram R, Kuo AD (2001) Mechanical and metabolic determinants of the preferred step width in human walking. Proc Biol Sci 268:1985–1992
Donelan JM, Shipman DW, Kram R, Kuo AD (2004) Mechanical and metabolic requirements for active lateral stabilization in human walking. J Biomech 37:827–835
Duysens J, Van de Crommert HW (1998) Neural control of locomotion; the central pattern generator from cats to humans. Gait Posture 7:131–141
Eng JJ, Winter DA, Patla AE (1997) Intralimb dynamics simplify reactive control strategies during locomotion. J Biomech 30:581–588
Fick A (1854) Die Bewegungen des menschlichen Augapfels. Z Rationelle Med 4:101–128
Goldstein H (1980) Classical mechanics. Addison-Wesley, Reading
Grasso R, Bianchi L, Lacquaniti F (1998) Motor patterns for human gait: backward versus forward locomotion. J Neurophysiol 80:1868–1885
Grasso R, Ivanenko YP, Zago M, Molinari M, Scivoletto G, Castellano V, Macellari V, Lacquaniti F (2004) Distributed plasticity of locomotor pattern generators in spinal cord injured patients. Brain 127:1019–1034
Grillner S (1975) Locomotion in vertebrates: central mechanisms and reflex interaction. Physiol Rev 55:247–304
Grillner S, Dubuc R (1988) Control of locomotion in vertebrates: spinal and supraspinal mechanisms. Adv Neurol 47:425–453
Grillner S, Halbertsma J, Nilsson J, Thorstensson A (1979) The adaptation to speed in human locomotion. Brain Res 165:177–182
Grillner S, Hellgren J, Menard A, Saitoh K, Wikstrom MA (2005) Mechanisms for selection of basic motor programs—roles for the striatum and pallidum. Trends Neurosci 28:364–370
Guadagnoli MA, Etnyre B, Rodrigue ML (2000) A test of a dual central pattern generator hypothesis for subcortical control of locomotion. J Electromyogr Kinesiol 10:241–247
Hebb O (1949) Organization of behavior. Wiley, New York
Hepp K, Henn V (1983) Spatio-temporal recoding of rapid eye movement signals in the monkey paramedian pontine reticular formation (PPRF). Exp Brain Res 52:105–120
Hirasaki E, Moore ST, Raphan T, Cohen B (1999) Effects of walking velocity on vertical head and body movements during locomotion. Exp Brain Res 127:117–130
Hof AL (1996) Scaling gait data to body size. Gait Posture 4:222–223
Hurmuzlu Y, Basdogan C, Carollo JJ (1994) Presenting joint kinematics of human locomotion using phase plane portraits and Poincare maps. J Biomech 27:1495–1499
Imai T, Moore ST, Raphan T, Cohen B (2001) Interaction of the body, head, and eyes during walking and turning. Exp Brain Res 136:1–18
Inman VT, Ralston HJ, Todd F (1981) Human walking. Williams and Wilkins, Baltimore
Ivanenko YP, Grasso R, Macellari V, Lacquaniti F (2002) Control of foot trajectory in human locomotion: role of ground contact forces in simulated reduced gravity. J Neurophysiol 87:3070–3089
Ivanenko YP, Grasso R, Zago M, Molinari M, Scivoletto G, Castellano V, Macellari V, Lacquaniti F (2003) Temporal components of the motor patterns expressed by the human spinal cord reflect foot kinematics. J Neurophysiol 90:3555–3565
Ivanenko YP, Poppele RE, Lacquaniti F (2004) Five basic muscle activation patterns account for muscle activity during human locomotion. J Physiol 556:267–282
Jahn K, Deutschlander A, Stephan T, Strupp M, Wiesmann M, Brandt T (2004) Brain activation patterns during imagined stance and locomotion in functional magnetic resonance imaging. Neuroimage 22:1722–1731
Keller EL (1974) Participation of medial pontine reticular formation in eye movement generation in monkey. J Neurophysiol 37:316–332
Komatsuzaki A, Harris HE, Alpert J, Cohen B (1969) Horizontal nystagmus of rhesus monkeys. Acta Otolaryngol 67:535–551
Kunin M, Delman B, Cohen B, Raphan T (2004) Head yaw rotation axes and their relationship to the orientation of the dens. In: Society for Neuroscience Abstracts, San Diego, pp 134–135
Kuo AD (1999) Stabilization of lateral motion in passive dynamic walking. Int J Robot Res 18:917–930
Kuo AD (2001) A simple model of bipedal walking predicts the preferred speed-step length relationship. J Biomech Eng 123:264–269
Lacquaniti F (1989) Central representations of human limb movement as revealed by studies of drawing and handwriting. Trends Neurosci 12:287–291
Lashley K (1933) Integrative function of the cerebral cortex. Physiol Rev 13:1–42
Lee CR, Farley CT (1998) Determinants of the center of mass trajectory in human walking and running. J Exp Biol 201:2935–2944
MacKinnon CD, Winter DA (1993) Control of whole body balance in the frontal plane during human walking. J Biomech 26:633–644
Mazzaro N, Grey MJ, do Nascimento OF, Sinkjaer T (2006) Afferent-mediated modulation of the soleus muscle activity during the stance phase of human walking. Exp Brain Res (in press)
Mena D, Mansour JM, Simon SR (1981) Analysis and synthesis of human swing leg motion during gait and its clinical applications. J Biomech 14:823–832
Mergner T, Becker W (2003) A modeling approach to the human spatial orientation system. Ann N Y Acad Sci 1004:303–315
Miller S, Scott PD (1977) The spinal locomotor generator. Exp Brain Res 30:387–403
Minetti AE (2001a) Invariant aspects of human locomotion in different gravitational environments. Acta Astronaut 49:191–198
Minetti AE (2001b) Walking on other planets. Nature 409:467–469
Mochon S, McMahon TA (1980) Ballistic walking. J Biomech 13:49–57
Ogino M, Tsukinoki I, Hosoda K, Asada M (2004) Controlling lateral stepping of a biped robot by swinging torso toward energy efficient walking. In: IEEE/RSJ International Conference on Intelligent Robots and Systems, pp 1359–1364
Patla AE, Calvert TW, Stein RB (1985) Model of a pattern generator for locomotion in mammals. Am J Physiol 248:R484–R494
Patla AE, Davies TC, Niechwiej E (2004) Obstacle avoidance during locomotion using haptic information in normally sighted humans. Exp Brain Res 155:173–185
Patla AE, Prentice SD (1995) The role of active forces and intersegmental dynamics in the control of limb trajectory over obstacles during locomotion in humans. Exp Brain Res 106:499–504
Patla AE, Rietdyk S, Martin C, Prentice S (1996) Locomotor patterns of the leading and the trailing limbs as solid and fragile obstacles are stepped over: some insights into the role of vision during locomotion. J Mot Behav 28:35–47
Perry J (1992) Gait analysis; normal and pathological function. Slack, Thorofare
Pozzo T, Berthoz A, Lefort L (1990) Head stabilization during various locomotor tasks in humans. I. Normal subjects. Exp Brain Res 82:97–106
Raphan T (1976) A parameter adaptive approach to oculomotor system modeling. In: Electical Engineering. City University of New York, New York, p 204
Raphan T, Matsuo V, Cohen B (1979) Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res 35:229–248
Redfern MS, Schumann T (1994) A model of foot placement during gait. J Biomech 27:1339–1346
Robinson DA (1968) The oculomotor control system. Proc IEEE 56:1032–1049
Robinson DA (1973) Models of the saccadic eye movement control system. Kybernetic 14:71–83
Saunders JB, Inman VT, Eberhart HD (1953) The major determinants in normal and pathological gait. J Bone Joint Surg Am 35-A:543–558
Selles RW, Bussmann JB, Wagenaar RC, Stam HJ (2001) Comparing predictive validity of four ballistic swing phase models of human walking. J Biomech 34:1171–1177
Shik ML, Orlovsky GN (1976) Neurophysiology of locomotor automatism. Physiol Rev 56:465–501
Soechting JF, Ross B (1984) Psychophysical determination of coordinate representation of human arm orientation. Neuroscience 13:595–604
Stark L (1971) The control system for versional eye movements. In: Bach-Y-Rita P, Collins CC, Hyde JE (eds) The control of eye movements. Academic, New York, pp 363–428
Stark L, Zangemeister WH, Edwards J, Grinberg J, Jones A, Lehman S, Lubock P, Narayan V, Nystrom M (1980) Head rotation trajectories compared with eye saccades by main sequence relationships. Invest Ophthalmol Vis Sci 19:986–988
Sutherland D (1988) Developement of mature walking. MacKeith press, Philadelphia
Sutherland DH, Kaufman KR, Moitoza JR (1994) Kinematics of normal human walking. Williams & Wilkins, Baltimore
Van de Crommert HW, Mulder T, Duysens J (1998) Neural control of locomotion: sensory control of the central pattern generator and its relation to treadmill training. Gait Posture 7:251–263
Vieilledent S, Kerlirzin Y, Dalbera S, Berthoz A (2001) Relationship between velocity and curvature of a human locomotor trajectory. Neurosci Lett 305:65–69
Whittle MW (1996) Gait analysis: an introduction, 2nd edn. Reed Educational and Professional Publishing, Ltd., Oxford
Winter DA (1983) Biomechanical motor patterns in normal walking. J Mot Behav 15:302–330
Winter DA, Rogers MW (1992) Foot trajectory in human gait: a precise and multifactorial motor control task. Phys Ther 72:45–53; discussion 54–46
Witte H, Preuschoft H, Recknagel S (1991) Human body proportions explained on the basis of biomechanical principles. Z Morphol Anthropol 78:407–423
Zadeh L, Desoer CA (1963) Linear system theory: the state space approach. McGraw Hill, New York
Zatsiorky VM, Werner SL, Kaimin MA (1994) Basic kinematics of walking. Step length and step frequency. A review. J Sports Med Phys Fitness 34:109–134
Acknowledgments
This work was supported by DC05222 and DC05204.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Measurement coordinate system
The lower left LED was the origin (Fig. 1a, LED 4). The line connecting LEDs 4 and 3 was the Z S-axis (Fig. 1a). It was aligned with the spatial vertical and directed upward. The line connecting LEDs 4 and 1 was the X S direction. It was aligned with the spatial horizontal in the direction of walking and was orthogonal to the Z S-axis. The line normal to the LED plate and positive to the left side of the walking direction was taken as the Y S-axis. These vectors formed a right-handed spatial coordinate frame (Fig. 1a).
Rigid bodies for the foot and shank consisted of four markers embedded 48 mm apart on a plastic frame (Fig. 1b). Five additional markers were placed on the right leg to form shank and foot coordinate frames (Fig. 1b). The foot coordinate markers were placed at the lateral malleolus, the lateral calcaneus (heel) and the fifth metatarsal (toe) (Fig. 1b). The foot rigid body, which was fixed on the shoe, was placed at the proximal part of the foot, approximately on the cuboid bone and lateral cuneiform. The positive Z f-axis of the foot coordinate frame was upward and spatially vertical when standing erect. The X f-axis was a cross product of the Z f-axis and a vector normal to a plane that contained the lateral malleolus, the lateral calcaneus and the fifth metatarsal (positive forward). The Y f-axis was a cross product of the Z f- and X f-axes (positive left).
Shank coordinate markers were placed at the medial and lateral epicondyles of the knee and lateral malleolus of the ankle (Fig. 1b). The shank rigid body was located in the middle of the knee and the ankle on the front-lateral side. The positive Z sh-axis of the shank coordinate frame was upward and spatially vertical when standing erect. The Y sh-axis was a cross product of the Z sh-axis and a vector normal to a plane that contained the medial and lateral epicondyles and the lateral malleolus (positive left). The X sh-axis was a cross product of the Z sh- and Y sh-axes (positive forward).
All rigid bodies were computed dynamically using Toolbench (Northern Digital, Inc) as part of the calibration procedure. Calibration was done by asking the subject to stand erect in a natural hip-wide, fully-extended knee position on a platform (Fig. 1b) and not to move one body part relative to another while holding onto a bar connected to the rotator. The platform was then rotated slowly by hand over a 30 s time period. This calibration procedure allowed the OPTOTRAK sensors to sample all LED coordinates several times so that the coordinate frames and rigid bodies could be computed accurately. After the data were accessed, a program built the coordinate frames and rigid bodies. This procedure took from 20 s to 1 min on a dual processor Opteron (AMD)-based computer.
A yaw angle of 0° of the foot indicated that X f-axis, which is the projection of the line between the lateral calcaneus and the fifth metatarsal (lateral border of the foot) onto the X–Y plane, was in parallel with the spatial X S-axis. During calibration, the averaged yaw angle between the X h- and X f-axes across subjects was −9°. This indicates that the coordinate frame of the foot was rotated laterally relative to the head during calibration, because the X f-axis was directed laterally relative to the line between the calcaneus and big toe. As the subjects were instructed to fixate a visual target placed in the middle of the trajectory, the X-axis of the head (X h) during locomotion was almost equal to the X S-axis, the walking direction. Thus, the yaw angle of the foot relative to space was ≈−9° in the calibrated position, which we denote as primary yaw positions of the foot.
Computation of orientation and translation of body part relative to space
Data were processed by programs written in MATLAB (Mathworks, Inc., Natick, MA, USA). The rotation and translation of the foot and shank were obtained as follows: At each orientation, a plane was fitted to the LED coordinates and a unit normal vector, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{1} , \) was determined (Fig. 1c). A vector was then determined as the average vector connecting the midpoint between LEDs 1 and 2 and the midpoint of LEDs 3 and 4 \( ({\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{{\mathbf{d}}} ) \) (Fig. 1c, dashed vector). The vectors, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{1} \) and \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{{\mathbf{d}}} , \) were then averaged over 11 samples and a normalized cross product between the averaged vector, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{{\mathbf{d}}} , \) and the averaged normal-to-plane vector, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{1} , \) was computed as a unit vector, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{2} . \) The vector \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{2} \) was orthogonal to the averaged vector \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{1} \) and lay in the average plane that was fitted to the LED coordinates. A third vector, \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{3} , \) was determined as the cross product of the averaged \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{1} \) and \( {\mathbf{\ifmmode\expandafter\hat\else\expandafter\^\fi{n}}}_{2} , \) which formed the coordinate triple for the rigid body. A matrix whose columns were the three coordinate vectors was the rotation matrix for the rigid body relative to space over the observation window. The rotation of the rigid body in space was obtained by multiplying the rotation matrix by the inverse of the rotation matrix obtained during calibration. The translation of the rigid body was obtained by subtracting the coordinates of the LEDs after these had been corrected for their rotation and averaging over the four LEDs. Thus, the relationship of the rigid body on the any body part was determined relative to the coordinate frame of that body part, using the markers on the anatomical landmarks. The rotations and translations of the rigid body could then be converted to rotations and translations of the coordinate frame and in turn related to anatomical markers. The toe movement was separately obtained from an LED at the fifth metatarsal.
The rotation and translation of the head were determined from four visible markers on the headband. Since head direction is the most reliable measure of heading during straight walking (Imai et al. 2001), it was used to compute the medial–lateral position of the subject on the treadmill and to obtain the yaw orientation of the foot. Angular rotations of the foot about the ankle were calculated from the rotation matrices of the shank and foot. Although somewhat different from anatomical descriptions of the ankle joint, this relative rotation was defined as the ‘joint angle’ for simplicity.
Definition of the stance and swing phases
Components of the stride cycle (stance and swing phases) were determined from Heel contact (HC) and Toe-Off (TO) which were the points where the velocity of the X component of the heel were equal to zero. Our method for determining HC and TO from the heel movement is approximately equivalent to the determinations of HC and TO in terms of force exerted on a force plate during locomotion (Borghese et al. 1996; Grasso et al. 1998; Ivanenko et al. 2002, 2003). In these studies, a virtual line connecting the greater trochanter (GT) and the lateral malleolus (LM) projected into the sagittal plane is defined as the limb axis. The time of maximum (forward) and minimum (backward) elevation of the limb axis is taken as the heel contact and toe-off respectively. Their estimated times of HC and TO were within 2% of the gait cycle determined by the data from the force plate (2 N/cm2 as threshold) (Borghese et al. 1996; Grasso et al. 1998; Ivanenko et al. 2002, 2003). Since the elevation angle was given by Tan−1[(LMx-GTx)/(LMz-GTz)], maximum and minimum values of this parameter occur when LM reaches its forward and backward limit and when the X velocity of the ankle becomes zero. It is close to the time when heel X velocity becomes 0, which is how HC and TO were determined in the present study. Waveforms representing forward movement obtained during treadmill walking at 1.5 m/s (0.52 Froude) had consistent linear backward stance movements and sigmoidal swing phases (Fig. 1d). Waveforms at other velocities of walking had similar characteristics.
Rights and permissions
About this article
Cite this article
Osaki, Y., Kunin, M., Cohen, B. et al. Three-dimensional kinematics and dynamics of the foot during walking: a model of central control mechanisms. Exp Brain Res 176, 476–496 (2007). https://doi.org/10.1007/s00221-006-0633-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-006-0633-1