Abstract
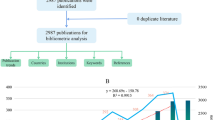
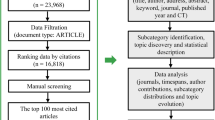
Glucagon-like peptide 1 receptor agonist (GLP-1RA) is a new class of glucose-lowing agents with the kidney benefit effect. This paper aims at finding the current state and hotspots of the research on GLP-1RA in kidney disease by using bibliometric methodologies and visualization maps to analyze publications and provide the direction for future studies on that topic. Literature information was obtained by retrieving the WoSCC database. Then, software like Microsoft Excel, VOSviewer, and CiteSpace was used to analyze and process obtained data. Bibliometric analysis and visualization of nations, authors, organizations, journals, keywords, and references were also done by VOSviewer and CiteSpace. A total of 991 publications written by 4747 authors from 1637 organizations in 75 countries on GLP-1RA in renal disease in Web of Science Core Collection were retrieved. The number of publications and citations kept growing from 2015 to 2022. The USA, Univ Copenhagen, and Rossing Peter are the leading country, organization, and author on this topic, respectively. All literature was published in 346 journals, and DIABETES OBESITY & METABOLISM is the journal with the most contributions. Meanwhile, most references are from DIABETES CARE. “Cardiovascular outcome” is the most frequent keyword in the total publications, and the reference cited most times is “Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes” by Marso SP. The topic of GLP-1RA in renal disease has attracted more and more attention all over the world. Existing studies are mainly about clinical use in patients with diabetes, and studies on the mechanism are lacking.






Similar content being viewed by others
Data availability
The data presented in this article are available on the WoSCC database using the search query listed in the “Materials and methods” section; further inquiries can be directed to the corresponding authors.
References
Andersen A, Lund A, Knop FK, Vilsbøll T (2018) Glucagon-like peptide 1 in health and disease. Nat Rev Endocrinol 14(7):390–403. https://doi.org/10.1038/s41574-018-0016-2
Ashley GW, Henise J, Reid R, Santi DV (2013) Hydrogel drug delivery system with predictable and tunable drug release and degradation rates. Proc Natl Acad Sci USA 110(6):2318–2323. https://doi.org/10.1073/pnas.1215498110
Bakris GL, Fonseca VA, Sharma K, Wright EM (2009) Renal sodium-glucose transport: role in diabetes mellitus and potential clinical implications. Kidney Int 75(12):1272–1277. https://doi.org/10.1038/ki.2009.87
Buchan AM, Polak JM, Capella C, Solcia E, Pearse AG (1978) Electronimmunocytochemical evidence for the K cell localization of gastric inhibitory polypeptide (GIP) in man. Histochemistry 56(1):37–44. https://doi.org/10.1007/BF00492251
Buse JB, Wexler DJ, Tsapas A, Rossing P, Mingrone G, Mathieu C, D’Alessio DA, Davies MJ (2020) 2019 update to: Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 43(2):487–493. https://doi.org/10.2337/dci19-0066
Carraro-Lacroix LR, Malnic G, Girardi ACC (2009) Regulation of Na+/H+ exchanger NHE3 by glucagon-like peptide 1 receptor agonist exendin-4 in renal proximal tubule cells. Am J Physiol Renal Physiol 297(6):F1647-1655. https://doi.org/10.1152/ajprenal.00082.2009
Chen C (2004) Searching for intellectual turning points: progressive knowledge domain visualization. Proc Natl Acad Sci USA 101(Suppl 1):5303–5310. https://doi.org/10.1073/pnas.0307513100
Davies MJ, D’Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, Rossing P, Tsapas A, Wexler DJ, Buse JB (2018a) Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 61(12):2461–2498. https://doi.org/10.1007/s00125-018-4729-5
Davies MJ, D’Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, Rossing P, Tsapas A, Wexler DJ, Buse JB (2018b) Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 41(12):2669–2701. https://doi.org/10.2337/dci18-0033
Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, Maruthur NM, Rosas SE, Del Prato S, Mathieu C, Mingrone G, Rossing P, Tankova T, Tsapas A, Buse JB (2022) Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 65:1925–1966. https://doi.org/10.1007/s00125-022-05787-2
De Nicola L, Zoccali C (2016) Chronic kidney disease prevalence in the general population: heterogeneity and concerns. Nephrol Dial Transplant: Official Publication of the European Dialysis and Transplant Association—European Renal Association 31(3):331–335. https://doi.org/10.1093/ndt/gfv427
Deacon CF, Johnsen AH, Holst JJ (1995) Degradation of glucagon-like peptide-1 by human plasma in vitro yields an N-terminally truncated peptide that is a major endogenous metabolite in vivo. J Clin Endocrinol Metab 80(3):952–957. https://doi.org/10.1210/jcem.80.3.7883856
Drucker DJ (2016) Evolving concepts and translational relevance of enteroendocrine cell biology. J Clin Endocrinol Metab 101(3):778–786. https://doi.org/10.1210/jc.2015-3449
Dupre J, Ross SA, Watson D, Brown JC (1973) Stimulation of insulin secretion by gastric inhibitory polypeptide in man. 1. J Clin Endocrinol Metab 37(5):826–828. https://doi.org/10.1210/jcem-37-5-826
Giugliano D, Longo M, Signoriello S, Maiorino MI, Solerte B, Chiodini P, Esposito K (2022) The effect of DPP-4 inhibitors, GLP-1 receptor agonists and SGLT-2 inhibitors on cardiorenal outcomes: a network meta-analysis of 23 CVOTs. Cardiovasc Diabetol 21(1):42. https://doi.org/10.1186/s12933-022-01474-z
He T, Ao J, Duan C, Yan R, Li X, Liu L, Zhang J, Li X (2022) Bibliometric and visual analysis of nephrotoxicity research worldwide. Front Pharmacol 13:940791. https://doi.org/10.3389/fphar.2022.940791
Hendarto H, Inoguchi T, Maeda Y, Ikeda N, Zheng J, Takei R, Yokomizo H, Hirata E, Sonoda N, Takayanagi R (2012) GLP-1 analog liraglutide protects against oxidative stress and albuminuria in streptozotocin-induced diabetic rats via protein kinase A-mediated inhibition of renal NAD(P)H oxidases. Metab Clin Exp 61(10):1422–1434. https://doi.org/10.1016/j.metabol.2012.03.002
Holst JJ (2007) The physiology of glucagon-like peptide 1. Physiol Rev 87(4):1409–1439. https://doi.org/10.1152/physrev.00034.2006
Hussien NI, Sorour SM, El-Kerdasy HI, Abdelrahman BA (2018) The glucagon-like peptide-1 receptor agonist Exendin-4, ameliorates contrast-induced nephropathy through suppression of oxidative stress, vascular dysfunction and apoptosis independent of glycaemia. Clin Exp Pharmacol Physiol 45(8):808–818. https://doi.org/10.1111/1440-1681.12944
International Diabetes Federation (2023) Kidney and diabetes. https://www.idf.org/our-activities/care-prevention/diabetes-and-the-kidney.html. Accessed 21 February 2023
Katagiri D, Hamasaki Y, Doi K, Okamoto K, Negishi K, Nangaku M, Noiri E (2013) Protection of glucagon-like peptide-1 in cisplatin-induced renal injury elucidates gut-kidney connection. J Am Soc Nephrol 24(12):2034–2043. https://doi.org/10.1681/ASN.2013020134
Kawanami D, Takashi Y (2020) GLP-1 Receptor agonists in diabetic kidney disease: from clinical outcomes to mechanisms. Front Pharmacol 11:967. https://doi.org/10.3389/fphar.2020.00967
Kim M, Platt MJ, Shibasaki T, Quaggin SE, Backx PH, Seino S, Simpson JA, Drucker DJ (2013) GLP-1 receptor activation and Epac2 link atrial natriuretic peptide secretion to control of blood pressure. Nat Med 19(5):567–575. https://doi.org/10.1038/nm.3128
King A, Lock J, Xu G, Bonner-Weir S, Weir GC (2005) Islet transplantation outcomes in mice are better with fresh islets and exendin-4 treatment. Diabetologia 48(10):2074–2079. https://doi.org/10.1007/s00125-005-1922-0
Kjems LL, Holst JJ, Vølund A, Madsbad S (2003) The influence of GLP-1 on glucose-stimulated insulin secretion: effects on beta-cell sensitivity in type 2 and nondiabetic subjects. Diabetes 52(2):380–386. https://doi.org/10.2337/diabetes.52.2.380
Kodera R, Shikata K, Kataoka HU, Takatsuka T, Miyamoto S, Sasaki M, Kajitani N, Nishishita S, Sarai K, Hirota D, Sato C, Ogawa D, Makino H (2011) Glucagon-like peptide-1 receptor agonist ameliorates renal injury through its anti-inflammatory action without lowering blood glucose level in a rat model of type 1 diabetes. Diabetologia 54(4):965–978. https://doi.org/10.1007/s00125-010-2028-x
Li XC, Zhuo JL (2007) Targeting glucagon receptor signalling in treating metabolic syndrome and renal injury in Type 2 diabetes: theory versus promise. Clin Sci (London, England: 1979) 113(4):183–193. https://doi.org/10.1042/CS20070040
Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jodar E, Leiter LA, Lingvay I, Rosenstock J, Seufert J, Warren ML, Woo V, Hansen O, Holst AG, Pettersson J, Vilsboll T (2016a) Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 375(19):1834–1844. https://doi.org/10.1056/NEJMoa1607141
Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JFE, Nauck MA, Nissen SE, Pocock S, Poulter NR, Ravn LS, Steinberg WM, Stockner M, Zinman B, Bergenstal RM, Buse JB, Steering Committee LEADER, Trial Investigators LEADER (2016b) Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 375(4):311–322. https://doi.org/10.1056/NEJMoa1603827
Moellmann J, Klinkhammer BM, Onstein J, Stöhr R, Jankowski V, Jankowski J, Lebherz C, Tacke F, Marx N, Boor P, Lehrke M (2018) Glucagon-like peptide 1 and its cleavage products are renoprotective in murine diabetic nephropathy. Diabetes 67(11):2410–2419. https://doi.org/10.2337/db17-1212
Nauck MA, Bartels E, Orskov C, Ebert R, Creutzfeldt W (1993a) Additive insulinotropic effects of exogenous synthetic human gastric inhibitory polypeptide and glucagon-like peptide-1-(7–36) amide infused at near-physiological insulinotropic hormone and glucose concentrations. J Clin Endocrinol Metab 76(4):912–917. https://doi.org/10.1210/jcem.76.4.8473405
Nauck MA, Heimesaat MM, Orskov C, Holst JJ, Ebert R, Creutzfeldt W (1993b) Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J Clin Investig 91(1):301–307. https://doi.org/10.1172/JCI116186
Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Køber LV, Lawson FC, Ping L, Wei X, Lewis EF, Maggioni AP, McMurray JJV, Probstfield JL, Riddle MC, Solomon SD, Tardif J-C, Investigators ELIXA (2015) Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 373(23):2247–2257. https://doi.org/10.1056/NEJMoa1509225
Rossing P, Caramori ML, Chan JCN, Heerspink HJL, Hurst C, Khunti K, Liew A, Michos ED, Navaneethan SD, Olowu WA, Sadusky T, Tandon N, Tuttle KR, Wanner C, Wilkens KG, Zoungas S, de Boer IH (2022) KDIGO 2022 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int 102(5):S1–S127. https://doi.org/10.1016/j.kint.2022.06.008
Rowlands J, Heng J, Newsholme P, Carlessi R (2018) Pleiotropic effects of GLP-1 and analogs on cell signaling, metabolism, and function. Front Endocrinol 9:672. https://doi.org/10.3389/fendo.2018.00672
Schlatter P, Beglinger C, Drewe J, Gutmann H (2007) Glucagon-like peptide 1 receptor expression in primary porcine proximal tubular cells. Regul Pept 141(1–3):120–128. https://doi.org/10.1016/j.regpep.2006.12.016
Sullivan SD, Alfonso-Cristancho R, Conner C, Hammer M, Blonde L (2009) Long-term outcomes in patients with type 2 diabetes receiving glimepiride combined with liraglutide or rosiglitazone. Cardiovasc Diabetol 8:12. https://doi.org/10.1186/1475-2840-8-12
Sullivan Sean D, Alfonso-Cristancho Rafael, Conner Chris, Hammer Mette, Blonde Lawrence (2009) A simulation of the comparative long-term effectiveness of liraglutide and glimepiride monotherapies in patients with type 2 diabetes mellitus. Pharmacotherapy 29(11):1280–1288. https://doi.org/10.1592/phco.29.11.1280
Tuttle KR, Lakshmanan MC, Rayner B, Busch RS, Zimmermann AG, Woodward DB, Botros FT (2018) Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate-to-severe chronic kidney disease (AWARD-7): a multicentre, open-label, randomised trial. Lancet Diabetes Endocrinol 6(8):605–617. https://doi.org/10.1016/S2213-8587(18)30104-9
Wang XX, Levi J, Luo Y, Myakala K, Herman-Edelstein M, Qiu L, Wang D, Peng Y, Grenz A, Lucia S, Dobrinskikh E, D’Agati VD, Koepsell H, Kopp JB, Rosenberg AZ, Levi M (2017) SGLT2 protein expression is increased in human diabetic nephropathy: SGLT2 protein inhibition decreases renal lipid accumulation, inflammation, and the development of nephropathy in diabetic mice. J Biol Chem 292(13):5335–5348. https://doi.org/10.1074/jbc.M117.779520
Wang J, Li C, Xia C (2018) Improved centrality indicators to characterize the nodal spreading capability in complex networks. Appl Math Comput 334:388–400. https://doi.org/10.1016/j.amc.2018.04.028
Wang X, Li Z, Huang X, Li F, Liu J, Li Z, Bai D (2019) An experimental study of exenatide effects on renal injury in diabetic rats1. Acta Cirurgica Brasileira 34(1):e20190010000001. https://doi.org/10.1590/s0102-865020190010000001
Wei Y, Jiang Z (2021) The evolution and future of diabetic kidney disease research: a bibliometric analysis. BMC Nephrol 22(1):158. https://doi.org/10.1186/s12882-021-02369-z
Wright EM, Turk E (2004) The sodium/glucose cotransport family SLC5. Pflugers Arch 447(5):510–518. https://doi.org/10.1007/s00424-003-1063-6
Xie Y, Bowe B, Mokdad AH, Xian H, Yan Y, Li T, Maddukuri G, Tsai C-Y, Floyd T, Al-Aly Z (2018) Analysis of the Global Burden of Disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int 94(3):567–581. https://doi.org/10.1016/j.kint.2018.04.011
Xie Y, Bowe B, Gibson AK, McGill JB, Maddukuri G, Yan Y, Al-Aly Z (2020) Comparative effectiveness of SGLT2 inhibitors, GLP-1 receptor agonists, DPP-4 inhibitors, and sulfonylureas on risk of kidney outcomes: emulation of a target trial using health care databases. Diabetes Care 43(11):2859–2869. https://doi.org/10.2337/dc20-1890
Xu J, Wang J, Cheng Y, Li X, He M, Zhu J, Han H, Wei G, Kong H, Xie W, Wang H, Zuo X (2018) Glucagon-like peptide-1 mediates the protective effect of the dipeptidyl peptidase IV inhibitor on renal fibrosis via reducing the phenotypic conversion of renal microvascular cells in monocrotaline-treated rats. Biomed Res Int 2018:1864107. https://doi.org/10.1155/2018/1864107
Yang H, Li H, Wang Z, Shi Y, Jiang G, Zeng F (2013) Exendin-4 ameliorates renal ischemia-reperfusion injury in the rat. J Surg Res 185(2):825–832. https://doi.org/10.1016/j.jss.2013.06.042
Zhang J, Yu Q, Zheng F, Long C, Lu Z, Duan Z (2016) Comparing keywords plus of WOS and author keywords: a case study of patient adherence research. J Am Soc Inf Sci 67(4):967–972. https://doi.org/10.1002/asi.23437
Zhang Y, Jin D, Duan Y, Zhang Y, Duan L, Lian F, Tong X (2022) Bibliometric analysis of renal fibrosis in diabetic kidney disease from 1985 to 2020. Front Public Health 10:767591. https://doi.org/10.3389/fpubh.2022.767591
Funding
This study was supported by grants from the Natural Science Foundation of China (No. 81870505, No. 82270778) and the program for Shenyang Bureau of Science and Technology (No. 21–173-9–50).
Author information
Authors and Affiliations
Contributions
LS provided project funding and designed the paper overall. YM and YW searched and filtered publications. WF and YM wrote the manuscript. MZ, JH, and HW plotted figures and constructed tables. YW and LS modified the article. LS reviewed the article. All authors reviewed and approved the manuscript. The authors confirm that no paper mill and artificial intelligence was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, Y., Wang, Y., Fu, W. et al. Global trends and focuses of GLP-1RA in renal disease: a bibliometric analysis and visualization from 2005 to 2022. Naunyn-Schmiedeberg's Arch Pharmacol 396, 3347–3361 (2023). https://doi.org/10.1007/s00210-023-02575-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02575-6