Abstract
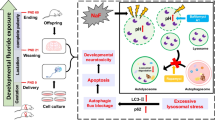
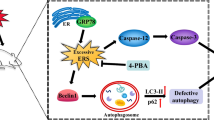
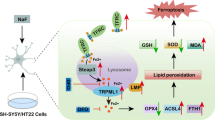
Fluoride neurotoxicity is associated with mitochondrial disruption. Mitochondrial fission/fusion dynamics is crucial to maintain functional mitochondria, yet little is known about how fluoride perturbs this dynamics and whether such perturbation contributes to impaired neurodevelopment. Here in human neuroblastoma SH-SY5Y cells treated with sodium fluoride (NaF, 20, 40 and 60 mg/L), mitochondrial fission suppression exerted a central role in NaF-induced mitochondrial abnormalities and the resulting autophagy deficiency, apoptosis augmentation, and compromised neuronal survival. Mechanically, pharmacological inhibition of mitochondrial fission exacerbated NaF-induced mitochondrial defects and cell death through promoting apoptosis despite partial autophagy restoration. Conversely, genetic enhancement of mitochondrial fission alleviated NaF-produced detrimental mitochondrial and cellular outcomes by elevating autophagy and inhibiting apoptosis. Further suppressing autophagy was harmful, while blocking apoptosis was beneficial for cellular survival in this context. Consistently, using Sprague–Dawley rats developmentally exposed to NaF (10, 50, and 100 mg/L) from pre-pregnancy until 2 months of delivery to mimic human exposure, we showed that perinatal exposure to environmentally relevant levels of fluoride caused learning and memory impairments, accompanied by hippocampal mitochondrial morphological alterations manifested as fission suppression and fusion acceleration, along with defective autophagy, excessive apoptosis and neuronal loss. Intriguingly, the disturbed circulating levels of identified mitochondrial fission/fusion molecules were closely associated with intellectual loss in children under long-term environmental drinking water fluoride exposure. Collectively, our results suggest that mitochondrial fission inhibition induces mitochondrial abnormalities, triggering abnormal autophagy and apoptosis, thus contributing to neuronal death, and that the mitochondrial dynamics molecules may act as promising indicators for developmental fluoride neurotoxicity.








Similar content being viewed by others
References
Angmar-Mansson B, Whitford GM (1984) Enamel fluorosis related to plasma F levels in the rat. Caries Res 18(1):25–32
Barbier O, Arreola-Mendoza L, Del Razo LM (2010) Molecular mechanisms of fluoride toxicity. Chem Biol Interact 188(2):319–333
Bartos M, Gumilar F, Bras C, Gallegos CE, Giannuzzi L, Cancela LM, Minetti A (2015) Neurobehavioural effects of exposure to fluoride in the earliest stages of rat development. Physiol Behav 147:205–212
Bashash M, Thomas D, Hu H, Martinez-Mier EA, Sanchez BN, Basu N, Peterson KE, Ettinger AS, Wright R, Zhang Z, Liu Y, Schnaas L, Mercado-Garcia A, Tellez-Rojo MM, Hernandez-Avila M (2017) Prenatal fluoride exposure and cognitive outcomes in children at 4 and 6–12 years of age in Mexico. Environ Health Perspect 125(9):097017
Choi AL, Sun G, Zhang Y, Grandjean P (2012) Developmental fluoride neurotoxicity: a systematic review and meta-analysis. Environ Health Perspect 120(10):1362–1368
Choi AL, Zhang Y, Sun G, Bellinger DC, Wang K, Yang XJ, Li JS, Zheng Q, Fu Y, Grandjean P (2015) Association of lifetime exposure to fluoride and cognitive functions in Chinese children: a pilot study. Neurotoxicol Teratol 47:96–101
Corrado M, Scorrano L, Campello S (2012) Mitochondrial dynamics in cancer and neurodegenerative and neuroinflammatory diseases. Int J Cell Biol 2012(3):1–13
Dec K, Lukomska A, Maciejewska D, Jakubczyk K, Baranowska-Bosiacka I, Chlubek D, Wasik A, Gutowska I (2017) The Influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol Trace Elem Res 177(2):224–234
Du L, Wan C, Cao X, Liu J (2008) The effect of fluorine on the developing human brain. Fluoride 41:327–330
Encinas M, Iglesias M, Liu Y, Wang H, Muhaisen A, Cena V, Gallego C, Comella JX (2000) Sequential treatment of SH-SY5Y cells with retinoic acid and brain-derived neurotrophic factor gives rise to fully differentiated, neurotrophic factor-dependent, human neuron-like cells. J Neurochem 75(3):991–1003
Friedman JR, Nunnari J (2014) Mitochondrial form and function. Nature 505(7483):335–343
Grandjean P, Landrigan PJ (2007) Developmental neurotoxicity of industrial chemicals. Lancet 369(9564):821–821
Gui CZ, Ran LY, Li JP, Guan ZZ (2010) Changes of learning and memory ability and brain nicotinic receptors of rat offspring with coal burning fluorosis. Neurotoxicol Teratol 32(5):536–541
Guo X, Disatnik MH, Monbureau M, Shamloo M, Mochlyrosen D, Qi X (2013) Inhibition of mitochondrial fragmentation diminishes Huntington’s disease–associated neurodegeneration. J Clin Invest 123(12):5371–5388
Hirzy JW, Connett P, Xiang Q, Spittle BJ, Kennedy DC (2016) Developmental neurotoxicity of fluoride: a quantitative risk analysis towards establishing a safe daily dose of fluoride for children. Fluoride 49(4):379–400
Ikeda Y, Shirakabe A, Maejima Y, Zhai P, Sciarretta S, Toli J, Nomura M, Mihara K, Egashira K, Ohishi M, Abdellatif M, Sadoshima J (2014) Endogenous Drp1 mediates mitochondrial autophagy and protects the heart against energy stress. Circ Res 116(2):264–278
Inoue-Yamauchi A, Oda H (2012) Depletion of mitochondrial fission factor DRP1 causes increased apoptosis in human colon cancer cells. Biochem Biophys Res Commun 421(1):81–85
Izquierdo-Vega JA, Sanchez-Gutierrez M, Del Razo LM (2008) Decreased in vitro fertility in male rats exposed to fluoride-induced oxidative stress damage and mitochondrial transmembrane potential loss. Toxicol Appl Pharmacol 230(3):352–357
Jiang C, Zhang S, Liu H, Guan Z, Zeng Q, Zhang C, Lei R, Xia T, Wang Z, Yang L, Chen Y, Wu X, Zhang X, Cui Y, Yu L, Wang A (2014) Low glucose utilization and neurodegenerative changes caused by sodium fluoride exposure in rat’s developmental brain. Neuromolecular Med 16(1):94–105
Kageyama Y, Zhang Z, Roda R, Fukaya M, Wakabayashi J, Wakabayashi N, Kensler TW, Reddy PH, Iijima M, Sesaki H (2012) Mitochondrial division ensures the survival of postmitotic neurons by suppressing oxidative damage. J Cell Biol 197(4):535–551
Kim J, Park BH, Lee JH, Park SK, Kim JH (2011) Cell type-specific alterations in the nucleus accumbens by repeated exposures to cocaine. Biol Psychiatry 69(11):1026–1034
Kim B, Kim JS, Yoon Y, Santiago MC, Brown MD, Park JY (2013) Inhibition of Drp1-dependent mitochondrial division impairs myogenic differentiation. Am J Physiol Regul Integr Comp Physiol 305(8):R927–R938
Liesa M, Shirihai OS (2013) Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metab 17(4):491–506
Lin JR, Shen WL, Yan C, Gao PJ (2015) Downregulation of dynamin-related protein 1 contributes to impaired autophagic flux and angiogenic function in senescent endothelial cells. Arterioscler Thromb Vasc Biol 35(6):1413–1422
Liu HL, Lam LT, Zeng Q, Han SQ, Fu G, Hou CC (2009) Effects of drinking water with high iodine concentration on the intelligence of children in Tianjin, China. J Public Health (Oxf) 31(1):32–38
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2– ∆∆CT Method. Methods 25(4):402–408
Lou DD, Guan ZZ, Liu YJ, Liu YF, Zhang KL, Pan JG, Pei JJ (2013) The influence of chronic fluorosis on mitochondrial dynamics morphology and distribution in cortical neurons of the rat brain. Arch Toxicol 87(3):449–457
Manczak M, Calkins MJ, Reddy PH (2011) Impaired mitochondrial dynamics and abnormal interaction of amyloid beta with mitochondrial protein Drp1 in neurons from patients with Alzheimer’s disease: implications for neuronal damage. Hum Mol Genet 20(13):2495–2509
Messer JS (2017) The cellular autophagy/apoptosis checkpoint during inflammation. Cell Mol Life Sci 74(7):1281–1296
Mishra P, Chan DC (2014) Mitochondrial dynamics and inheritance during cell division, development and disease. Nat Rev Mol Cell Biol 15(10):634–646
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11(1):47–60
Mumtaz N, Pandey G, Labhasetwar PK (2015) Global fluoride occurrence, available technologies for fluoride removal, and electrolytic defluoridation: a review. Crit Rev Env Sci Tec 45(21):2357–2389
Niu Q, Chen J, Xia T, Li P, Zhou G, Xu C, Zhao Q, Dong L, Zhang S, Wang A (2018) Excessive ER stress and the resulting autophagic flux dysfunction contribute to fluoride-induced neurotoxicity. Environ Pollut 233:889–899
Nunnari J, Suomalainen A (2012) Mitochondria: in sickness and in health. Cell 148(6):1145–1159
Parone PA, James DI, Da Cruz S, Mattenberger Y, Donze O, Barja F, Martinou JC (2006) Inhibiting the mitochondrial fission machinery does not prevent Bax/Bak-dependent apoptosis. Mol Cell Biol 26(20):7397–7408
Peng K, Yang L, Wang J, Ye F, Dan G, Zhao Y, Cai Y, Cui Z, Ao L, Liu J, Zou Z, Sai Y, Cao J (2017) The interaction of mitochondrial biogenesis and fission/fusion mediated by PGC-1alpha regulates rotenone-induced dopaminergic neurotoxicity. Mol Neurobiol 54(5):3783–3797
Priault M, Salin B, Schaeffer J, Vallette FM, di Rago JP, Martinou JC (2005) Impairing the bioenergetic status and the biogenesis of mitochondria triggers mitophagy in yeast. Cell Death Differ 12(12):1613–1621
Rana A, Oliveira MP, Khamoui AV, Aparicio R, Rera M, Rossiter HB, Walker DW (2017) Promoting Drp1-mediated mitochondrial fission in midlife prolongs healthy lifespan of Drosophila melanogaster. Nat Commun 8(1):1–14
Sarkar C, Pal S, Das N, Dinda B (2014) Ameliorative effects of oleanolic acid on fluoride induced metabolic and oxidative dysfunctions in rat brain: experimental and biochemical studies. Food Chem Toxicol 66(4):224–236
Sheridan C, Martin SJ (2010) Mitochondrial fission/fusion dynamics and apoptosis. Mitochondrion 10(6):640–648
Suzuki M, Bartlett JD (2014) Sirtuin1 and autophagy protect cells from fluoride-induced cell stress. Biochim Biophys Acta 1842(2):245–255
Teng Y, Zhang J, Zhang Z, Feng J (2017) The effect of chronic fluorosis on calcium ions and CaMKIIα, and c-fos expression in the Rat hippocampus. Biol Trace Elem Res 182(6):295–302
Tu W, Zhang Q, Liu Y, Han L, Wang Q, Chen P, Zhang S, Wang A, Zhou X (2018) Fluoride induces apoptosis via inhibiting SIRT1 activity to activate mitochondrial p53 pathway in human neuroblastoma SH-SY5Y cells. Toxicol Appl Pharmacol 347:60–69
Westermann B (2010) Mitochondrial fusion and fission in cell life and death. Nat Rev Mol Cell Biol 11(12):872–884
Yan N, Liu Y, Liu S, Cao S, Wang F, Wang Z, Xi S (2016) Fluoride-induced neuron apoptosis and expressions of inflammatory factors by activating microglia in Rat brain. Mol Neurobiol 53(7):4449–4460
Youle RJ, Karbowski M (2005) Mitochondrial fission in apoptosis. Nat Rev Mol Cell Biol 6(8):657–663
Youle RJ, van der Bliek AM (2012) Mitochondrial fission, fusion, and stress. Science 337(6098):1062–1065
Yu X, Chen J, Li Y, Liu H, Hou C, Zeng Q, Cui Y, Zhao L, Li P, Zhou Z (2018) Threshold effects of moderately excessive fluoride exposure on children’s health: a potential association between dental fluorosis and loss of excellent intelligence. Environ Int 118:116–124
Zhang Z, Kageyama Y, Sesaki H (2012) Mitochondrial division prevents neurodegeneration. Autophagy 8(10):1531–1533
Zhang BB, Wang DG, Guo FF, Xuan C (2015a) Mitochondrial membrane potential and reactive oxygen species in cancer stem cells. Fam Cancer 14(1):19–23
Zhang S, Zhang X, Liu H, Qu W, Guan Z, Zeng Q, Jiang C, Gao H, Zhang C, Lei R, Xia T, Wang Z, Yang L, Chen Y, Wu X, Cui Y, Yu L, Wang A (2015b) Modifying effect of COMT gene polymorphism and a predictive role for proteomics analysis in children’s intelligence in endemic fluorosis area in Tianjin, China. Toxicol Sci 144(2):238–245
Zhang S, Chen Y, Xue W, Hui G, Ma R, Jiang C, Gang K, Zhao G, Tao X, Zhang X (2016a) The pivotal role of Ca2+ homeostasis in PBDE-47-induced neuronal apoptosis. Mol Neurobiol 53(10):7078–7088
Zhang S, Niu Q, Gao H, Ma R, Lei R, Zhang C, Xia T, Li P, Xu C, Wang C, Chen J, Dong L, Zhao Q, Wang A (2016b) Excessive apoptosis and defective autophagy contribute to developmental testicular toxicity induced by fluoride. Environ Pollut 212:97–104
Zhang J, Zhu Y, Shi Y, Han Y, Liang C, Feng Z, Zheng H, Eng M, Wang J (2017) Fluoride-induced autophagy via the regulation of phosphorylation of mammalian targets of rapamycin in mice leydig cells. J Agric Food Chem 65(40):8966–8976
Acknowledgements
We would like to express our sincere thanks to all children donors who volunteered to participate in this study. We also sincerely thank the Tianjin Center for Disease Control and Prevention for its assistance for sample collection. This work was supported by grants from the State Key Program of National Natural Science of China (Grant No. 81430076), the National Natural Science Foundation of China (Grants No. 81502785 and No. 81773388) and the Fundamental Research Funds for the Central Universities (HUST 2016YXMS221 and HUST 2015ZDTD052).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Authors declared that there are no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, Q., Niu, Q., Chen, J. et al. Roles of mitochondrial fission inhibition in developmental fluoride neurotoxicity: mechanisms of action in vitro and associations with cognition in rats and children. Arch Toxicol 93, 709–726 (2019). https://doi.org/10.1007/s00204-019-02390-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02390-0