Abstract
Summary
In this nationwide register-based cohort study, we found no difference in the risk of fractures in patients discontinuing versus continuing alendronate (ALN) treatment after 5 years.
Introduction
Information on fracture risk in patients discontinuing ALN in a real-life setting is sparse. We aimed to examine ALN discontinuation patterns, compare fracture rates in patients discontinuing versus continuing ALN after 5 years of treatment, and define determinants of fractures in ALN discontinuers.
Methods
A nationwide population-based cohort study using Danish health registry data. Our source population was individuals who had redeemed ≥ 2 ALN prescriptions between January 1, 1995, and September 1, 2017.
Results
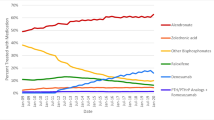
We found that 25% of all ALN initiators used ALN for less than 1 year and 43% continued treatment for at least 5 years. We classified n = 1865 as ALN discontinuers and n = 29,619 as ALN continuers. Using Cox proportional hazards regression analysis and an “as-treated” approach, we observed no increased risk of any fracture (incidence rate ratio (IRR) 1.06, 95% CI 0.92–1.23), vertebral fracture (IRR 0.59, 95% CI 0.33–1.05), hip fracture (IRR 1.04, 95% CI 0.75–1.45), or major osteoporotic fracture (IRR 1.05, 95% CI 0.88–1.25) in the ALN discontinuers compared to continuers during a follow-up time of 1.84 ± 1.56 years (mean ± SD) and 2.51 ± 1.60 years, respectively. ALN re-initiation was a major determinant of follow-up among the discontinuers. Old age (> 80 vs. 50–60 years, unadjusted IRR 2.92, 95% CI 1.18–7.24) was the strongest determinant for fractures following ALN discontinuation.
Conclusion
In a real-world setting, less than 50% continued ALN treatment for 5 years. We found no difference in the risk of fractures in patients discontinuing versus continuing ALN after 5 years.




Similar content being viewed by others
Data availability
The study protocol will be available following publication to investigators who provide a methodologically sound proposal to the corresponding author.
References
Hernlund E, Svedbom A, Ivergård M, Compston J, Cooper C, Stenmark J, McCloskey EV, Jönsson B, Kanis JA (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. Arch Osteoporos 8(136):1–2
Russell RGG, Watts NB, Ebetino FH, Rogers MJ (2008) Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int 19(6):733–759
Porras AG, Holland SD, Gertz BJ (1999) Pharmacokinetics of alendronate. Clin Pharmacokinet 36(5):315–328
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348(9041):1535–1541
Liberman UA, Weiss SR, Bröll J, Minne HW, Quan H, Bell NH, Rodriguez-Portales J, Downs RW Jr, Dequeker J, Favus M, Seeman E, Recker RR, Capizzi T, Santora AC, Lombardi A, Shah RV, Hirsch LJ, Karpf DB (1995) Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med 333(22):1437–1443
Black DM, Schwartz AV, Ensrud KE, Cauley JA, Levis S, Quandt SA, Satterfield S, Wallace RB, Bauer DC, Palermo L, Wehren LE, Lombardi A, Santora AC, Cummings SR, FLEX Research Group (2006) Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA 296(24):2927–2938
Schwartz AV, Bauer DC, Cummings SR, Cauley JA, Ensrud KE, Palermo L, Wallace RB, Hochberg MC, Feldstein AC, Lombardi A, Black DM, for the FLEX Research Group (2010) Efficacy of continued alendronate for fractures in women with and without prevalent vertebral fracture: the FLEX trial. J Bone Miner Res 25(5):976–982
Shane E, Burr D, Abrahamsen B, Adler RA, Brown TD, Cheung AM, Cosman F, Curtis JR, Dell R, Dempster DW, Ebeling PR, Einhorn TA, Genant HK, Geusens P, Klaushofer K, Lane JM, McKiernan F, McKinney R, Ng A, Nieves J, O'Keefe R, Papapoulos S, Howe TS, van der Meulen MCH, Weinstein RS, Whyte MP (2014) Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American society for bone and mineral research. J Bone Miner Res 29(1):1–23
Ruggiero SL, Dodson TB, Fantasia J, Goodday R, Aghaloo T, Mehrotra B, O'Ryan F, American Association of Oral and Maxillofacial Surgeons (2014) American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw—2014 update. J Oral Maxillofac Surg 72(10):1938–1956
Langdahl BL (2019) Management of endocrine disease: treatment breaks in long-term management of osteoporosis. Eur J Endocrinol 180(1):R29–R35
Schmidt M, Pedersen L, Sørensen HT (2014) The Danish Civil Registration System as a tool in epidemiology. Eur J Epidemiol 29(8):541–549
Schmidt M, Schmidt SAJ, Sandegaard JL, Ehrenstein V, Pedersen L, Sørensen HT (2015) The Danish National Patient Registry: a review of content, data quality, and research potential. Clin Epidemiol 7:449
WHO (2010) International statistical classification of diseases and related health problems 10th revision
Pottegård A, Schmidt SAJ, Wallach-Kildemoes H, Sørensen HT, Hallas J, Schmidt M (2017) Data Resource Profile: The Danish National Prescription Registry. Int J Epidemiol 46(3):798–798f
Zhu D, Chung HF, Pandeya N, Dobson AJ, Cade JE, Greenwood DC, Crawford SL, Avis NE, Gold EB, Mitchell ES, Woods NF, Anderson D, Brown DE, Sievert LL, Brunner EJ, Kuh D, Hardy R, Hayashi K, Lee JS, Mizunuma H, Giles GG, Bruinsma F, Tillin T, Simonsen MK, Adami HO, Weiderpass E, Canonico M, Ancelin ML, Demakakos P, Mishra GD (2018) Relationships between intensity, duration, cumulative dose, and timing of smoking with age at menopause: a pooled analysis of individual data from 17 observational studies. PLoS Med 15(11):e1002704. https://doi.org/10.1371/journal.pmed.1002704
Lund JL, Richardson DB, Stürmer T (2015) The active comparator, new user study design in pharmacoepidemiology: historical foundations and contemporary application. Curr Epidemiol Rep 2(4):221–228
Landfeldt E, Ström O, Robbins S, Borgström F (2012) Adherence to treatment of primary osteoporosis and its association to fractures--the Swedish Adherence Register Analysis (SARA). Osteoporos Int 23(2):433–443
Forbes CA, Deshpande S, Sorio-Vilela F, Kutikova L, Duffy S, Gouni-Berthold I, Hagström E (2018) A systematic literature review comparing methods for the measurement of patient persistence and adherence. Curr Med Res Opin 34(9):1613–1625
Malo S, Aguilar-Palacio I, Feja C, Lallana MJ, Rabanaque MJ, Armesto J, Menditto E (2017) Different approaches to the assessment of adherence and persistence with cardiovascular-disease preventive medications. Curr Med Res Opin 33(7):1329–1336
Hjelholt TJ, Edwards NM, Vesterager JD, Kristensen PK, Pedersen AB (2020) The positive predictive value of hip fracture diagnoses and surgical procedure codes in the danish multidisciplinary hip fracture registry and the danish national patient registry. Clin Epidemiol 12:123–131
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Thygesen SK, Christiansen CF, Christensen S, Lash TL, Sørensen HT (2011) The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish National Registry of Patients. BMC Med Res Methodol 11(1):83
Abrahamsen B (2012) Varigheden af behandling med medicin mod osteoporose. Ration Farmakoter (2)
Nandy S, Parsons S, Cryer C, Underwood M, Rashbrook E, Carter Y, Eldridge S, Close J, Skelton D, Taylor S, Feder G, Falls Prevention Pilot Steering Group (2004) Development and preliminary examination of the predictive validity of the Falls Risk Assessment Tool (FRAT) for use in primary care. J Public Health 26(2):138–143
Andersen PK, Geskus RB, de Witte T, Putter H (2012) Competing risks in epidemiology: possibilities and pitfalls. Int J Epidemiol 41(3):861–870
Kanis JA, Hans D, Cooper C, Baim S, Bilezikian JP, Binkley N, Cauley JA, Compston JE, Dawson-Hughes B, Fuleihan GE, Johansson H, Leslie WD, Lewiecki EM, Luckey M, Oden A, Papapoulos SE, Poiana C, Rizzoli R, Wahl DA, McCloskey EV, Task Force of the FRAX Initiative (2011) Interpretation and use of FRAX in clinical practice. Osteoporos Int 22(9):2395–2411
Thygesen LC, Daasnes C, Thaulow I, Brønnum-Hansen H (2011) Introduction to Danish (nationwide) registers on health and social issues: structure, access, legislation, and archiving. Scand J Public Health 39(7 Suppl):12–16
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, STROBE Initiative (2014) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int J Surg 12(12):1495–1499
Fink HA, Milavetz DL, Palermo L, Nevitt MC, Cauley JA, Genant HK, Black DM, Ensrud KE, Fracture Intervention Trial Research Group (2005) What proportion of incident radiographic vertebral deformities is clinically diagnosed and vice versa? J Bone Min Res 20(7):1216–1222
Hudson M, Avina-Zubieta A, Lacaille D, Bernatsky S, Lix L, Jean S (2013) The validity of administrative data to identify hip fractures is high - a systematic review. J Clin Epidemiol 66(3):278–285
Curtis JR, Westfall AO, Cheng H, Delzell E, Saag KG (2008) Risk of hip fracture after bisphosphonate discontinuation: implications for a drug holiday. Osteoporos Int 19(11):1613–1620
Mignot MA, Taisne N, Legroux I, Cortet B, Paccou J (2017) Bisphosphonate drug holidays in postmenopausal osteoporosis: effect on clinical fracture risk. Osteoporos Int 28(12):3431–3438
Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH (2001) Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344(19):1434–1441
Bone HG, Wagman RB, Brandi ML, Brown JP, Chapurlat R, Cummings SR , Czerwiński E, Fahrleitner-Pammer A, Kendler DL, Lippuner K, Reginster J-Y, Roux C, Malouf J, Bradley MN, Daizadeh NS, Wang A, Dakin P, Pannacciulli N, Dempster DW, Papapoulos S (2017) 10 years of denosumab treatment in postmenopausal women with osteoporosis: results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol 5(7):513–523
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J, Glüer CC, Krueger K, Cohen FJ, Eckert S, Ensrud KE, Avioli LV, Lips P, Cummings SR (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators. JAMA 282(7):637–645
Roux C, Reginster J-Y, Fechtenbaum J, Kolta S, Sawicki A, Tulassay Z, Luisetto G, Padrino J-Y, Doyle D, Prince R, Fardellone P, Sorensen OH, Meunier PJ (2006) Vertebral Fracture Risk Reduction With Strontium Ranelate in Women With Postmenopausal Osteoporosis Is Independent of Baseline Risk Factors. J Bone Min. Res 21(4):536–542
Curtis J, et al (2017) The Impact of the Duration of Bisphosphonate Drug Holidays on Hip Fracture Rates. ACR/ARHP Annunal Meetting 2017, Abstract 828
Curtis JR, Saag KG, Arora T, Wright NC, Yun H, Daigle S, Matthews R, Delzell E (2020) Duration of bisphosphonate drug holidays and associated fracture risk. Med Care 58(5):419–426
Adams AL, Adams JL, Raebel MA, Tang BT, Kuntz JL, Vijayadeva V, McGlynn EA, Gozansky WS (2018) Bisphosphonate Drug Holiday and Fracture Risk: A Population-Based Cohort Study. J Bone Min Res 33(7):1252–1259
Nayak S, Greenspan SL (2019) A systematic review and meta-analysis of the effect of bisphosphonate drug holidays on bone mineral density and osteoporotic fracture risk. Osteoporos Int 30(4):705–720
Fatoye F, Smith P, Gebrye T, Yeowell G (2019) Real-world persistence and adherence with oral bisphosphonates for osteoporosis: a systematic review. BMJ Open 9(4):e027049
Pottegård A, dePont Christensen R, Houji A, Christiansen CB, Paulsen MS, Thomsen JL, Hallas J (2014) Primary non-adherence in general practice: a Danish register study. Eur J Clin Pharmacol 70(6):757–763
Hansen C, Pedersen BD, Konradsen H, Abrahamsen B (2013) Anti-osteoporotic therapy in Denmark - predictors and demographics of poor refill compliance and poor persistence. Osteoporos Int 24(7):2079–2097
Adami G, Jaleel A, Curtis JR, Delzell E, Chen R, Yun H, Daigle S, Arora T, Danila MI, Wright NC, Cadarette SM, Mudano A, Foster J, Saag KG (2019) Temporal trends and factors associated with bisphosphonate discontinuation and restart. J Bone Miner Res 35(3):478–487
Chiha M, Myers LE, Ball CA, Sinacore JM, Camacho PM (2013) Long-term follow-up of patients on drug holiday from bisphosphonates: real-world setting. Endocr Pract 19(6):989–994
Bindon B, Adams W, Balasubramanian N, Sandhu J, Camacho P (2018) Osteoporotic fractures during bisphosphonates drug holiday. Endocr Pract 24(2):163–169
Hernán MA, Robins JM (2017) Per-protocol analyses of pragmatic trials. N Engl J Med 377(5):1391–1398
Schneeweiss S, Avorn J (2005) A review of uses of health care utilization databases for epidemiologic research on therapeutics. J Clin Epidemiol 58(4):323–337
Rothman K (2008) Modern Epidemiology, Third edn. Lippincott, Williams, & Wilkins, Philadelphia
Funding
The study was initiated by the investigators. Aarhus University and The Research Foundation of the Department of Endocrinology and Internal Medicine and the Department of Clinical Epidemiology, Aarhus University Hospital, have granted financial support.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study.
Corresponding author
Ethics declarations
Conflicts of interest
Anne Sophie Sølling has nothing to disclose.
Diana Hedevang Christensen has nothing to disclose.
Bianka Darvalics has nothing to disclose.
Torben Harsløf received lecture fees from Amgen, Astra Zeneca, and Eli Lilly.
Reimar Wernich Thomsen has nothing to disclose.
Bente Langdahl is an editor-in-chief of Bone Reports and an associate editor of the European Journal of Endocrinology and has received research funding to her institution from Amgen and Novo Nordisk. Bente Langdahl serves on advisory boards and speaker’s bureau for Amgen, Eli Lilly, Gedeon-Richter, Gilead, and UCB.
The Department of Clinical Epidemiology is involved in studies of osteoporosis with funding from various companies as research grants to (and administered by) Aarhus University.
Disclaimer
The Research Foundation was not involved in the conduct of the study, data collection, interpretation of the data, or the preparation of the manuscript.
Code availability
Please see supplementary Table 1 for International Classification of Diseases, Tenth Revision codes, procedure and NOMESCO surgery codes, and Anatomical Therapeutic Chemical classification system codes used in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sølling, A., Christensen, D., Darvalics, B. et al. Fracture rates in patients discontinuing alendronate treatment in real life: a population-based cohort study. Osteoporos Int 32, 1103–1115 (2021). https://doi.org/10.1007/s00198-020-05745-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-020-05745-x