Abstract
Introduction and hypothesis
Surgical management of uterine prolapse varies greatly and recently uterus-preserving techniques have been gaining popularity. The aim of this study was to compare patient-reported outcomes after cervical amputation versus vaginal hysterectomy, with or without concomitant anterior colporrhaphy, in women suffering from pelvic organ prolapse.
Method
We carried out a population-based longitudinal cohort study with data from the Swedish National Quality Register for Gynecological Surgery. Between 2006 and 2013, a total of 3,174 patients with uterine prolapse were identified, who had undergone primary surgery with either cervical amputation or vaginal hysterectomy, with or without concomitant anterior colporrhaphy. Pre- and postoperative prolapse-related symptoms and patient satisfaction were assessed, in addition to complications and adverse events. Between-group comparisons were performed using univariate and multivariate logistic regression.
Results
There were no differences between the two groups in neither symptom relief nor patient satisfaction. In both groups a total of 81 % of the women reported the absence of vaginal bulging 1 year after surgery and a total of 89 % were satisfied with the result of the operation. The vaginal hysterectomy group had a higher rate of severe complications than the cervical amputation group, 1.9 % vs 0.2 % (p < 0.001). The vaginal hysterectomy group also had a longer duration of surgery and greater perioperative blood loss, in addition to longer hospitalization.
Conclusions
Cervical amputation seems to perform equally well in comparison to vaginal hysterectomy in the treatment of uterine prolapse, but with less morbidity and a lower rate of severe complications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pelvic organ prolapse (POP) is a common condition amongst the female population and the lifetime risk for prolapse or incontinence surgery is 11 % by the age of 80 years, with a 29 % risk of reoperation [1]. Surgical management of uterine prolapse varies greatly and there are currently insufficient data to guide practice [2]. Vaginal hysterectomy, including some kind of vaginal vault suspension, has traditionally been the most common surgical procedure in the treatment of uterine prolapse [1, 3], but uterus-preserving techniques are gaining popularity [4, 5]. Recent studies have shown a desire from the patients to retain the uterus in the case of equal outcome with hysterectomy [6, 7]. Uterus-preserving techniques have been shown to be associated with less morbidity and shorter hospitalization [8] and it has also been suggested that hysterectomy causes damage to the vascular and nerve supply of the pelvis, resulting in bladder dysfunction and recurrence of prolapse [9, 10]. Procedures such as abdominal sacrocolpopexy and sacrospinous fixation have been evaluated in randomized trials and have shown high success rates in comparison to vaginal hysterectomy [2, 11, 12]. Randomized trials evaluating techniques which include amputation of the cervix are lacking. In some papers cervical amputation is even described as an obsolete procedure [12]. However, based on the current literature, procedures including amputation of the cervix appear to be equally as effective with regard to anatomical cure and recurrence rates, but with less morbidity than vaginal hysterectomy [8, 13–15].
The aim of the present study was to compare the outcomes of cervical amputation versus vaginal hysterectomy, with or without concomitant anterior vaginal wall repair, in women suffering from uterine prolapse, using a population-based cohort selected from a national quality register.
Materials and methods
Data source
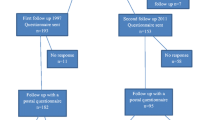
In this longitudinal cohort study data were collected from the Swedish National Quality Register for Gynecological Surgery. The register was established in 1997 and the majority (44 out of 55) of Sweden’s gynecological departments participate. Collection of data concerning POP surgery started in 2006. Data are collected prospectively using patient questionnaires and forms completed by the physicians. The physicians report data on admission, surgery, and discharge, including preoperative gynecological examination of the patient. The preoperative patient questionnaires include questions concerning demographic data and medical history in addition to questions about symptoms associated with POP. Postoperative questionnaires are filled in 8 weeks and 1 year after surgery and include questions about POP symptoms, complications, and patient satisfaction. The study design, including the use of data from the register, was approved by the Regional Board of Ethics in Stockholm, Sweden (Dnr 2014/958-31/4).
Study population
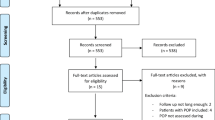
The patients included in the study were women with symptomatic uterine prolapse stage I–IV, with or without anterior vaginal wall prolapse stage I–IV, according to the validated Pelvic Organ Prolapse Quantification System (POP-Q) [16], who have undergone either cervical amputation or vaginal hysterectomy, with or without concomitant anterior colporrhaphy, between January 2006 and December 2013. Exclusion criteria included previous prolapse or urinary incontinence surgery, a uterus larger than 12 weeks’ gestation, and concomitant posterior colporrhaphy or enterocele repair. A total of 4,047 patients, meeting the criteria described above, were identified in the register database.
Description of the procedures
In the case of concomitant anterior colporrhaphy, this procedure is performed first. Anterior colporrhaphy involves a transvaginal incision of the anterior vaginal wall, 2–3 cm posterior to the external urethral meatus almost to the vaginal vault or neck of the cervix, and dissection of the bladder from the vagina. The pubocervical fascia is then adapted in the midline, after which the vaginal epithelium is closed.
In cervical amputation, the cervix is circumcised and the bladder is dissected from the cervical neck. The peritoneum is not opened. After amputation of the cervix, the vaginal epithelium and the cardinal ligaments are often sutured to the stump.
In vaginal hysterectomy the vaginal wall around the cervix is circumcised. After bladder dissection, the anterior and posterior peritoneum is opened. The uterus is released in several steps using clamps and sutures or an electro-thermal bipolar tissue sealing and dividing system. Vault suspension is often performed by suturing the uterosacral or cardinal ligaments to the vaginal vault. The mucosa is then closed.
Outcomes
Baseline data were collected both from forms completed by the physician (age, functional status, and POP-Q) and from the preoperative questionnaire completed by the patient (BMI, parity, smoking, cesarean deliveries, use of estrogen replacement therapy, and menopausal status). Functional outcome measures were retrieved from the questionnaires completed by the patients. The primary outcome was patient-reported vaginal bulging at the 1 year follow-up questionnaire: “Do you experience a feeling of bulging or protrusion in the vaginal area?” The question was dichotomized from five answer options (never or almost never into “no” and 1–3 times per month, 1–3 times per week, and daily into “yes”). Secondary outcomes included surgical complications and adverse events related to the procedure reported by the physician, patient-reported satisfaction, changes in urinary and bowel symptoms, and sexual function 1 year after surgery. Questions about urinary and bowel symptoms were recoded from five answer options into three (never and almost never into “never,” 1–3 times per month, 1–3 times per week into “sometimes,” and daily into “daily”). Sexual activity was reported by answering the question “Have you had coitus in the past 3 months?” Data on complications were reported by the physician at the time of surgery and up to 1 year postoperatively. The severe complications included severe intra-abdominal bleeding, severe intra-abdominal infections or sepsis, ureteric injuries, bowel injuries, myocardial infarction, and severe complications related to the anesthesia. The mild to moderate complications consisted of bladder injuries, lower urinary tract infections or other mild postoperative infections, obstipation, nausea, and prolonged postoperative pain, vaginal bleeding, or fatigue.
Statistical analysis
Baseline characteristics are presented as means ± SD or medians and range for continuous variables and as frequencies for categorical variables. For the comparison of baseline variables between groups we used the Mann–Whitney U test when analyzing continuous data and Fisher’s exact test for categorical variables. Categorical endpoints were analyzed using univariate and multivariate logistic regression, dichotomized outcomes with binary regression and multiple answer outcomes with the proportional odds model. Outcome measures were analyzed using logistic regression. The regression model adjusted for the following variables: surgical method, age, parity, body mass index (BMI), preoperative degree of anterior and apical compartment prolapse (according to the POP-Q), concomitant anterior colporrhaphy, symptoms at baseline, and menopausal status. Results of the logistic regression analyses are presented as odds ratios with 95 % confidence intervals. Comparison of outcomes within groups were analyzed using the Wilcoxon signed rank and McNemar tests. A p value of <0.05 was considered significant for all comparisons. Statistical analyses were performed using SPSS version 22 software (IBM, Armonk, NY, USA).
Results
Study population
Of the 4,047 patients eligible for the study, a total of 3,174 (78 %) women responded to the primary outcome question (sensation of vaginal bulging) in the 1-year follow-up questionnaire. Of the 3,174 respondents, 1,195 patients (38 %) were treated with vaginal hysterectomy and 1,979 patients (62 %) with cervical amputation. There were no significant differences in baseline characteristics between the groups, except for menopausal status (Table 1). Concomitant anterior colporrhaphy was performed in 1,013 patients in the hysterectomy group (85 %) and in 1,757 patients in the cervical amputation group (89 %; Table 1). The relative number of cervical amputations in comparison to vaginal hysterectomies was greater in stage I–II apical descent, whereas the relative number of hysterectomies was higher in stage III–IV (Fig. 1).
Outcome measures
Table 2 describes the mechanical, urinary and bowel symptoms, and sexual activity before and 1 year after surgery within the treatment groups. One year postoperatively, there was significant relief from all urinary and bowel symptoms in both treatment groups. Sexual activity also improved significantly in both groups. Comparisons between vaginal hysterectomy and cervical amputation 1 year after surgery are shown in Table 3. There were no statistical differences between the two groups neither in symptom relief nor in patient satisfaction. In both groups a total of 81 % of the women with preoperative vaginal bulging were asymptomatic 1 year after surgery and a total of 89 % were satisfied with the result of the operation. The results persisted after multivariate logistic regression analysis.
The presence of a vaginal bulging sensation 1 year postoperatively was considered to be symptomatic recurrence and an analysis of the “cured prolapse” versus “recurrent prolapse” group was made to identify risk factors for recurrence. The two groups differed in that the patients with a symptomatic recurrence were somewhat younger (mean age ± SD; 61.5 ± 10.5 vs 63.4 ± 10.3, p = 0.001) and had a higher BMI (mean BMI ± SD; 26.2 ± 4.3 vs 25.7 ± 3.8, p = 0.04) compared with the patients with “cured prolapse.” There were no statistical differences when comparing the two groups considering the severity of prolapse in the apical or anterior compartment, functional status (ASA classification), parity, or whether or not concomitant anterior colporrhaphy was performed.
The primary outcome, the sensation of vaginal bulging 1 year postoperatively, was also evaluated in different subgroups (Table 4). Both techniques performed equally well in every subgroup analysis. The only exception was the subgroup without concomitant anterior colporrhaphy, in which cervical amputation was superior to vaginal hysterectomy.
Adverse events
Adverse events are presented in Table 5. The rate of severe complications was significantly higher in the hysterectomy group than in the cervical amputation group, 23 out of 1,195 (1.9 %) vs 4 out of 1,979 (0.2 %), p < 0.001. The severe complications in the hysterectomy group were: intra-abdominal bleeding (n = 8), severe intra-abdominal infection or sepsis (n = 7), ureteric injuries (n = 4), bowel injuries (n = 2), myocardial infarction (n = 1), and severe complications related to the anesthesia (n = 1). The severe complications in the cervical amputation group consisted of: severe bleeding (n = 2) and severe infection (n = 2). There was no difference in the rate of mild to moderate complications between the groups, 146 out of 1,195 (12.2 %) in the vaginal hysterectomy group versus 246 out of 1,979 (12.4 %) in the cervical amputation group, p = 0.9.
The hysterectomy group had a significantly longer mean duration of surgery (76.2 vs 50.0 min, p < 0.001) and greater mean amount of blood loss (100.1 vs 44.6 ml, p < 0.001) compared with the cervical amputation group. The hysterectomy group also received prophylactic antibiotics and prophylaxis against venous thrombosis to a greater extent, and they had a longer hospitalization (1.7 vs 0.8 days, p < 0.001) and a longer period until return to normal activities of daily living (6.1 vs 4.8 days, p < 0.001) than the cervical amputation group.
Discussion
The results of this large, population-based register study indicate that cervical amputation performs equally as well as vaginal hysterectomy in the treatment of women with uterine prolapse. There was no difference in patient-reported symptom relief and satisfaction 1 year after surgery, but the rate of severe complications was significantly lower in the cervical amputation group. The findings corroborate those of previous studies comparing these two techniques [8, 13, 14]. In the subgroup of patients where no concomitant anterior colporrhaphy was performed, the cervical amputation group had a significantly lower symptomatic recurrence rate 1 year after surgery than the vaginal hysterectomy group, indicating that cervical amputation may be superior to vaginal hysterectomy in the treatment of apical descent.
In this study, cured prolapse was solely symptomatically defined, as we did not have information about postoperative POP-Q status. An absence of vaginal bulge symptoms strongly correlates with patient-reported improvement and treatment success [17]. Using the definition described above, treatment success was similar (81 %) in each group. In a study by Thys et al. comparing the Manchester Fothergill (MF) procedure with vaginal hysterectomy (VH), the objective recurrence rates were 18 % in the MF group and 19 % in the VH group with a median follow-up time of 75 months and there were no differences in POP-related symptoms postoperatively [13]. De Boer et al. compared the modified Manchester procedure (cervical amputation with uterosacral ligament plication) with vaginal hysterectomy. Both procedures performed excellently in the middle compartment [14]. In a review article from 2009 by Dietz et al., the anatomical cure rate in apical support ranged between 93 and 100 % in the Manchester procedure group.
In the present study, the preoperative degree of uterine prolapse was more pronounced in the vaginal hysterectomy group compared with the cervical amputation group (Table 1). This may reflect a general conception that suspension of the vaginal cuff is needed for proper repair of advanced apical prolapse. However, similar treatment results persisted after a multivariate regression analysis adjusting for preoperative POP-Q stage, among other variables. When comparing the results of vaginal hysterectomy with cervical amputation in each subgroup of preoperative POP-Q stage, there were no significant differences. Hence, cervical amputation with ligament attachment can also be considered in advanced uterine prolapse.
Uterine prolapse is often associated with co-existing prolapse in the anterior vaginal wall [18], which is reflected in this study, where the vast majority of patients had concomitant anterior vaginal wall repair. This study cannot answer the question: does the presence of prolapse symptoms postoperatively represent a failure in the apical or the anterior compartment? In both groups, significant improvement was seen in all self-reported bladder dysfunction symptoms, as expected after an anterior colporrhaphy [19, 20]. It was notable, however, that self-reported symptoms of obstructive defecation also became significantly less frequent, indicating that symptoms of uterovaginal prolapse do not necessarily correlate with compartment-specific defects [21].
Increasing age and excess weight are established risk factors for pelvic organ prolapse [1]. In the present study, women with a lower BMI had less symptomatic recurrence than the group with higher BMI. However, symptomatic recurrence correlated inversely with age and the preoperative grade of prolapse did not affect the risk of recurrence. One could speculate that younger women might be more physically active and thus performing “heavy activities” postoperatively to a greater extent, which increases the risk of recurrence.
Various surgical methods have been developed to improve the outcome after surgery for apical prolapse. Sacrocolpopexy and sacrospinous fixation can be performed not only in the treatment of vaginal vault prolapse, but also as uterus-sparing techniques. Open abdominal sacrocolpopexy is the most successful method in the surgical treatment of apical prolapse regarding recurrence rates, but the procedure is associated with an increased length of hospital stay, analgesic requirements, and costs compared with transvaginal procedures [2, 22]. Laparoscopic and robot-assisted sacrocolpopexies also provide excellent short- to medium-term reconstructive outcomes for patients with POP, but involve a shorter recovery time than with open procedures [23]. These techniques, though, can only partly replace the traditional ones, as they require both high-technology operating facilities and experienced surgeons. Their cost-effectiveness is currently unclear [23]. The use of mesh in prolapse surgery has reduced recurrence rates and has therefore been used more frequently over the last decade. However, the benefits must be weighed against the disadvantages, such as mesh erosion (5–10 %) and dyspareunia [24–26]. Sacrospinous hysteropexy is another uterus-preserving technique that is aimed at fixing the uterus to the sacrospinous ligament—most commonly to the right side to prevent lesions of the rectum. A newly published randomized trial by Detollenaere et al. showed equal outcomes of sacrospinous hysteropexy compared with vaginal hysterectomy [11].
Strengths of this study include prospective data collection and a large study population treated in a routine medical care setting. The operating clinics vary from large-scale teaching hospitals to smaller private practitioners and the geographical distribution of the patients is wide, which also increases the external validity. The overall response rate of 78 % must be considered an acceptable figure in a questionnaire-based study and the possibility of response bias is relatively low [27].
The lack of objective measures postoperatively is a limitation of our study, as this could have provided relevant information about prolapse symptoms in relation to anatomical outcome after uterine prolapse surgery. However, the aim of POP surgery is symptom relief regardless of postoperative anatomy. The register data provide no information about the position of the isthmus and cervical length. Therefore, it is not possible to evaluate the influence of possible cervix elongation on the choice of surgical method. The literature contains no clear definition of cervical elongation. Some authors suggest a definition including the corpus uteri/cervix ratio (CCR) of < 1.5 [28, 29]. Using that definition, Mothes et al. found that cervical elongation was present in 97.6 % of patients undergoing hysterectomy due to objective and symptomatic uterine POP stage II–IV, which would suggest that a considerable amount of the patients in the present study might have had some degree of cervical elongation [28]. Suspension of the vaginal apex or cervical end is a standard procedure in vaginal hysterectomy and cervical amputation, but the methods vary. The database does not contain information regarding whether a suspension of the cuff/stump was performed and details of the suspension techniques are not registered. It is, in our opinion, highly important to perform some kind of cuff suspension to restore apical support, as it is not the hysterectomy alone that corrects the prolapse.
The follow-up time of 1 year allows us to evaluate only short-term outcomes. One long-term disadvantage of cervical amputation is the possibility of cervical stenosis and thus complications such as hematometra and difficulties in diagnosing endometrial pathological conditions using histological samples. These complications are not possible to evaluate in the present study.
Another limitation of this study is that the patient questionnaires used in the register database are not validated POP questionnaires. However, the primary outcome question regarding vaginal bulging sensation has been validated.
In conclusion, this study shows no difference between cervical amputation and vaginal hysterectomy in symptomatic outcomes or patient satisfaction in the treatment of uterine prolapse 1 year after surgery. Cervical amputation is a less invasive procedure with a short operation time, reduced blood loss, low complication rates, and fast postoperative recovery time compared with vaginal hysterectomy. The use of prophylactic antibiotics and low molecular weight heparin can also be reduced when performing a cervical amputation rather than a vaginal hysterectomy. Based on these outcomes, we suggest that cervical amputation is a reliable option with few complications in the treatment of women with uterine prolapse, when no other indication for removal of the uterus exists. Randomized controlled trials with long-term follow-up are still needed to evaluate a comparison of cervical amputation not only with vaginal hysterectomy, but also with other uterine-preserving procedures.
References
Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol. 1997;89(4):501–6. doi:10.1016/s0029-7844(97)00058-6.
Maher C, Feiner B, Baessler K, Schmid C. Surgical management of pelvic organ prolapse in women. Cochrane Data Syst Rev. 2013;4:Cd004014. doi:10.1002/14651858.CD004014.pub5.
Jha S, Moran P. The UK national prolapse survey: 5 years on. Int Urogynecol J. 2011;22(5):517–28. doi:10.1007/s00192-011-1379-2.
Detollenaere RJ, den Boon J, Kluivers KB, Vierhout ME, van Eijndhoven HW. Surgical management of pelvic organ prolapse and uterine descent in the Netherlands. Int Urogynecol J. 2013;24(5):781–8. doi:10.1007/s00192-012-1934-5.
Wu MP, Long CY, Huang KH, Chu CC, Liang CC, Tang CH. Changing trends of surgical approaches for uterine prolapse: an 11-year population-based nationwide descriptive study. Int Urogynecol J. 2012;23(7):865–72. doi:10.1007/s00192-011-1647-1.
Frick AC, Barber MD, Paraiso MF, Ridgeway B, Jelovsek JE, Walters MD. Attitudes toward hysterectomy in women undergoing evaluation for uterovaginal prolapse. Female Pelvic Med Reconstruct Surg. 2013;19(2):103–9. doi:10.1097/SPV.0b013e31827d8667.
Korbly NB, Kassis NC, Good MM, Richardson ML, Book NM, Yip S, et al. Patient preferences for uterine preservation and hysterectomy in women with pelvic organ prolapse. Am J Obstet Gynecol. 2013;209(5):470.e1–6. doi:10.1016/j.ajog.2013.08.003.
Dietz V, Schraffordt Koops SE, van der Vaart CH. Vaginal surgery for uterine descent; which options do we have? a review of the literature. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20(3):349–56. doi:10.1007/s00192-008-0779-4.
Altman D, Falconer C, Cnattingius S, Granath F. Pelvic organ prolapse surgery following hysterectomy on benign indications. Am J Obstet Gynecol. 2008;198(5):572.e1–6. doi:10.1016/j.ajog.2008.01.012.
Altman D, Granath F, Cnattingius S, Falconer C. Hysterectomy and risk of stress-urinary-incontinence surgery: nationwide cohort study. Lancet (London, England). 2007;370(9597):1494–9. doi:10.1016/s0140-6736(07)61635-3.
Detollenaere RJ, den Boon J, Stekelenburg J, IntHout J, Vierhout ME, Kluivers KB, et al. Sacrospinous hysteropexy versus vaginal hysterectomy with suspension of the uterosacral ligaments in women with uterine prolapse stage 2 or higher: multicentre randomised non-inferiority trial. BMJ. 2015;351:h3717. doi:10.1136/bmj.h3717.
Betschart C, Cervigni M, Contreras Ortiz O, Doumouchtsis SK, Koyama M, Medina C, et al. Management of apical compartment prolapse (uterine and vault prolapse): a FIGO Working Group report. Neurourol Urodyn. 2015. doi:10.1002/nau.22916.
Thys SD, Coolen A, Martens IR, Oosterbaan HP, Roovers J, Mol B, et al. A comparison of long-term outcome between Manchester Fothergill and vaginal hysterectomy as treatment for uterine descent. Int Urogynecol J. 2011;22(9):1171–8. doi:10.1007/s00192-011-1422-3.
De Boer TA, Milani AL, Kluivers KB, Withagen MI, Vierhout ME. The effectiveness of surgical correction of uterine prolapse: cervical amputation with uterosacral ligament plication (modified Manchester) versus vaginal hysterectomy with high uterosacral ligament plication. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20(11):1313–9. doi:10.1007/s00192-009-0945-3.
Diwan A, Rardin CR, Kohli N. Uterine preservation during surgery for uterovaginal prolapse: a review. Int Urogynecol J Pelvic Floor Dysfunct. 2004;15(4):286–92.
Bump RC, Mattiasson A, Bo K, Brubaker LP, DeLancey JO, Klarskov P, et al. The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obstet Gynecol. 1996;175(1):10–7.
Barber MD, Brubaker L, Nygaard I, Wheeler 2nd TL, Schaffer J, Chen Z, et al. Defining success after surgery for pelvic organ prolapse. Obstet Gynecol. 2009;114(3):600–9. doi:10.1097/AOG.0b013e3181b2b1ae.
Summers A, Winkel LA, Hussain HK, DeLancey JO. The relationship between anterior and apical compartment support. Am J Obstet Gynecol. 2006;194(5):1438–43. doi:10.1016/j.ajog.2006.01.057.
Miedel A, Tegerstedt G, Morlin B, Hammarstrom M. A 5-year prospective follow-up study of vaginal surgery for pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(12):1593–601. doi:10.1007/s00192-008-0702-z.
Weber AM, Walters MD, Piedmonte MR, Ballard LA. Anterior colporrhaphy: a randomized trial of three surgical techniques. Am J Obstet Gynecol. 2001;185(6):1299–304. doi:10.1067/mob.2001.119081. discussion 1304–1296.
Ellerkmann RM, Cundiff GW, Melick CF, Nihira MA, Leffler K, Bent AE. Correlation of symptoms with location and severity of pelvic organ prolapse. Am J Obstet Gynecol. 2001;185(6):1332–7, discussion 1337–8. doi:10.1067/mob.2001.119078.
Sze EH, Kohli N, Miklos JR, Roat T, Karram MM. A retrospective comparison of abdominal sacrocolpopexy with Burch colposuspension versus sacrospinous fixation with transvaginal needle suspension for the management of vaginal vault prolapse and coexisting stress incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 1999;10(6):390–3.
Lee RK, Mottrie A, Payne CK, Waltregny D. A review of the current status of laparoscopic and robot-assisted sacrocolpopexy for pelvic organ prolapse. Eur Urol. 2014;65(6):1128–37. doi:10.1016/j.eururo.2013.12.064.
Cundiff GW, Varner E, Visco AG, Zyczynski HM, Nager CW, Norton PA, et al. Risk factors for mesh/suture erosion following sacral colpopexy. Am J Obstet Gynecol. 2008;199(6):688.e1–5. doi:10.1016/j.ajog.2008.07.029.
Nygaard I, Brubaker L, Zyczynski HM, Cundiff G, Richter H, Gantz M, et al. Long-term outcomes following abdominal sacrocolpopexy for pelvic organ prolapse. JAMA. 2013;309(19):2016–24. doi:10.1001/jama.2013.4919.
Miklos JR, Chinthakanan O, Moore RD, Mitchell GK, Favors S, Karp DR, et al. The IUGA/ICS classification of synthetic mesh complications in female pelvic floor reconstructive surgery: a multicenter study. Int Urogynecol J. 2015. doi:10.1007/s00192-015-2913-4.
Draugalis JR, Coons SJ, Plaza CM. Best practices for survey research reports: a synopsis for authors and reviewers. Am J Pharm Educ. 2008;72(1):11.
Mothes AR, Mothes H, Frober R, Radosa MP, Runnebaum IB. Systematic classification of uterine cervical elongation in patients with pelvic organ prolapse. Eur J Obstet Gynecol Reprod Biol. 2016;200:40–4. doi:10.1016/j.ejogrb.2016.02.029.
Berger MB, Ramanah R, Guire KE, DeLancey JO. Is cervical elongation associated with pelvic organ prolapse? Int Urogynecol J. 2012;23(8):1095–103. doi:10.1007/s00192-012-1747-6.
Acknowledgements
The authors thank Lina Benson at the Department of Clinical Science and Education at Södersjukhuset for support with the statistical analyses. The database was kindly provided by the Swedish National Quality Register of Gynecologic Surgery.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bergman, I., Söderberg, M.W., Kjaeldgaard, A. et al. Cervical amputation versus vaginal hysterectomy: a population-based register study. Int Urogynecol J 28, 257–266 (2017). https://doi.org/10.1007/s00192-016-3119-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-016-3119-0