Abstract
Aneurysmal subarachnoid haemorrhage (aSAH) is a rare yet profoundly debilitating condition associated with high global case fatality and morbidity rates. The key determinants of functional outcome include early brain injury, rebleeding of the ruptured aneurysm and delayed cerebral ischaemia. The only effective way to reduce the risk of rebleeding is to secure the ruptured aneurysm quickly. Prompt diagnosis, transfer to specialized centers, and meticulous management in the intensive care unit (ICU) significantly improved the prognosis of aSAH. Recently, multimodality monitoring with specific interventions to correct pathophysiological imbalances has been proposed. Vigilance extends beyond intracranial concerns to encompass systemic respiratory and haemodynamic monitoring, as derangements in these systems can precipitate secondary brain damage. Challenges persist in treating aSAH patients, exacerbated by a paucity of robust clinical evidence, with many interventions showing no benefit when tested in rigorous clinical trials. Given the growing body of literature in this field and the issuance of contemporary guidelines, our objective is to furnish an updated review of essential principles of ICU management for this patient population. Our review will discuss the epidemiology, initial stabilization, treatment strategies, long-term prognostic factors, the identification and management of post-aSAH complications. We aim to offer practical clinical guidance to intensivists, grounded in current evidence and expert clinical experience, while adhering to a concise format.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The review highlights significant progress in managing aneurysmal subarachnoid hemorrhage (aSAH), underscoring the crucial role of timely diagnosis, transfer to specialized centers, and intensive care in improving prognosis. It presents monitoring and intervention strategies in intensive care aimed at addressing both intracranial and systemic complications, despite persistent challenges due to the lack of robust evidence for many treatments. Future research focusing on closing knowledge gaps, particularly in early and late complications management, is deemed essential to enhance long-term outcomes for aSAH patients. |
Epidemiology and need for intensive care management
Aneurysmal subarachnoid haemorrhage (aSAH) is characterized by bleeding in the subarachnoid space from the rupture of an intracranial aneurysm (Fig. 1) [1]. aSAH accounts for only 2–5% of all strokes, and global incidence declined from 10.2 per 100,000 person-years in 1980 to 6.1 in 2010, with significant variabilities across regions, age, and sex [1]. The global decrease in aSAH incidence paralleled a global reduction in prevalence of hypertension and smoking [1]. The incidence of aSAH increases with age and is 1.3 times higher in women than in men [1, 2]. Although the prognosis of aSAH has improved over the last decades, 12% of patients die before reaching the hospital, and the 90-day case-fatality of patients hospitalized for aSAH is approximately 30% [3]. Survivors often suffer from functional and cognitive sequelae that decrease quality of life and hamper the ability to return to work. Since half of patients with aSAH are younger than 55 years, many are in their most productive working years. Hence, the economic burden is high due to the loss of productive years of life[4]. In the United Kingdom, total costs from aSAH are estimated to be £510 million ($645, €591) per year. On a community level, the loss of productive years of life after aSAH is similar in magnitude to that of ischaemic stroke.
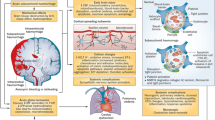
After the rupture of an intracranial aneurysm, a cascade of events ensues. Arterial blood under pressure enters the subarachnoid space, inducing swift mechanical effects, such as abrupt increases in intracranial pressure and related cerebral impact. This sets off intracranial repercussions in the form of early brain injury, accompanied by immediate systemic consequences, impacting cardiovascular and respiratory functions. The presence of blood in the subarachnoid space may contribute to cerebral vasospasm, delayed cerebral ischemia, hydrocephalus, and seizures. Systemically, there can be hyperglycaemia, an inflammatory response, electrolyte imbalances (primarily hypo/hypernatremia), and hormonal disturbances. ICP intracranial pressure, CBF cerebral blood flow, NPO neurogenic pulmonary oedema, ECG electrocardiography, BBB brain blood barrier, DCI delayed cerebral ischemia, LV left ventricle
Major determinants of poor functional outcome and case fatality are early brain injury, rebleeding of the ruptured aneurysm, and delayed cerebral ischaemia (DCI) [5, 6]. Rapid diagnosis, transfer to dedicated centres, management strategies to prevent rebleeding, and haemodynamic management to preserve organ perfusion in the early phase after aSAH can improve the chances of favourable outcomes [7]. Admission to high-volume centres, defined by the management of at least 35 patients per year, is also associated with better functional outcomes after aSAH [8].
Criteria for admission to the intensive care unit (ICU) are not well defined but determined by the need for specialized care to manage early and delayed intracranial and extracranial complications. Management in dedicated neuroICUs has been shown to improve patient outcomes [7]. The optimal duration of ICU management is not well defined. In studies comparing neurointensivist-managed ICUs, the median ICU stay was around 11–12 days, which parallels the time window to address aSAH-related complications [9]. However, these studies do not take into account disease severity. aSAH patients may necessitate critical care management for many weeks. In contrast, good-grade patients may require fewer days of monitoring in the ICU, although often even good-grade aSAH patients benefit from close neurologic observation by specially trained staff during the risk period for vasospasm and DCI.
Clinical presentation and severity scales
Although the clinical presentation of patients with aSAH may vary, the characteristic presenting symptom is a thunderclap headache, a sudden severe headache generally described as the “worst headache of life”. Other symptoms include nausea, vomiting, confusion, disturbance in vision and language, focal neurological deficits and loss of consciousness [1].
Severity scales are based on neurological examinations and early computed tomography (CT) findings [10]. The most widely utilized clinical assessment scale to capture clinical severity in patients with aSAH is the World Federation of Neurosurgical Societies (WFNS) scale [10], (electronic supplementary material, ESM, Figure S1). The WFNS scale (full range 0–5) transforms the Glasgow Coma Scale (GCS) score into different levels (3–6, 7–12, 13–14 with/without motor deficit, and 15). Similarly, the Hunt and Hess Scale is a clinical grading system ranging from a score of 1 to 5, based on neurological symptoms and signs ranging from mild headache to a comatose state [11].
Imaging-based scales such as the Fisher scale [12] or modified Fisher scale [13, 14] quantify the extent of subarachnoid, intraventricular, and intraparenchymal haemorrhage and are associated with outcomes. The Subarachnoid haemorrhage Early Brain Oedema Score (SEBES) has been proposed to quantify global cerebral oedema [15], and seems to be associated with increased intracranial pressure and outcomes after aSAH [16].
Early complications before aneurysm securing
Early brain injury, defined as a cascade of events that develop in the first hours after subarachnoid haemorrhage, is related to a variety of pathophysiological mechanisms, which include cerebral oedema from bleeding, microcirculatory alterations, oxidative and inflammatory cascade, and blood–brain-barrier breakdown, thus leading to neuronal damage [17].
Early management of patients includes intubation and mechanical ventilation in comatose patients to protect the airway and optimize ventilation, neuroimaging (non-contrast head CT, and if negative lumbar puncture, CT angiography, digital subtraction angiography) to establish the diagnosis and guide interventions (i.e. modality of aneurysm treatment, need for diversion of cerebrospinal fluid), treatment of haemodynamic instability, and reversal of coagulopathy [18]. At this stage, a multidisciplinary case discussion for further treatment planning (neurologist, neurosurgeon, interventional neuro-radiologist) is recommended.
Rebleeding
Rebleeding after acute rupture of an intracranial aneurysm markedly increases the risk of poor outcome [18]. It occurs in approximately 3–6% of patients within the first 24 h; the risk rapidly declines afterwards. The only effective way to reduce the risk of rebleeding is to secure the ruptured aneurysm quickly.
For this reason, guidelines recommend aneurysm repair by surgical or endovascular means as soon as feasible, preferably within 24 h. Nevertheless, logistical challenges render this goal difficult, and patients should be haemodynamically stable before aneurysm treatment can be safely pursued [19]. Interestingly, studies have found no benefit with repair within 24 h compared to 24–72 h [20].
The initial instability of the blood clot sealing the defect in the aneurysm is the target of antifibrinolytics. In several initial, randomized-controlled trials, tranexamic acid had been associated with reduced rebleeding [21], but longer-term use (weeks) was also associated with an increased risk of DCI. However, a recent large, adequately powered randomized controlled trial (RCT) that investigated ultra-early and short-term treatment with tranexamic acid found no difference in outcome, and antifibrinolytic use has since been widely abandoned [22].
Acute aSAH can be associated with a sympathetic response and rise in blood pressure, aggravating the risk of rebleeding. While intuitive, no high-quality data exist that treatment of hypertension reduces the risk of rebleeding [23]. Additionally, hypotension should also be avoided, as the lack of adequate perfusion of viable brain in the setting of elevated intracranial pressure may lead to acute cerebral ischaemia. The thresholds to lower systolic arterial blood pressure, commonly set at a goal of < 160 mmHg, for these risks are likely unique to each patient and ideally should be individualized in relation to the patient's status and baseline arterial blood pressure values.
Hydrocephalus
Hydrocephalus is a common acute or, less often, delayed complication after aSAH (Figs. 1, 2). Although it is more frequent in patients with intraventricular haemorrhage (especially when the third and fourth ventricles are filled with blood), it can develop in the absence of visible intraventricular extension of the bleeding on head CT [24]. Its mechanism can be obstructive (typically by clot impeding cerebrospinal fluid flow through the aqueduct), malabsorptive (from lack of cerebrospinal fluid absorption by the arachnoid granulations) [24], or both. Clinical presentation is characterized by a depressed level of alertness and, in severe cases, by downgaze deviation (from compression of the mesencephalic tectum by the dilated third ventricle), and a combination of bradycardia and hypertension.
Intracranial complications after aneurysmal subarachnoid haemorrhage (aSAH) and their management. ICP intracranial pressure, EVD external ventricular drain, DCI delayed cerebral ischemia, TCD transcranial doppler, TC computed tomography, CSF cerebrospinal fluid, EEG electroencephalography, MRI magnetic resonance imaging, ABP arterial blood pressure
Cerebrospinal fluid drainage by external ventricular drain (EVD), lumbar drain or lumbar puncture should be performed in patients with hydrocephalus to reduce intracranial pressure, thereby improving cerebral perfusion [25]. Ventriculostomy is the method of choice in aSAH patients with obstructive hydrocephalus and those with extensive intraventricular haemorrhage. Inserting an EVD before aneurysm occlusion can protect against abrupt ICP increases in case of rebleeding [26]. However, attention should be paid to excessive ventricular drainage before aneurysm obliteration which could prompt rerupture.
Treatment of the aneurysm (surgical clipping/endovascular treatment)
Timely repair of ruptured aneurysms reduces the risk of rebleeding and allows for more targeted and safer management of DCI [27]. Studies suggest that early aneurysm securement, i.e. within 24–72 h from onset of aSAH, yields better outcomes than delayed treatment after 7–10 days [28]. Complete obliteration of the aneurysm during initial treatment is vital to minimize rebleeding risk and risks associated with the need for retreatment. In cases where complete obliteration is not immediately feasible, securing the rupture site during the acute phase reduces the risk of early rebleeding. Retreatment within 1–3 months is then recommended to prevent future rebleeding [29].
The choice of the method to treat the ruptured aneurysm is complex and necessitates a careful balance between securing the aneurysm and the associated procedural risks [30]. This multidisciplinary decision-making process involves evaluating patient- and aneurysm-specific factors, including age, aneurysm characteristics (such as location, size, and configuration) and degree of intraparenchymal clot burden.
The two primary approaches for aneurysm treatment are open surgical techniques and endovascular methods. Each option presents distinct advantages and disadvantages. Coiling is generally preferred over open clipping if the aneurysm is amenable to such a technique, although differential considerations apply based on the setting. Good-grade aSAH patients from ruptured aneurysms of the anterior circulation are often equally suitable for both primary coiling and clipping; primary coiling is generally preferred to clipping as it is linked to improved 1-year functional outcomes [30, 31]. Similarly, some demographical subpopulations, such as older patients, may be preferably treated with coiling, whereas in case of longer life expectancy and better long-term protection from re-rupture, clipping should be taken into consideration [27]. In addition, most posterior circulation aneurysms benefit from endovascular coiling over surgical clipping [31]. Finally, aneurysm morphology (e.g. wide neck) may favour one procedure over another, and placing high-density stents (such as pipeline) over the necks of aneurysms unsuitable for clipping or coiling can be considered. However, the evidence supporting treatment recommendations is somewhat limited, particularly concerning the comparison of various, especially newer, endovascular techniques with each other and surgical methods, and in most cases, the choice is made according to clinical features but also local practice and expertise [30].
Management in the ICU
After aneurysm securement, the ICU management of aSAH involves close monitoring, optimization of systemic function and preventing and treating cranial and systemic complications. A comprehensive multidisciplinary team is fundamental to provide appropriate and aggressive treatment of these patients [32]. Minimizing or avoiding sedation seems to provide a shorter duration of mechanical ventilation and length of hospital stay and allows appropriate clinical neuromonitoring for early detection of neurodeterioration [33]. However, sedation may be required to manage intracranial hypertension, pain, agitation, status epilepticus or severe respiratory failure. The choice of sedative needs to be balanced with the patients' clinical conditions. Midazolam and propofol are most frequently adopted as first-line sedatives, but ketamine appears promising [33].
Intracranial complications
Delayed cerebral ischaemia
DCI occurs in about 30% of patients with aneurysmal aSAH, mostly between days 4–14 after ictus, and is the leading cause of functional disability in survivors [34, 35] (Figs. 2, 3). It is often associated with angiographic vasospasm, but documentation of vasospasm is not a sine-qua-non for the diagnosis of DCI [36]. Only half of patients with angiographic vasospasm develop ischaemic symptoms, and DCI may develop without angiographic vasospasm [34]. Recent work has suggested that other factors may contribute to DCI; it also occurs in cortical spreading depolarization [37], impaired autoregulation with reduced regional blood flow, intravascular volume contraction, and microthrombosis [38]. Vasospasm can affect small vessels escaping angiographic detection. Poor initial clinical (i.e. Hunt Hess or WFNS score) and radiographic (i.e. modified Fisher, Hijdra) grades [39] are established predictors of DCI. Other risk factors, variably associated, include female sex [40], smoking, hydrocephalus, hyperglycaemia, and diabetes [41].
Detection of causes of delayed cerebral ischemia (DCI). CT computed tomography, CTA CT angiography, DSA digital subtraction angiography, CTP perfusion computed tomography, MRI magnetic resonance imaging, TCD transcranial Doppler, PbtO2 brain tissue oxygen, ICP intracranial pressure, EEG electroencephalography, NPi neurological pupillary index, ADR alpha/delta ratio, LR Lindegaard ratio
Guideline-recommended strategies to reduce DCI risk include enteral administration of nimodipine and prevention of hypovolaemia and hypotension [42]. Nimodipine reduces infarction rates and improves functional outcomes despite not significantly preventing angiographic vasospasm [43]. Intravenous nimodipine more often leads to hypotension with compared to oral nimodipine, with significant drops in blood pressure in one-third of patients after the start of intravenous nimodipine as opposed to after every tenth oral intake [30] Hypotension may require dose adjustment, discontinuation, or vasopressor support increase [44]. Nimodipine can also be used as a potential intraarterial vasodilator [45], although robust data on eventual burden of DCI are lacking for such use.
A summary of measures aimed at DCI prevention and treatment is provided in Table 1, and Fig. 3.
As preventive measures are limited, detecting and treating DCI before it leads to cerebral infarction is important. Clinical monitoring through serial neurologic assessments is fundamental but misses DCI-related neurologic changes in one-fifth of patients [46]. Hence, imaging aimed at vasospasm detection and assessment of cerebral perfusion is commonly entertained. The gold standard for diagnosis of large-vessel vasospasm, digital subtraction angiography, enables endovascular treatment if indicated. Computed tomography-angiography (CTA) reaches 82% and 97% sensitivity and specificity for angiographic vasospasm detection [47]. CT perfusion may allow recognition of impaired perfusion, which may occur independently of vasospasm [48].
Transcranial Doppler ultrasonography (TCD) is noninvasive and can be performed daily or even more often, but it lacks high sensitivity for vasospasm detection, reaching only 38% in a recent review [49]. In addition, agreement between CTA and TCD is limited [50].
Continuous electroencephalography (cEEG) with quantitative analysis can predict the development of DCI earlier than TCD. When combining cEEG and TCD, diagnostic accuracy to predict DCI increases [51]. Invasive DCI monitoring modalities include cerebral oxygenation, brain-tissue biochemistry, electrocorticography, and intracortical EEG (Fig. 3) [52]. If DCI is detected, physiological and biological factors such as sedation, hyperthermia, and arterial blood gases should be considered (Fig. 3).
No data are available to support the treatment of subclinical vasospasm detected by monitoring. Therefore, at the current stage of knowledge, these techniques mainly aim at identifying patients at higher risk of DCI and may guide stricter monitoring or lower thresholds to trigger further examination or more advanced management.
Treating DCI aims to improve perfusion and minimize or prevent infarction. Haemodynamic augmentation through induced hypertension is often used as a primary intervention in patients with DCI in the absence of cardiac failure and severe baseline hypertension. Induced hypertension may reduce the risk of DCI-related cerebral infarction and lead to better outcomes [52], but optimal target blood pressures remain unclear, and the only randomized controlled trial on this topic failed to support induced hypertension in this setting but likely was underpowered [53]. Haemodynamic augmentation appears safe in the presence of other small, unruptured and untreated aneurysms [54]. Vasopressor selection depends on cardiovascular status; norepinephrine and phenylephrine are commonly used. While cardiac output changes generally do not affect cerebral blood flow, they may do so in SAH patients. Accordingly, inotropic agents, such as milrinone (that also has cerebral vasodilatory properties), may be useful in patients with ventricular dysfunction.
Endovascular rescue therapy with either intraarterial infusion of vasodilators (i.e. verapamil, nicardipine or milrinone) and/or balloon angioplasty may be considered when haemodynamic augmentation fails or is contraindicated [55]. There is no evidence supporting these interventions from phase 3 RCTs, although a recent small phase 2 RCT [56] showed worse outcomes in patients treated with endovascular rescue therapy than those treated with hypertension induction. Retrospective data suggest improved outcomes in centres offering endovascular intervention for DCI compared to those that do not [56].
Seizures and status epilepticus
Seizures in aSAH may be encountered at the time of bleeding, during hospitalization, and months or years later (ESM, Table S1). Underlying mechanisms are debated and may include gliosis, neuroinflammation [57], and cerebral hyperaemia [58]. The SAFARI score may help identify those at risk for convulsive in-hospital seizures based on older age, pre-hospitalization seizures, ruptured anterior circulation aneurysms, and ventricular drainage [59]. Recent guidelines [30] state that cEEG is reasonable, primarily for aSAH patients with impaired consciousness or fluctuating examination, ruptured middle cerebral artery aneurysm, high-grade aSAH, intracranial haemorrhage, hydrocephalus, and cortical infarction. Periodic discharges not qualifying as seizures [60] are prevalent and of controversial significance. Long-term epilepsy appears less likely in patients who underwent coil embolization [27].
Electrographic seizures [61] and high-frequency periodic discharges (> 2.5 Hz) [62] have been associated with increased metabolic demand that may be insufficiently compensated for by local increases in cerebral blood flow. In aSAH patients during and post-hospitalization [63], electrographic seizures have been associated with worse outcomes. Importantly, these associations do not imply causal effects and possibly relate to other secondary complications, such as late diagnosis of delayed cerebral ischaemia [64].
No adequately powered clinical trial has been conducted to guide seizure prophylaxis or treatment in aSAH patients. Recent guidelines [30] do not recommend the routine use of prophylactic antiseizure medication (ASM), although ASM may be reasonable in patients at high risk for seizures. Phenytoin as ASM should be avoided since it may induce harm. In an underpowered, prospective, single-centre, randomized, open-label trial, the benefit from prolonged levetiracetam prophylaxis was greatest for those with imaging evidence of early brain injury [65] and others have suggested a possible benefit against DCI with newer ASMs (levetiracetam and perampanel) [66]. All clinical or electrographic seizures that are diagnosed should be treated for at least 7 days and for those with delayed seizures or those with risk factors for seizures, more prolonged treatment should be considered. Management of periodic discharges or other ictal-interictal patterns is highly controversial, and the benefit of treatment has not been demonstrated [67].
ICP indications, management and cerebral oxygenation
The practice of intracranial pressure (ICP) monitoring in patients with aSAH remains a matter of debate due to the absence of high-level evidence tailored to this condition [68] (Fig. 2). While the Neurocritical Care Society suggests ICP monitoring for acute brain injuries at risk of elevated ICP based on clinical and imaging features, there are no distinct indications for aSAH patients [69]. However, in certain circumstances, ICP monitoring should be considered in aSAH, including GCS ≤ 8, neurological deterioration, acute hydrocephalus, cerebral oedema, intracranial mass lesions, and need for perioperative or drainage of cerbrospinal fluid (CSF) [30].
The gold standard method for ICP measurement is via a ventricular catheter connected to a pressure transducer [39,40,41]. This approach also allows for CSF drainage and recalibration. Of note, while potential benefits of fibrinolytic treatment of intraventricular haemorrhage have been demonstrated for primary intracranial haemorrhage, there are no data to support this for the aSAH population [70, 71].
Drainage practices after aneurysm treatment vary across centres and include open drainage by adjustment of the drainage system above the foramen of Monro or intermittent drainage based on ICP values [26]. Timing, triggers of weaning and how to discontinue ventricular drainage are subject to debate [26]. Reports suggest that direct clamping may be preferable to weaning before clamping [72], but the evidence is weak.
A lumbar drain is considered a less invasive alternative to EVD but may be precluded in case or obstructive hydrocephalus and contraindicated based on head CT findings [73]. In a recent trial, prophylactic lumbar drainage after aSAH reduced secondary infarctions and the rate of an unfavourable outcome at 6 months [74]. These findings support the use of lumbar drains after aSAH, but additional research is necessary to determine the value of lumbar drainage to improve outcomes after aSAH, especially because an earlier RCT found no effect of lumbar drainage on outcome at 6 months [75].
Patients receiving lumbar punctures need to be carefully monitored to confirm that they do not need additional CSF diversion.
Ventriculoperitoneal shunting is necessary in patients with persistent hydrocephalus. Risk factors for requiring ventriculoperitoneal shunting include intraventricular haemorrhage and higher radiological grade (such as modified Fisher), older age, higher clinical grade (Hunt and Hess or WFNS), more significant acute ventricular dilatation, rebleeding, posterior location of the ruptured aneurysm, and multiple clamp failures [76].
Alternative noninvasive methods for estimating ICP are being explored, including ultrasound assessment of optic nerve sheath diameter, although their reliability and use in aSAH patients is questionable [77].
The appropriate threshold for treating elevated ICP remains poorly understood. While the classic threshold is around 20–22 mmHg, poor outcomes have also been linked to lower values [78], suggesting a potential need to lower ICP targets. Recent studies indicate that a “dose” concept, considering both the magnitude and duration of exposure to high ICP, may better quantify the ICP burden and predict outcomes [79].
Elevated ICP in aSAH may arise primarily from hydrocephalus (30%), intracerebral haemorrhage (ICH), and early or delayed global cerebral oedema (GCO) (8–12%) [68, 80]. Less common causes include subdural hematoma, cerebral infarction, and extracranial factors. aSAH-related ICP elevation can occur acutely, subacutely, within a few days from bleeding, or delayed. Early GCO risk is linked to cerebral hypoperfusion, vasomotor paralysis, and blood volume increase [81]. Later, ICP may normalize due to reduced blood, improved CSF dynamics, and brain oedema reduction. Further risk of GCO arises from ischaemic injury, ion channel dysfunction, and cellular swelling.
The management of increased ICP in aSAH patients lacks specific guidelines, prompting the application of recommendations from traumatic brain injury (TBI) guidelines [76].
The SYNAPSE-ICU study [82] revealed that episodes of high ICP necessitating treatment were frequent in aSAH patients, especially those with parenchymal devices, and ICP monitoring and treatment were associated with improved outcomes [83].
When intracranial hypertension is caused by hydrocephalus, CSF removal is vital for controlling ICP, with current guidelines recommending CSF diversion for acute symptomatic hydrocephalus [84].
Hyperventilation, head elevation, and osmotherapy are commonly used to manage high ICP. The transient effect of hyperventilation makes it suitable for short-term ICP control but carries the risk of cerebral ischemia and should be avoided in patients at risk for DCI, while the choice of osmotherapy between mannitol and hypertonic saline remains debatable [84].
Hypothermia and high-dose barbiturates are reserved for refractory cases due to their risks and limited supporting evidence [84]. Decompressive craniectomy (DC) has shown efficacy in reducing ICP, but its impact on functional outcomes is questionable [84]. While evidence for the routine use of DC for managing elevated ICP in SAH is lacking, it remains a last resort option when medical management fails. Finally, cerebral monitoring with brain tissue oxygen tension (PbtO2) can be taken into consideration—although evidence on PbtO2 monitoring mainly stems from traumatic brain-injured patients; reductions in brain oxygen tension have been associated with angiographic vasospasm or altered regional cerebral blood flow [85], and recent evidence supports its use for the detection of DCI in selected patients [85].
Extracranial complications
Cardiac complications
While the primary concern in the setting of aSAH is its direct effects on the brain through primary and secondary brain injury, the post-bleeding course can be complicated by an array of cardiac adverse events ranging from arrhythmias and myocardial dysfunction to myocardial infarction [86]. Cardiac complications increase the complexity of medical management and can worsen prognosis of patients with aSAH [87]. Therefore, prompt medical management of both the neurological and cardiac aspects of aSAH is crucial to improve outcomes and reduce the risk of further complications (Figs. 1, 4).
Extracranial complications after aneurysmal subarachnoid haemorrhage. T temperature, ABG arterial blood gases, MAP mean arterial pressure, ECG electrocardiography, PBW predicted body weight, DP driving pressure, Pplateau Plateau pressure, PEEP positive end-expiratory pressure, TV tidal volume, PPlat plateau pressure, SpO2 oxygen saturation, NPO neurogenic pulmonary oedema, VAP ventilator-associated pneumonia, ARDS acute respiratory distress syndrome, PE pulmonary embolism, DVT deep venous thrombosis, SIADH syndrome of inappropriate antidiuretic hormone, CWS cerebral salt-wasting, CXR chest X-ray, CT computed tomography, LMWH low molecular weight heparin
aSAH can disrupt the electrical sinus pacing or cause conduction abnormalities, leading to arrhythmias [88]. Encountered rhythm abnormalities include sinus arrhythmia, atrial fibrillation, ventricular tachycardia, or ventricular fibrillation. Prompt correction of electrolyte abnormalities is important, and use of antiarrhythmic medications may be required. Serial electrocardiograms (ECG), troponin sampling, and continuous monitoring of vital signs in an intensive care setting are recommended [89]. In addition, more advanced haemodynamic monitoring, including pulse index continuous cardiac output (PICCO), pulse dye densitometry, and pulmonary artery catheterization, can be considered to minimize cerebral and haemodynamic complications in specific cases [90].
aSAH can also result in wall motion abnormalities [91]. These changes are thought to result from the massive catecholamine release and sympathetic surge during ictus bleed and are typically not reflective of an anatomic territory of the coronary arteries. Decreased cardiac output and impaired ejection fraction may ensue, which could result in hypotension, fluid overload, and heart failure. In most cases, these changes are reversible and will resolve without sequelae.
In more severe cases, patients may present with profound neurogenic myocardial stunning, called Takotsubo cardiomyopathy [92]. This condition is characterized by biventricular apical hypo- or akinesia, leading to signs and symptoms mimicking a myocardial infarction, such as chest pain, shortness of breath, abnormal ECG with changes characterized by ST-segment elevation, and troponin elevation [93]. Echocardiography can assist in the differential diagnosis of myocardial infarction. The important distinction from ischaemic cardiac disease is that aSAH-related cardiac findings are non-ischaemic due to disturbance of the contraction bands in cardiac muscle and are fully reversible.
Infrequently, aSAH can trigger ischaemic myocardial infarction [94]. This complication can occur due to a combination of factors, including increased sympathetic activity, arterial spasm, and microvascular dysfunction in a susceptible individual, mostly in the setting of underlying coronary artery disease. The risk of myocardial infarction is highest within the first few days after aSAH. A multidisciplinary approach involving cardiology, neurosurgery, and neurointensivists for determination of the appropriateness of prompt initiation of management of acute coronary syndrome should be coordinated in a timely fashion.
Pulmonary complications
aSAH patients are at risk for pulmonary complications, which contribute to worse outcomes and mortality. Patients with a decrease in neurologic function also are at increased risk for aspiration pneumonia due to loss of control of swallowing [95]. Mechanical ventilation for airway protection can lead to ventilator-associated pneumonia. In a large prospective multicentre study of aSAH in the United States (US) and Canada from 1995 (50 hospitals), pneumonia occurred in 22% of aSAH cases [96] (Fig. 4).
Neurogenic pulmonary oedema (NPO), caused by increased interstitial and alveolar lung fluid, occurs as a consequence of central nervous system injury and, when most severe, can result in acute respiratory distress syndrome (ARDS) [97]. It is an early complication, occurring in up to 23% of cases, and is thought to be related to sympathetic activation due to the SAH [96]. Treatment of NPO is supportive, including mechanical ventilation with positive end-expiratory pressure (PEEP), and reduction of ICP. Euvolemia is central to the management of NPO and aSAH and must balance the slightly competing imperatives (avoidance of hypervolemia in NPO and avoidance of hypovolemia in aSAH).
Classic ARDS, characterized by hypoxemic respiratory failure associated with diffuse lung inflammation and increased alveolar-capillary permeability, generally occurs as a response to injury (such as pneumonia, sepsis, and aspiration) in post-aSAH patients [98]. The incidence of ARDS increases with aSAH severity, including cerebral oedema, cardiac arrest and cardiogenic shock. Unsurprisingly, ARDS is also associated with significantly worse outcomes [99]. It is encouraging that the incidence of ARDS after aSAH has decreased from 38% in 2008 to 4% in 2014 [100]. It is also noted to be of similarly low incidence (< 4%) in a large multicentre European cohort in 2021 [101]. Lung-protective ventilation strategies for ARDS (low tidal volume, titrated positive end-expiratory pressure, PEEP, limiting plateau pressure and driving pressure, cautious recruitment manoeuvres and permissive hypercapnia) started to become the standard of care around 2000 (Figs. 2, 3) [102]. The declining incidence of ARDS with evolving management of both aSAH and ARDS suggests that this is a preventable condition and should be a target for improving outcomes after aSAH [103].
Metabolic and fluid management
Metabolic and fluid management in aSAH patients is an essential aspect of neurointensive care, aiming to maintain an even fluid balance, prevent complications, and support optimal cerebral perfusion [104]. A key concept of haemodynamic management is the optimization of cardiovascular function to ensure sufficient cerebral perfusion pressure and oxygen delivery to the brain. Haemodynamic optimization is essentially achieved by avoiding hypovolemia, maintaining adequate blood pressure, and targeting optimal cardiac output by ensuring adequate preload (intravascular volume) and contractility based on the Starling curve principle. Goal-directed therapy is a reasonable approach to achieve the hemodynamic optimization goals. The specific targets may vary based on individual patient factors and institutional protocols; commonly used parameters include mean arterial pressure (MAP) goals with a target MAP > 65 to 70 mmHg and cardiac output (CO) or cardiac index (CI), to guide fluid administration, vasopressor dosing and inotropic support [105].
Invasive blood pressure monitoring with arterial catheterization is advisable for most cases. Noninvasive cardiac output monitoring, central venous catheterization, or—in select and severe cases—pulmonary artery catheterization may be utilized to closely monitor hemodynamic parameters, especially in patients requiring active intervention with fluids and vasopressors or inotropes. Accurate and frequent monitoring of fluid intake, urine output, and clinical assessment of hydration status is crucial.
Assessing fluid responsiveness through dynamic indices (e.g. stroke volume variation, pulse pressure variation) or bedside echocardiography can help guide fluid administration, and these approaches are likely most reliable in determining the intravascular volume and guiding fluid administration.
Electrolyte imbalances, including hypokalaemia, hypomagnesemia and hypophosphatemia, are common in aSAH patients, primarily due to excess renal losses. They can significantly affect neurological and cardiac function [106]; monitoring and prompt correction of electrolyte abnormalities are important aspects of aSAH management to prevent arrhythmias or other cardiac abnormalities.
Hyponatremia has been attributed to the syndrome of inappropriate antidiuretic hormone (SIADH), excessive salt wasting, hypovolemia or a combination of both [107]. Determination of intravascular volume status along with urinary osmolarity and sodium excretion may be used to determine the cause of hyponatremia and guide the approach to its correction that may include free water optimization, avoiding fluid restriction, sodium supplementation (hypertonic saline or enteral replacement), or occasionally administration of mineralocorticoids, even if debatable [108].
Adequate nutrition is essential for recovery. Oral or enteral feeding is preferred over parenteral nutrition when possible. Hyperglycaemia is associated with worse outcomes [109]. Glucose levels should be closely monitored, and if elevated, appropriate insulin therapy may be initiated to maintain glycaemia in a typical range between 140 and 180 mg/dL [110].
It is important to note that individual patient characteristics, the severity of aSAH, and other co-existing conditions may influence the specific approach to metabolic and fluid management. Thus, these strategies should be tailored to each patient's unique circumstances, and close monitoring and collaboration with a multidisciplinary team are crucial for optimal management.
Fever is extremely common in patients after aSAH; early, i.e. within the first 3 days, fever is frequently neurogenic. Its occurrence (regardless of the cause, neurogenic, infective, or drug-related) is associated with worse outcomes. Recent recommendations suggest active fever treatment, continuous core temperature monitoring, and avoiding temperatures > 37.5° [111]. Other principles for the general management of these patients and their complications include deep venous thrombosis prophylaxis after aneurysm treatment to prevent thromboembolic events, and screening/ treatment for systemic infective complications.
Long-term outcomes of SAH patients
Long-term outcomes of aSAH have improved over the years, even in patients presenting with very severe illness [112]. A favourable functional outcome is achieved in a third of poor-grade aSAH patients [112]. Many factors have likely led to this improvement, including earlier timing of aneurysm treatment [113], and specialized neurocritical care management [114]. There is tremendous variability in aSAH care [115] and associated outcomes [116], and evidence-based and guideline-driven care [30] have the potential to improve population outcomes for aSAH. Long-term outcomes in aSAH trials and observational studies have been historically measured on scales of function such as modified Rankin Score (mRS) or Glasgow Outcome Scale or Extended (GOS/GOSE), and are dichotomized by researchers inconsistently into good or poor outcomes.
Importantly, functional outcome measures can have poor agreement with patients' self-assessed outcomes after aSAH [117]. The recorded outcome measures often miss important aspects, as reported by patients and families: cognitive, psychological, and social dependence [118]. Even if patients improve in physical function, most patients suffer from neurocognitive morbidity, which is a driver of the economic burden of aSAH [35]. Memory deficits and impairment in executive functions are most common and are closely related to impaired quality of life, which is reported in a third of survivors a year after ictus [35]. More than half of patients with good neurological outcomes a year after aSAH still had mild to moderate difficulties even 20 years out [119]. Unfortunately, these symptoms can be underrecognized and underdocumented, and current prognostic models are limited in their assessment [120]. The high prevalence of neurocognitive morbidity despite functional independence underscores the importance of measures of quality of life in outcomes assessments and as targets of prevention and intervention. Outcome tools for the prognosis after aSAH have been proposed, such as the SAHOT (subarachnoid haemorrhage outcome tool) for individual assessment of prognosis after aSAH [121], but these need to be better implemented and validated in clinical practice. Importantly, post-ICU clinical trajectory needs to be taken into consideration through rehabilitation programs, assessments and follow-up to minimize long-term symptoms such as anxiety, depression, and fatigue and ensure a better quality of life.
Conclusions and future perspectives
aSAH still has high mortality and morbidity, which significantly depend on the initial severity and the occurrence of early and late complications. Management of this entity in specialized centres with dedicated personnel may improve outcomes. A growing body of evidence is available regarding the ICU management of these patients, but still significant knowledge gaps and a lack of high-quality data remain. Future research and randomized controlled trials are warranted in order to understand better the management of early and late complications in this population, especially regarding DCI detection and treatment (in the ESM we present a list of the ongoing trials on aSAH). In particular, research should focus on the aim to address the gaps in the understanding of aSAH pathophysiology and strategies that could improve long term outcomes.
References
Etminan N, Chang H-S, Hackenberg K, De Rooij NK, Vergouwen MDI, Rinkel GJE, Algra A (2019) Worldwide incidence of aneurysmal subarachnoid haemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: a systematic review and meta-analysis. JAMA Neurol 76:588. https://doi.org/10.1001/jamaneurol.2019.0006
Rinkel GJE, Djibuti M, Algra A, Van Gijn J (1998) Prevalence and risk of rupture of intracranial aneurysms: a systematic review. Stroke 29:251–256. https://doi.org/10.1161/01.STR.29.1.251
Lovelock CE, Rinkel GJE, Rothwell PM (2010) Time trends in outcome of subarachnoid haemorrhage: population-based study and systematic review. Neurology 74:1494–1501. https://doi.org/10.1212/WNL.0b013e3181dd42b3
Rivero-Arias O, Gray A, Wolstenholme J (2010) Burden of disease and costs of aneurysmal subarachnoid haemorrhage (aSAH) in the United Kingdom. Cost Eff Resour Alloc 8:6. https://doi.org/10.1186/1478-7547-8-6
Roos YBWEM (2000) Complications and outcome in patients with aneurysmal subarachnoid haemorrhage: a prospective hospital based cohort study in The Netherlands. J Neurol Neurosurg Psychiatry 68:337–341. https://doi.org/10.1136/jnnp.68.3.337
Vergouwen MDI, Jong-Tjien-Fa AV, Algra A, Rinkel GJE (2016) Time trends in causes of death after aneurysmal subarachnoid haemorrhage: a hospital-based study. Neurology 86:59–63. https://doi.org/10.1212/WNL.0000000000002239
Busl KM, Bleck TP, Varelas PN (2019) Neurocritical care outcomes, research, and technology: a review. JAMA Neurol 76:612. https://doi.org/10.1001/jamaneurol.2018.4407
Leifer D, Fonarow GC, Hellkamp A, Baker D, Hoh BL, Prabhakaran S, Schoeberl M, Suter R, Washington C, Williams S, Xian Y, Schwamm LH (2021) Association between hospital volumes and clinical outcomes for patients with nontraumatic subarachnoid haemorrhage. J Am Heart Assoc 10:e018373. https://doi.org/10.1161/JAHA.120.018373
Egawa S, Hifumi T, Kawakita K, Okauchi M, Shindo A, Kawanishi M, Tamiya T, Kuroda Y (2016) Impact of neurointensivist-managed intensive care unit implementation on patient outcomes after aneurysmal subarachnoid haemorrhage. J Crit Care 32:52–55. https://doi.org/10.1016/j.jcrc.2015.11.008
Teasdale GM, Drake CG, Hunt W, Kassell N, Sano K, Pertuiset B, De Villiers JC (1988) A universal subarachnoid haemorrhage scale: report of a committee of the World Federation of Neurosurgical Societies. J Neurol Neurosurg Psychiatry 51:1457–1457. https://doi.org/10.1136/jnnp.51.11.1457
Hunt WE, Hess RM (1968) Surgical risk as related to time of intervention in the repair of intracranial aneurysms. J Neurosurg 28:14–20. https://doi.org/10.3171/jns.1968.28.1.0014
Fisher CM, Kistler JP, Davis JM (1980) Relation of cerebral vasospasm to subarachnoid haemorrhage visualized by computerized tomographic scanning. Neurosurgery 6:1–9. https://doi.org/10.1227/00006123-198001000-00001
Claassen J, Bernardini GL, Kreiter K, Bates J, Du YE, Copeland D, Connolly ES, Mayer SA (2001) Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid haemorrhage: the fisher scale revisited. Stroke 32:2012–2020. https://doi.org/10.1161/hs0901.095677
Frontera JA, Claassen J, Schmidt JM, Wartenberg KE, Temes R, Connolly ES, Macdonald RL, Mayer SA (2006) Prediction of symptomatic vasospasm after subarachnoid haemorrhage: the modified fisher scale. Neurosurgery 59:21–27. https://doi.org/10.1227/01.neu.0000243277.86222.6c
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA (2002) Global cerebral oedema after subarachnoid haemorrhage: frequency, predictors, and impact on outcome. Stroke 33:1225–1232. https://doi.org/10.1161/01.STR.0000015624.29071.1F
Ahn S-H, Savarraj JP, Pervez M, Jones W, Park J, Jeon S-B, Kwon SU, Chang TR, Lee K, Kim DH, Day AL, Choi HA (2018) The subarachnoid haemorrhage early brain oedema score predicts delayed cerebral ischemia and clinical outcomes. Neurosurgery 83:137–145. https://doi.org/10.1093/neuros/nyx364
Lauzier DC, Jayaraman K, Yuan JY, Diwan D, Vellimana AK, Osbun JW, Chatterjee AR, Athiraman U, Dhar R, Zipfel GJ (2023) Early brain injury after subarachnoid haemorrhage: incidence and mechanisms. Stroke 54:1426–1440. https://doi.org/10.1161/STROKEAHA.122.040072
Brilstra EH, Rinkel GJE, Algra A, Van Gijn J (2000) Rebleeding, secondary ischemia, and timing of operation in patients with subarachnoid haemorrhage. Neurology 55:1656–1660. https://doi.org/10.1212/WNL.55.11.1656
Connolly ES, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J, Higashida RT, Hoh BL, Kirkness CJ, Naidech AM, Ogilvy CS, Patel AB, Thompson BG, Vespa P (2012) Guidelines for the management of aneurysmal subarachnoid haemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 43:1711–1737. https://doi.org/10.1161/STR.0b013e3182587839
Oudshoorn SC, Rinkel GJE, Molyneux AJ, Kerr RS, Dorhout Mees SM, Backes D, Algra A, Vergouwen MDI (2014) Aneurysm treatment <24 versus 24–72 h after subarachnoid haemorrhage. Neurocrit Care 21:4–13. https://doi.org/10.1007/s12028-014-9969-8
Vermeulen M, Lindsay KW, Murray GD, Cheah F, Hijdra A, Muizelaar JP, Schannong M, Teasdale GM, Van Crevel H, Van Gijn J (1984) Antifibrinolytic treatment in subarachnoid haemorrhage. N Engl J Med 311:432–437. https://doi.org/10.1056/NEJM198408163110703
Post R, Germans MR, Tjerkstra MA, Vergouwen MDI, Jellema K, Koot RW, Kruyt ND, Willems PWA, Wolfs JFC, De Beer FC, Kieft H, Nanda D, Van Der Pol B, Roks G, De Beer F, Halkes PHA, Reichman LJA, Brouwers PJAM, Van Den Berg-Vos RM, Kwa VIH, Van Der Ree TC, Bronner I, Van De Vlekkert J, Bienfait HP, Boogaarts HD, Klijn CJM, Van Den Berg R, Coert BA, Horn J, Majoie CBLM, Rinkel GJE, Roos YBWEM, Vandertop WP, Verbaan D, Post R, Germans MR, Tjerkstra MA, Vergouwen MDI, Jellema K, Koot RW, Kruyt ND, Willems PWA, Wolfs JFC, De Beer FC, Kieft H, Nanda D, Van Der Pol B, Roks G, De Beer F, Halkes PHA, Reichman LJA, Brouwers PJAM, Van Den Berg-Vos RM, Kwa VIH, Van Der Ree TC, Bronner I, Bienfait HP, Boogaarts HD, Klijn CJM, Van Bilzen M, Dieks HJG, De Gans K, Ten Holter JBM, De Kruijk JR, Leijzer CTJM, Molenaar D, Van Oostenbrugge RJ, Van Pamelen J, Spaander FHM, Vermeer SE, Van De Vlekkert J, Voorend JM, Van Den Berg R, Coert BA, Horn J, Majoie CBLM, Rinkel GJE, Roos YBWEM, Vandertop WP, Verbaan D (2021) Ultra-early tranexamic acid after subarachnoid haemorrhage (ULTRA): a randomised controlled trial. The Lancet 397:112–118. https://doi.org/10.1016/S0140-6736(20)32518-6
Rosengart AJ, Schultheiss KE, Tolentino J, Macdonald RL (2007) Prognostic factors for outcome in patients with aneurysmal subarachnoid haemorrhage. Stroke 38:2315–2321. https://doi.org/10.1161/STROKEAHA.107.484360
Chen S, Luo J, Reis C, Manaenko A, Zhang J (2017) Hydrocephalus after subarachnoid haemorrhage: pathophysiology, diagnosis, and treatment. BioMed Res Int 2017:1–8. https://doi.org/10.1155/2017/8584753
Van Asch CJJ, Van Der Schaaf IC, Rinkel GJE (2010) Acute hydrocephalus and cerebral perfusion after aneurysmal subarachnoid haemorrhage. Am J Neuroradiol 31:67–70. https://doi.org/10.3174/ajnr.A1748
Chung DY, Leslie-Mazwi TM, Patel AB, Rordorf GA (2017) Management of external ventricular drains after subarachnoid haemorrhage: a multi-institutional survey. Neurocrit Care 26:356–361. https://doi.org/10.1007/s12028-016-0352-9
Molyneux AJ, Kerr RS, Yu L-M, Clarke M, Sneade M, Yarnold JA, Sandercock P (2005) International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomized comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet 366:809–817. https://doi.org/10.1016/S0140-6736(05)67214-5
Rawal S, Alcaide-Leon P, Macdonald RL, Rinkel GJE, Victor JC, Krings T, Kapral MK, Laupacis A (2017) Meta-analysis of timing of endovascular aneurysm treatment in subarachnoid haemorrhage: inconsistent results of early treatment within 1 day. J Neurol Neurosurg Psychiatry 88:241–248. https://doi.org/10.1136/jnnp-2016-314596
Campi A, Ramzi N, Molyneux AJ, Summers PE, Kerr RSC, Sneade M, Yarnold JA, Rischmiller J, Byrne JV (2007) Retreatment of ruptured cerebral aneurysms in patients randomized by coiling or clipping in the international subarachnoid aneurysm trial (ISAT). Stroke 38:1538–1544. https://doi.org/10.1161/STROKEAHA.106.466987
Hoh BL, Ko NU, Amin-Hanjani S, Hsiang-Yi Chou S, Cruz-Flores S, Dangayach NS, Derdeyn CP, Du R, Hänggi D, Hetts SW, Ifejika NL, Johnson R, Keigher KM, Leslie-Mazwi TM, Lucke-Wold B, Rabinstein AA, Robicsek SA, Stapleton CJ, Suarez JI, Tjoumakaris SI, Welch BG (2023) 2023 Guideline for the management of patients with aneurysmal subarachnoid haemorrhage: a guideline from the American Heart Association/American Stroke Association. Stroke. https://doi.org/10.1161/STR.0000000000000436
Algra A, Brilstra E, Clarke M, Van Gijn J, Molyneux A, Rinkel G, Van Rooij W (2001) Endovascular coiling versus neurosurgical clipping for patients with aneurysmal subarachnoid haemorrhage. In: The Cochrane Collaboration (ed) The Cochrane Database of systematic reviews. Wiley, Chichester, p CD003085
Green DM, Burns JD, DeFusco CM (2013) ICU management of aneurysmal subarachnoid haemorrhage. J Intensive Care Med 28:341–354. https://doi.org/10.1177/0885066611434100
Oddo M, Crippa IA, Mehta S, Menon D, Payen J-F, Taccone FS, Citerio G (2016) Optimizing sedation in patients with acute brain injury. Crit Care 20:128. https://doi.org/10.1186/s13054-016-1294-5
Vergouwen MDI, Vermeulen M, Van Gijn J, Rinkel GJE, Wijdicks EF, Muizelaar JP, Mendelow AD, Juvela S, Yonas H, Terbrugge KG, Macdonald RL, Diringer MN, Broderick JP, Dreier JP, Roos YBWEM (2010) Definition of delayed cerebral ischemia after aneurysmal subarachnoid haemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke 41:2391–2395. https://doi.org/10.1161/STROKEAHA.110.589275
Thompson JC, Chalet F-X, Manalastas EJ, Hawkins N, Sarri G, Talbot DA (2022) Economic and humanistic burden of cerebral vasospasm and its related complications after aneurysmal subarachnoid haemorrhage: a systematic literature review. Neurol Ther 11:597–620. https://doi.org/10.1007/s40120-022-00348-6
Vergouwen MDI, Ilodigwe D, Macdonald RL (2011) Cerebral infarction after subarachnoid haemorrhage contributes to poor outcome by vasospasm-dependent and -independent effects. Stroke 42:924–929. https://doi.org/10.1161/STROKEAHA.110.597914
Dreier JP, Woitzik J, Fabricius M, Bhatia R, Major S, Drenckhahn C, Lehmann T-N, Sarrafzadeh A, Willumsen L, Hartings JA, Sakowitz OW, Seemann JH, Thieme A, Lauritzen M, Strong AJ (2006) Delayed ischaemic neurological deficits after subarachnoid haemorrhage are associated with clusters of spreading depolarizations. Brain 129:3224–3237. https://doi.org/10.1093/brain/awl297
Vergouw LJM, Egal M, Bergmans B, Dippel DWJ, Lingsma HF, Vergouwen MDI, Willems PWA, Oldenbeuving AW, Bakker J, Van Der Jagt M (2020) High early fluid input after aneurysmal subarachnoid haemorrhage: combined report of association with delayed cerebral ischemia and feasibility of cardiac output-guided fluid restriction. J Intensive Care Med 35:161–169. https://doi.org/10.1177/0885066617732747
Van Der Steen WE, Leemans EL, Van Den Berg R, Roos YBWEM, Marquering HA, Verbaan D, Majoie CBLM (2019) Radiological scales predicting delayed cerebral ischemia in subarachnoid haemorrhage: systematic review and meta-analysis. Neuroradiology 61:247–256. https://doi.org/10.1007/s00234-019-02161-9
Rehman S, Phan HT, Chandra RV, Gall S (2022) Is sex a predictor for delayed cerebral ischaemia (DCI) and hydrocephalus after aneurysmal subarachnoid haemorrhage (aSAH)? A systematic review and meta-analysis. Acta Neurochir (Wien) 165:199–210. https://doi.org/10.1007/s00701-022-05399-0
De Rooij NK, Rinkel GJE, Dankbaar JW, Frijns CJM (2013) Delayed cerebral ischemia after subarachnoid haemorrhage: a systematic review of clinical, laboratory, and radiological predictors. Stroke 44:43–54. https://doi.org/10.1161/STROKEAHA.112.674291
Oddo M, Poole D, Helbok R, Meyfroidt G, Stocchetti N, Bouzat P, Cecconi M, Geeraerts T, Martin-Loeches I, Quintard H, Taccone FS, Geocadin RG, Hemphill C, Ichai C, Menon D, Payen J-F, Perner A, Smith M, Suarez J, Videtta W, Zanier ER, Citerio G (2018) Fluid therapy in neurointensive care patients: ESICM consensus and clinical practice recommendations. Intensive Care Med 44:449–463. https://doi.org/10.1007/s00134-018-5086-z
Hao G, Chu G, Pan P, Han Y, Ai Y, Shi Z, Liang G (2022) Clinical effectiveness of nimodipine for the prevention of poor outcome after aneurysmal subarachnoid haemorrhage: a systematic review and meta-analysis. Front Neurol 13:982498. https://doi.org/10.3389/fneur.2022.982498
Rass V, Kindl P, Lindner A, Kofler M, Altmann K, Putnina L, Ianosi B-A, Schiefecker AJ, Beer R, Pfausler B, Helbok R (2023) Blood pressure changes in association with nimodipine therapy in patients with spontaneous subarachnoid haemorrhage. Neurocrit Care 39:104–115. https://doi.org/10.1007/s12028-023-01760-y
Diringer MN, Bleck TP, Claude Hemphill J, Menon D, Shutter L, Vespa P, Bruder N, Connolly ES, Citerio G, Gress D, Hänggi D, Hoh BL, Lanzino G, Le Roux P, Rabinstein A, Schmutzhard E, Stocchetti N, Suarez JI, Treggiari M, Tseng M-Y, Vergouwen MDI, Wolf S, Zipfel G (2011) Critical care management of patients following aneurysmal subarachnoid haemorrhage: recommendations from the neurocritical care society’s multidisciplinary consensus conference. Neurocrit Care 15:211. https://doi.org/10.1007/s12028-011-9605-9
LaBuzetta JN, Hirshman BR, Malhotra A, Owens RL, Kamdar BB (2022) Practices and patterns of hourly neurochecks: analysis of 8,936 patients with neurological injury. J Intensive Care Med 37:784–792. https://doi.org/10.1177/08850666211029220
Allen JW, Prater A, Kallas O, Abidi SA, Howard BM, Tong F, Agarwal S, Yaghi S, Dehkharghani S (2022) Diagnostic performance of computed tomography angiography and computed tomography perfusion tissue time-to-maximum in vasospasm following aneurysmal subarachnoid haemorrhage. J Am Heart Assoc 11:e023828. https://doi.org/10.1161/JAHA.121.023828
Mir DIA, Gupta A, Dunning A, Puchi L, Robinson CL, Epstein H-AB, Sanelli PC (2014) CT Perfusion for detection of delayed cerebral ischemia in aneurysmal subarachnoid haemorrhage: a systematic review and meta-analysis. Am J Neuroradiol 35:866–871. https://doi.org/10.3174/ajnr.A3787
Khawaja AM, McNulty J, Thakur UV, Chawla S, Devi S, Liew A, Mirshahi S, Du R, Mekary RA, Gormley W (2022) Transcranial Doppler and computed tomography angiography for detecting cerebral vasospasm post-aneurysmal subarachnoid haemorrhage. Neurosurg Rev 46:3. https://doi.org/10.1007/s10143-022-01913-1
Van Der Harst JJ, Luijckx G-JR, Elting JWJ, Bokkers RPH, Van Den Bergh WM, Eshghi OS, Metzemaekers JDM, Groen RJM, Mazuri A, Van Dijk JMC, Uyttenboogaart M (2019) Transcranial Doppler versus CT-angiography for detection of cerebral vasospasm in relation to delayed cerebral ischemia after aneurysmal subarachnoid haemorrhage: a prospective single-center cohort study: the transcranial doppler and CT-angiography for Investigating Cerebral vasospasm in Subarachnoid haemorrhage (TACTICS) study. Crit Care Explor 1:e0001. https://doi.org/10.1097/CCE.0000000000000001
Gollwitzer S, Groemer T, Rampp S, Hagge M, Olmes D, Huttner HB, Schwab S, Madžar D, Hopfengaertner R, Hamer HM (2015) Early prediction of delayed cerebral ischemia in subarachnoid haemorrhage based on quantitative EEG: a prospective study in adults. Clin Neurophysiol 126:1514–1523. https://doi.org/10.1016/j.clinph.2014.10.215
Haegens NM, Gathier CS, Horn J, Coert BA, Verbaan D, Van Den Bergh WM (2018) Induced hypertension in preventing cerebral infarction in delayed cerebral ischemia after subarachnoid haemorrhage. Stroke 49:2630–2636. https://doi.org/10.1161/STROKEAHA.118.022310
Allen ML, Kulik T, Keyrouz SG, Dhar R (2019) Posterior reversible encephalopathy syndrome as a complication of induced hypertension in subarachnoid haemorrhage: a case-control study. Neurosurgery 85:223–230. https://doi.org/10.1093/neuros/nyy240
Hoh BL, Carter BS, Ogilvy CS (2002) Risk of haemorrhage from unsecured, unruptured aneurysms during and after hypertensive hypervolemic therapy. Neurosurgery 50:1207–1212. https://doi.org/10.1097/00006123-200206000-00006
Suwatcharangkoon S, De Marchis GM, Witsch J, Meyers E, Velazquez A, Falo C, Schmidt JM, Agarwal S, Connolly ES, Claassen J, Mayer SA (2019) Medical treatment failure for symptomatic vasospasm after subarachnoid haemorrhage threatens long-term outcome. Stroke 50:1696–1702. https://doi.org/10.1161/STROKEAHA.118.022536
Vatter H, Güresir E, König R, Durner G, Kalff R, Schuss P, Mayer TE, Konczalla J, Hattingen E, Seifert V, Berkefeld J (2022) Invasive diagnostic and therapeutic management of cerebral vasospasm after aneurysmal subarachnoid haemorrhage (IMCVS)—a phase 2 randomized controlled trial. J Clin Med 11:6197. https://doi.org/10.3390/jcm11206197
Claassen J, Albers D, Schmidt JM, De Marchis GM, Pugin D, Falo CM, Mayer SA, Cremers S, Agarwal S, Elkind MSV, Connolly ES, Dukic V, Hripcsak G, Badjatia N (2014) Nonconvulsive seizures in subarachnoid haemorrhage link inflammation and outcome: seizures in SAH. Ann Neurol 75:771–781. https://doi.org/10.1002/ana.24166
Alkhachroum A, Megjhani M, Terilli K, Rubinos C, Ford J, Wallace BK, Roh DJ, Agarwal S, Connolly ES, Boehme AK, Claassen J, Park S (2020) Hyperemia in subarachnoid haemorrhage patients is associated with an increased risk of seizures. J Cereb Blood Flow Metab 40:1290–1299. https://doi.org/10.1177/0271678X19863028
Jaja BNR, Schweizer TA, Claassen J, Le Roux P, Mayer SA, Macdonald RL, Collaborators SAHIT, Noble A, Molyneux A, Quinn A, Schatlo B, Lo B, Jaja BNR, Hanggi D, Hasan D, Wong GKC, Etminan N, Lantigua H, Fukuda H, Torner J, Singh J, Suarez JI, Spears J, Schaller K, Stienen MN, Vergouwen MDI, Cusimano MD, Todd M, Tseng M-Y, Le Roux P, Macdonald RL, Johnston SC, Yamagata S, Mayer S, Schenk T, Schweizer TA, Van Den Bergh W (2018) The SAFARI Score to assess the risk of convulsive seizure during admission for aneurysmal subarachnoid haemorrhage. Neurosurgery 82:887–893. https://doi.org/10.1093/neuros/nyx334
Hirsch LJ, Fong MWK, Leitinger M, LaRoche SM, Beniczky S, Abend NS, Lee JW, Wusthoff CJ, Hahn CD, Westover MB, Gerard EE, Herman ST, Haider HA, Osman G, Rodriguez-Ruiz A, Maciel CB, Gilmore EJ, Fernandez A, Rosenthal ES, Claassen J, Husain AM, Yoo JY, So EL, Kaplan PW, Nuwer MR, Van Putten M, Sutter R, Drislane FW, Trinka E, Gaspard N (2021) American clinical neurophysiology society’s standardized critical care EEG terminology: 2021 version. J Clin Neurophysiol 38:1–29. https://doi.org/10.1097/WNP.0000000000000806
Claassen J, Perotte A, Albers D, Kleinberg S, Schmidt JM, Tu B, Badjatia N, Lantigua H, Hirsch LJ, Mayer SA, Connolly ES, Hripcsak G (2013) Nonconvulsive seizures after subarachnoid haemorrhage: multimodal detection and outcomes: Claassen et al.: effects of Seizures after SAH. Ann Neurol 74:53–64. https://doi.org/10.1002/ana.23859
Messina A, Villa F, Lionetti G, Galarza L, Meyfroidt G, Van Der Jagt M, Monnet X, Pelosi P, Cecconi M, Robba C (2022) Hemodynamic management of acute brain injury caused by cerebrovascular diseases: a survey of the European Society of Intensive Care Medicine. Intensive Care Med Exp 10:42. https://doi.org/10.1186/s40635-022-00463-6
Bögli SY, Wang S, Romaguera N, Schütz V, Rafi O, Gilone M, Keller E, Imbach LL, Brandi G (2022) Impact of seizures and status epilepticus on outcome in patients with aneurysmal subarachnoid haemorrhage. Neurocrit Care 36:751–759. https://doi.org/10.1007/s12028-022-01489-0
Rosenthal ES, Biswal S, Zafar SF, O’Connor KL, Bechek S, Shenoy AV, Boyle EJ, Shafi MM, Gilmore EJ, Foreman BP, Gaspard N, Leslie-Mazwi TM, Rosand J, Hoch DB, Ayata C, Cash SS, Cole AJ, Patel AB, Westover MB (2018) Continuous electroencephalography predicts delayed cerebral ischemia after subarachnoid haemorrhage: a prospective study of diagnostic accuracy. Ann Neurol 83:958–969. https://doi.org/10.1002/ana.25232
Human T, Diringer MN, Allen M, Zipfel GJ, Chicoine M, Dacey R, Dhar R (2018) A randomized trial of brief versus extended seizure prophylaxis after aneurysmal subarachnoid haemorrhage. Neurocrit Care 28:169–174. https://doi.org/10.1007/s12028-017-0440-5
Suzuki H, Miura Y, Yasuda R, Yago T, Mizutani H, Ichikawa T, Miyazaki T, Kitano Y, Nishikawa H, Kawakita F, Fujimoto M, Toma N (2022) Effects of new-generation antiepileptic drug prophylaxis on delayed neurovascular events after aneurysmal subarachnoid haemorrhage. Transl Stroke Res. https://doi.org/10.1007/s12975-022-01101-9
Zafar SF, Rosenthal ES, Postma EN, Sanches P, Ayub MA, Rajan S, Kim JA, Rubin DB, Lee H, Patel AB, Hsu J, Patorno E, Westover MB (2022) Antiseizure medication treatment and outcomes in patients with subarachnoid haemorrhage undergoing continuous EEG monitoring. Neurocrit Care 36:857–867. https://doi.org/10.1007/s12028-021-01387-x
Addis A, Baggiani M, Citerio G (2023) Intracranial pressure monitoring and management in aneurysmal subarachnoid haemorrhage. Neurocrit Care. https://doi.org/10.1007/s12028-023-01752-y
Le Roux P, Menon DK, Citerio G, Vespa P, Bader MK, Brophy GM, Diringer MN, Stocchetti N, Videtta W, Armonda R, Badjatia N, Böesel J, Chesnut R, Chou S, Claassen J, Czosnyka M, De Georgia M, Figaji A, Fugate J, Helbok R, Horowitz D, Hutchinson P, Kumar M, McNett M, Miller C, Naidech A, Oddo M, Olson D, O’Phelan K, Provencio JJ, Puppo C, Riker R, Robertson C, Schmidt M, Taccone F (2014) Consensus summary statement of the International Multidisciplinary Consensus Conference on Multimodality Monitoring in Neurocritical Care: a statement for healthcare professionals from the Neurocritical Care Society and the European Society of Intensive Care Medicine. Intensive Care Med 40:1189–1209. https://doi.org/10.1007/s00134-014-3369-6
Hanley DF, Lane K, McBee N, Ziai W, Tuhrim S, Lees KR, Dawson J, Gandhi D, Ullman N, Mould WA, Mayo SW, Mendelow AD, Gregson B, Butcher K, Vespa P, Wright DW, Kase CS, Carhuapoma JR, Keyl PM, Diener-West M, Muschelli J, Betz JF, Thompson CB, Sugar EA, Yenokyan G, Janis S, John S, Harnof S, Lopez GA, Aldrich EF, Harrigan MR, Ansari S, Jallo J, Caron J-L, LeDoux D, Adeoye O, Zuccarello M, Adams HP, Rosenblum M, Thompson RE, Awad IA (2017) Thrombolytic removal of intraventricular haemorrhage in treatment of severe stroke: results of the randomized, multicentre, multiregion, placebo-controlled CLEAR III trial. Lancet 389:603–611. https://doi.org/10.1016/S0140-6736(16)32410-2
Kuramatsu JB, Gerner ST, Ziai W, Bardutzky J, Sembill JA, Sprügel MI, Mrochen A, Kölbl K, Ram M, Avadhani R, Falcone GJ, Selim MH, Lioutas V-A, Endres M, Zweynert S, Vajkoczy P, Ringleb PA, Purrucker JC, Volkmann J, Neugebauer H, Erbguth F, Schellinger PD, Knappe UJ, Fink GR, Dohmen C, Minnerup J, Reichmann H, Schneider H, Röther J, Reimann G, Schwarz M, Bäzner H, Claßen J, Michalski D, Witte OW, Günther A, Hamann GF, Lücking H, Dörfler A, Ishfaq MF, Chang JJ, Testai FD, Woo D, Alexandrov AV, Staykov D, Goyal N, Tsivgoulis G, Sheth KN, Awad IA, Schwab S, Hanley DF, Huttner HB, Sansing L, Matouk CC, Leasure A, Sobesky J, Bauer M, Schurig J, Rizos T, Haeusler KG, Müllges W, Kraft P, Schubert A-L, Stösser S, Ludolph AC, Nueckel M, Glahn J, Stetefeld H, Rahmig J, Fisse AL, Michels P, Schwert H, Hagemann G, Rakers F, Wöhrle JC, Alshammari F, Horn M, Bahner D, Urbanek C, Palm F, Grau A (2022) Association of intraventricular fibrinolysis with clinical outcomes in intracerebral haemorrhage: an individual participant data meta-analysis. Stroke 53:2876–2886. https://doi.org/10.1161/STROKEAHA.121.038455
Chung DY, Thompson BB, Kumar MA, Mahta A, Rao SS, Lai JH, Tadevosyan A, Kessler K, Locascio JJ, Patel AB, Mohamed W, Olson DM, John S, Rordorf GA (2022) Association of external ventricular drain wean strategy with shunt placement and length of stay in subarachnoid haemorrhage: a prospective multicenter study. Neurocrit Care 36:536–545. https://doi.org/10.1007/s12028-021-01343-9
Wolf S (2015) Rationale for lumbar drains in aneurysmal subarachnoid haemorrhage. Curr Opin Crit Care 21:120–126. https://doi.org/10.1097/MCC.0000000000000183
Wolf S, Mielke D, Barner C, Malinova V, Kerz T, Wostrack M, Czorlich P, Salih F, Engel DC, Ehlert A, Staykov D, Alturki AY, Sure U, Bardutzky J, Schroeder HWS, Schürer L, Beck J, Juratli TA, Fritsch M, Lemcke J, Pohrt A, Meyer B, Schwab S, Rohde V, Vajkoczy P, EARLYDRAIN Study Group, Baro N, Bauer M, Dengler NF, Von Dincklage F, Finger T, Francis R, Hotter B, Hunsicker O, Jussen D, Jüttler E, Schaumann A, Witsch J, Nagel C, Meier U, Podlesik D, Schackert G, Huttner H, Hagedorn S, Müller D, Müller O, Sarge R, Niesen W-D, Lange K, Päsler D, Reinhardt S, Regelsberger J, Sauvigny T, Westphal M, Gremmer R, Beyer C, Beyer D, Huthmann A, Landscheidt J, Schul DB, Ryang Y-M, Toeroek E, Arouk W, Al-Jehani H, Sinclair DB, Fung C, Soell N, Hildebrandt G, Huscher K, Lange H, Hutchinson P, Tseng M-Y (2023) Effectiveness of lumbar cerebrospinal fluid drain among patients with aneurysmal subarachnoid haemorrhage: a randomized clinical trial. JAMA Neurol 80:833. https://doi.org/10.1001/jamaneurol.2023.1792
Al-Tamimi YZ, Bhargava D, Feltbower RG, Hall G, Goddard AJP, Quinn AC, Ross SA (2012) Lumbar drainage of cerebrospinal fluid after aneurysmal subarachnoid haemorrhage: a prospective, randomized, controlled trial (LUMAS). Stroke 43:677–682. https://doi.org/10.1161/STROKEAHA.111.625731
Wilson CD, Safavi-Abbasi S, Sun H, Kalani MYS, Zhao YD, Levitt MR, Hanel RA, Sauvageau E, Mapstone TB, Albuquerque FC, McDougall CG, Nakaji P, Spetzler RF (2017) Meta-analysis and systematic review of risk factors for shunt dependency after aneurysmal subarachnoid haemorrhage. J Neurosurg 126:586–595. https://doi.org/10.3171/2015.11.JNS152094
McIver JI, Friedman JA, Wijdicks EFM, Piepgras DG, Pichelmann MA, Toussaint LG, McClelland RL, Nichols DA, Atkinson JLD (2002) Preoperative ventriculostomy and rebleeding after aneurysmal subarachnoid haemorrhage. J Neurosurg 97:1042–1044. https://doi.org/10.3171/jns.2002.97.5.1042
Cagnazzo F, Chalard K, Lefevre P-H, Garnier O, Derraz I, Dargazanli C, Gascou G, Riquelme C, Bonafe A, Perrini P, Di Carlo DT, Morganti R, Le Corre M, Pavillard F, Perrigault P-F, Costalat V (2021) Optimal intracranial pressure in patients with aneurysmal subarachnoid haemorrhage treated with coiling and requiring external ventricular drainage. Neurosurg Rev 44:1191–1204. https://doi.org/10.1007/s10143-020-01322-2
Magni F, Pozzi M, Rota M, Vargiolu A, Citerio G (2015) High-resolution intracranial pressure burden and outcome in subarachnoid haemorrhage. Stroke 46:2464–2469. https://doi.org/10.1161/STROKEAHA.115.010219
Claassen J, Park S (2022) Spontaneous subarachnoid haemorrhage. Lancet 400:846–862. https://doi.org/10.1016/S0140-6736(22)00938-2
Fang Y, Huang L, Wang X, Si X, Lenahan C, Shi H, Shao A, Tang J, Chen S, Zhang J, Zhang JH (2022) A new perspective on cerebrospinal fluid dynamics after subarachnoid haemorrhage: from normal physiology to pathophysiological changes. J Cereb Blood Flow Metab 42:543–558. https://doi.org/10.1177/0271678X211045748
Robba C, Graziano F, Rebora P, Elli F, Giussani C, Oddo M, Meyfroidt G, Helbok R, Taccone FS, Prisco L, Vincent J-L, Suarez JI, Stocchetti N, Citerio G, Abdelaty M, Abed Maillard S, Ahmed H, Albrecht L, Alsudani A, Amundarain ED, Anand S, Andersen JB, Anglada M, Arabi Y, Aragao I, Arias Verdu MD, Asehnoune K, Assunção F, Audibert G, Badenes R, Bajracharya T, Banco P, Batista D, Bertellini E, Berty Gutiérrez H, Besch G, Biston P, Blandino Ortiz A, Blazquez V, Bloria S, Bonetti C, Bresil P, Brunetti I, Buldini V, Caillard A, Calamai I, Carbonara M, Caricato A, Casadio MC, Casanova M, Cavaleiro P, Celaya Lopez M, Chan CY, Chauhan R, Cinotti R, Corral L, Cortegiani A, Cotoia A, Crippa IA, Davidovich V, Del Bianco S, Diakaki C, Dibu J, Dimoula A, Domeniconi G, Dominguez LJY, Dovbysh N, Duque P, Eddelien HS, Efthymiou A, Egmose Larsen T, Elhadi M, Favre Eva E, Fencl M, Forjan P, Freitas R, Fuest K, Fumale M, Gakuba C, Galarza L, García MF, Gasca López GA, Gelormini C, Gempeler A, Giannopoulos A, Giménez ME, Giugni A, Glorieux D, Gonzalez Perez MI, Gradisek P, Grandis M, Griesdale D, Gritsan A, Grotheer S, Gupta D, Hallt ED, Hawthorne C, Helbok R, Holm MO, Iasonidou C, Idowu O, Ioannoni E, Izzi A, Jibaja M, Kafle P, Kandamby DH, Khan MM, Khomiakov S, Kilapong B, Kletecka J, Kojder K, Kolias A, Kontoudaki E, Koukoulitsios G, Kovac N, Kozar S, Krieg SM, Kurtz P, Kyriazopoulos G, Lamperti M, Lavicka P, Lencastre L, Levin M, Lightfoot R, Lindner A, López Ojeda P, Lores A, Lucca M, Luthra A, Magni F, Majholm B, Makris D, Maldonado F, Marudi A, Maskey S, Mebis L, Mejia-Mantilla JH, Mendoza R, Milivojevic N, Miroz JP, Monleon B, Montes JM, Morelli P, Motta A, Mouloudi E, Muehlschlegel S, Ñamendys Silva SA, Nardai G, Nilam K, Olson D, Ozair A, Pacheco C, Padilla Juan J, Palli E, Panda N, Pantelas N, Pariente L, Pearson D, Pérez-Araos R, Picetti E, Pinedo Portilla JL, Pons B, Pozzi F, Provaznikova E, Quartarone MC, Quintard H, Rajbanshi L, Reade M, Ribaric SF, Rigamonti A, Rivera LL, Roberts J, Roka YB, Sabelnikovs O, Sapra H, Schaller SJ, Sekhon M, Sellami W, Seppelt I, Serrano A, Sharma K, Shrestha GS, Shum HP, Silva S, Simoes M, Sivakumar S, Siviter R, Skola J, Škoti M, Smitt M, Soley R, Sonneville R, Soragni A, Soyer B, Spatenkova V, Stamou EE, Stival E, Olson Z, Tánczos K, Thompson C, Thomsen J, Tsikriki S, Van De Velde S, Videtta W, Villa F, Vrbica K, Vrettou C, Westy Hoffmeyer H, Wolf S, Wolf S, Yasin Wayhs S, Zerbi SM (2021) Intracranial pressure monitoring in patients with acute brain injury in the intensive care unit (SYNAPSE-ICU): an international, prospective observational cohort study. Lancet Neurol 20:548–558. https://doi.org/10.1016/S1474-4422(21)00138-1
Baggiani M, Graziano F, Rebora P, Robba C, Guglielmi A, Galimberti S, Giussani C, Suarez JI, Helbok R, Citerio G (2023) Intracranial pressure monitoring practice, treatment, and effect on outcome in aneurysmal subarachnoid haemorrhage. Neurocrit Care 38:741–751. https://doi.org/10.1007/s12028-022-01651-8
Akinduro OO, Vivas-Buitrago TG, Haranhalli N, Ganaha S, Mbabuike N, Turnbull MT, Tawk RG, Freeman WD (2020) Predictors of ventriculoperitoneal shunting following subarachnoid haemorrhage treated with external ventricular drainage. Neurocrit Care 32:755–764. https://doi.org/10.1007/s12028-019-00802-8
Veldeman M, Albanna W, Weiss M, Park S, Hoellig A, Clusmann H, Helbok R, Temel Y, Alexander Schubert G (2021) Invasive multimodal neuromonitoring in aneurysmal subarachnoid haemorrhage: a systematic review. Stroke 52:3624–3632. https://doi.org/10.1161/STROKEAHA.121.034633
Hammer A, Ranaie G, Erbguth F, Hohenhaus M, Wenzl M, Killer-Oberpfalzer M, Steiner H-H, Janssen H (2020) Impact of complications and comorbidities on the intensive care length of stay after aneurysmal subarachnoid haemorrhage. Sci Rep 10:6228. https://doi.org/10.1038/s41598-020-63298-9
Van Der Bilt IAC, Hasan D, Vandertop WP, Wilde AAM, Algra A, Visser FC, Rinkel GJE (2009) Impact of cardiac complications on outcome after aneurysmal subarachnoid haemorrhage: a meta-analysis. Neurology 72:635–642. https://doi.org/10.1212/01.wnl.0000342471.07290.07
Sommargren CE (2002) Electrocardiographic abnormalities in patients with subarachnoid haemorrhage. Am J Crit Care Off Publ Am Assoc Crit-Care Nurses 11:48–56
Neifert SN, Chapman EK, Martini ML, Shuman WH, Schupper AJ, Oermann EK, Mocco J, Macdonald RL (2021) Aneurysmal subarachnoid haemorrhage: the last decade. Transl Stroke Res 12:428–446. https://doi.org/10.1007/s12975-020-00867-0
Simonassi F, Ball L, Badenes R, Millone M, Citerio G, Zona G, Pelosi P, Robba C (2021) Hemodynamic monitoring in patients with subarachnoid haemorrhage: a systematic review and meta-analysis. J Neurosurg Anesthesiol 33:285–292. https://doi.org/10.1097/ANA.0000000000000679
Pollick C, Cujec B, Parker S, Tator C (1988) Left ventricular wall motion abnormalities in subarachnoid haemorrhage: an echocardiographic study. J Am Coll Cardiol 12:600–605. https://doi.org/10.1016/S0735-1097(88)80044-5
Kerro A, Woods T, Chang JJ (2017) Neurogenic stunned myocardium in subarachnoid haemorrhage. J Crit Care 38:27–34. https://doi.org/10.1016/j.jcrc.2016.10.010
Medina De Chazal H, Del Buono MG, Keyser-Marcus L, Ma L, Moeller FG, Berrocal D, Abbate A (2018) Stress cardiomyopathy diagnosis and treatment. J Am Coll Cardiol 72:1955–1971. https://doi.org/10.1016/j.jacc.2018.07.072
Cerecedo-Lopez CD, Ng I, Nguyen HB, Lai PMR, Gormley WB, Patel N, Frerichs KU, Aziz-Sultan MA, Du R (2022) Incidence and outcomes of registry-based acute myocardial infarction after aneurysmal subarachnoid haemorrhage. Neurocrit Care 36:772–780. https://doi.org/10.1007/s12028-021-01365-3
Armstrong JR, Mosher BD (2011) Aspiration pneumonia after stroke: intervention and prevention. Neurohospitalist 1:85–93. https://doi.org/10.1177/1941875210395775
Solenski NJ, Haley ECJ, Kassell NF, Kongable G, Germanson T, Truskowski L, Torner JC (1995) Medical complications of aneurysmal subarachnoid haemorrhage: a report of the multicenter, cooperative aneurysm study. Crit Care Med 23:1007–1017. https://doi.org/10.1097/00003246-199506000-00004
Baumann A, Audibert G, McDonnell J, Mertes PM (2007) Neurogenic pulmonary oedema. Acta Anaesthesiol Scand 51:447–455. https://doi.org/10.1111/j.1399-6576.2007.01276.x
Fan E, Brodie D, Slutsky AS (2018) Acute respiratory distress syndrome: advances in diagnosis and treatment. JAMA 319:698. https://doi.org/10.1001/jama.2017.21907
Feldstein E, Ali S, Patel S, Raghavendran K, Martinez E, Blowes L, Ogulnick J, Bravo M, Dominguez J, Li B, Urhie O, Rosenberg J, Bowers C, Prabhakaran K, Bauershmidt A, Mayer SA, Gandhi CD, Al-Mufti F (2023) Acute respiratory distress syndrome in patients with subarachnoid haemorrhage: incidence, predictive factors, and impact on mortality. Interv Neuroradiol 29:189–195. https://doi.org/10.1177/15910199221082457
Veeravagu A, Chen Y-R, Ludwig C, Rincon F, Maltenfort M, Jallo J, Choudhri O, Steinberg GK, Ratliff JK (2014) Acute lung injury in patients with subarachnoid haemorrhage: a nationwide inpatient sample study. World Neurosurg 82:e235–e241. https://doi.org/10.1016/j.wneu.2014.02.030
Mazeraud A, Robba C, Rebora P, Iaquaniello C, Vargiolu A, Rass V, Bogossian EG, Helbok R, Taccone FS, Citerio G (2021) Acute distress respiratory syndrome after subarachnoid haemorrhage: incidence and impact on the outcome in a large multicenter, retrospective cohort. Neurocrit Care 34:1000–1008. https://doi.org/10.1007/s12028-020-01115-x
Brower RG, Matthay MA, Morris A et al (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342:1301–1308. https://doi.org/10.1056/NEJM200005043421801
Duggal A, Rezoagli E, Pham T, McNicholas BA, Fan E, Bellani G, Rubenfeld G, Pesenti AM, Laffey JG (2020) Patterns of use of adjunctive therapies in patients with early moderate to severe ARDS. Chest 157:1497–1505. https://doi.org/10.1016/j.chest.2020.01.041
Martini RP, Deem S, Brown M, Souter MJ, Yanez ND, Daniel S, Treggiari MM (2012) The association between fluid balance and outcomes after subarachnoid haemorrhage. Neurocrit Care 17:191–198. https://doi.org/10.1007/s12028-011-9573-0
Witsch J, Frey H-P, Schmidt JM, Velazquez A, Falo CM, Reznik M, Roh D, Agarwal S, Park S, Connolly ES, Claassen J (2017) Electroencephalographic periodic discharges and frequency-dependent brain tissue hypoxia in acute brain injury. JAMA Neurol 74:301. https://doi.org/10.1001/jamaneurol.2016.5325
Quinn L, Tian DH, Fitzgerald E, Flower O, Andersen C, Hammond N, Davidson K, Delaney A (2020) The association between hyponatraemia and long-term functional outcome in patients with aneurysmal subarachnoid haemorrhage: a single centre prospective cohort study. J Clin Neurosci 78:353–359. https://doi.org/10.1016/j.jocn.2020.06.003
Hannon MJ, Behan LA, O’Brien MMC, Tormey W, Ball SG, Javadpur M, Sherlock M, Thompson CJ (2014) Hyponatremia following mild/moderate subarachnoid haemorrhage is due to SIAD and glucocorticoid deficiency and not cerebral salt wasting. J Clin Endocrinol Metab 99:291–298. https://doi.org/10.1210/jc.2013-3032
Mori T, Katayama Y, Kawamata T, Hirayama T (1999) Improved efficiency of hypervolemic therapy with inhibition of natriuresis by fludrocortisone in patients with aneurysmal subarachnoid haemorrhage. J Neurosurg 91:947–952. https://doi.org/10.3171/jns.1999.91.6.0947
Kruyt ND, Biessels GJ, De Haan RJ, Vermeulen M, Rinkel GJE, Coert B, Roos YBWEM (2009) Hyperglycemia and clinical outcome in aneurysmal subarachnoid haemorrhage: a meta-analysis. Stroke. https://doi.org/10.1161/STROKEAHA.108.529974
Bilotta F, Spinelli A, Giovannini F, Doronzio A, Delfini R, Rosa G (2007) The effect of intensive insulin therapy on infection rate, vasospasm, neurologic outcome, and mortality in neurointensive care unit after intracranial aneurysm clipping in patients with acute subarachnoid haemorrhage: a randomized prospective pilot trial. J Neurosurg Anesthesiol 19:156–160. https://doi.org/10.1097/ANA.0b013e3180338e69
Lavinio A, Andrzejowski J, Antonopoulou I, Coles J, Geoghegan P, Gibson K, Gudibande S, Lopez-Soto C, Mullhi R, Nair P, Pauliah VP, Quinn A, Rasulo F, Ratcliffe A, Reddy U, Rhodes J, Robba C, Wiles M, Williams A (2023) Targeted temperature management in patients with intracerebral haemorrhage, subarachnoid haemorrhage, or acute ischaemic stroke: updated consensus guideline recommendations by the Neuroprotective Therapy Consensus Review (NTCR) group. Br J Anaesth 131:294–301. https://doi.org/10.1016/j.bja.2023.04.030
De Oliveira Manoel AL, Mansur A, Silva GS, Germans MR, Jaja BNR, Kouzmina E, Marotta TR, Abrahamson S, Schweizer TA, Spears J, Macdonald RL (2016) Functional outcome after poor-grade subarachnoid haemorrhage: a single-center study and systematic literature review. Neurocrit Care 25:338–350. https://doi.org/10.1007/s12028-016-0305-3
Konczalla J, Seifert V, Beck J, Güresir E, Vatter H, Raabe A, Marquardt G (2018) Outcome after Hunt and Hess Grade V subarachnoid haemorrhage: a comparison of pre-coiling era (1980–1995) versus post-ISAT era (2005–2014). J Neurosurg 128:100–110. https://doi.org/10.3171/2016.8.JNS161075
Wartenberg KE (2011) Critical care of poor-grade subarachnoid haemorrhage. Curr Opin Crit Care 17:85–93. https://doi.org/10.1097/MCC.0b013e328342f83d
Picetti E, Bouzat P, Bader MK, Citerio G, Helbok R, Horn J, Macdonald RL, McCredie V, Meyfroidt G, Righy C, Robba C, Sharma D, Smith WS, Suarez JI, Udy A, Wolf S, Taccone FS (2023) A survey on monitoring and management of cerebral vasospasm and delayed cerebral ischemia after subarachnoid haemorrhage: the mantra study. J Neurosurg Anesthesiol. https://doi.org/10.1097/ANA.0000000000000923
Shah VA, Kazmi SO, Damani R, Harris AH, Hohmann SF, Calvillo E, Suarez JI (2022) Regional variability in the care and outcomes of subarachnoid haemorrhage patients in the United States. Front Neurol 13:908609. https://doi.org/10.3389/fneur.2022.908609
Niznick N, Saigle V, Marti ML, Laframboise J, Zha X, Andersen C, Presseau J, Chassé M, McIntyre L, Fergusson D, Turgeon AF, Lauzier F, Mayer SA, Lahiri S, Ramsay T, English S, for the APOC Investigators and the Canadian Critical Care Trials Group (2023) Patient relevance of the modified Rankin scale in subarachnoid haemorrhage research: an international cross-sectional survey. Neurology 100:e1565–e1573. https://doi.org/10.1212/WNL.0000000000206879
Andersen CR, English SW, Delaney A (2022) Made to measure—selecting outcomes in aneurysmal subarachnoid haemorrhage research. Front Neurol 13:1000454. https://doi.org/10.3389/fneur.2022.1000454
Sonesson B, Kronvall E, Säveland H, Brandt L, Nilsson OG (2018) Long-term reintegration and quality of life in patients with subarachnoid haemorrhage and a good neurological outcome: findings after more than 20 years. J Neurosurg 128:785–792. https://doi.org/10.3171/2016.11.JNS16805
Wartenberg KE, Hwang DY, Haeusler KG, Muehlschlegel S, Sakowitz OW, Madžar D, Hamer HM, Rabinstein AA, Greer DM, Hemphill JC, Meixensberger J, Varelas PN (2019) Gap analysis regarding prognostication in neurocritical care: a joint statement from the German Neurocritical Care Society and the Neurocritical Care Society. Neurocrit Care 31:231–244. https://doi.org/10.1007/s12028-019-00769-6
Pace A, Mitchell S, Casselden E, Zolnourian A, Glazier J, Foulkes L, Bulters D, Galea I (2018) A subarachnoid haemorrhage-specific outcome tool. Brain 141:1111–1121. https://doi.org/10.1093/brain/awy003
Treggiari MM, Rabinstein AA, Busl KM, Caylor MM, Citerio G, Deem S, Diringer M, Fox E, Livesay S, Sheth KN, Suarez JI, Tjoumakaris S (2023) Guidelines for the neurocritical care management of aneurysmal subarachnoid hemorrhage. Neurocrit Care 39:1–28
Steiner T, Juvela S, Unterberg A, Jung C, Forsting M, Rinkel G, European Stroke O (2013) European Stroke Organization guidelines for the management of intracranial aneurysms and subarachnoid haemorrhage. Cerebrovasc Dis 35:93–112
Dayyani M, Mousavi Mohammadi E, Ashoorion V, Sadeghirad B, Javedani Yekta M, Grotta JC, Gonzalez NR, Zabihyan S (2022) Aneurysmal subarachnoid haemorrhage-cerebral vasospasm and prophylactic ibuprofen: a randomised controlled pilot trial protocol. BMJ Open 12:e058895
Zolnourian AH, Franklin S, Galea I, Bulters DO (2020) Study protocol for SFX-01 after subarachnoid haemorrhage (SAS): a multicentre randomised double-blinded, placebo controlled trial. BMJ Open 10:e028514
Bruder N, Higashida R, Santin-Janin H, Dubois C, Aldrich EF, Marr A, Roux S, Mayer SA (2022) The REACT study: design of a randomized phase 3 trial to assess the efficacy and safety of clazosentan for preventing deterioration due to delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. BMC Neurol 22:492
Idorsia (2023) Idorsia announces the results of REACT a Phase 3 study of clazosentan in patients following aneurysmal subarachnoid hemorrhage. In: Book Idorsia announces the results of REACT a Phase 3 study of clazosentan in patients following aneurysmal subarachnoid hemorrhage
Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, Vajkoczy P, Wanke I, Bach D, Frey A, Marr A, Roux S, Kassell N (2011) Clazosentan, an endothelin receptor antagonist, in patients with aneurysmal subarachnoid haemorrhage undergoing surgical clipping: a randomised, double-blind, placebo-controlled phase 3 trial (CONSCIOUS-2). Lancet Neurol 10:618–625
Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, Vajkoczy P, Wanke I, Bach D, Frey A, Nowbakht P, Roux S, Kassell N (2012) Randomized trial of clazosentan in patients with aneurysmal subarachnoid hemorrhage undergoing endovascular coiling. Stroke 43:1463–1469
Koopman I, Tack RW, Wunderink HF, Bruns AH, van der Schaaf IC, Cianci D, Gelderman KA, van de Ridder IM, Hol EM, Rinkel GJ, Vergouwen MD (2023) Safety and pharmacodynamic efficacy of eculizumab in aneurysmal subarachnoid hemorrhage (CLASH): a phase 2a randomized clinical trial. Eur Stroke J 8:23969873231194124
Castle-Kirszbaum M, Lai L, Maingard J, Asadi H, Danks RA, Goldschlager T, Chandra RV (2021) Intravenous milrinone for treatment of delayed cerebral ischaemia following subarachnoid haemorrhage: a pooled systematic review. Neurosurg Rev 44:3107–3124
Lannon M, Martyniuk A, Sharma S, (2022) Intravenous milrinone for delayed cerebral ischaemia in aneurysmal subarachnoid haemorrhage: a systematic review. Br J Neurosurg 26:1–6. https://doi.org/10.1080/02688697.2022.2125160
English SW, Fergusson D, Chassé M, Turgeon AF, Lauzier F, Griesdale D, Algird A, Kramer A, Tinmouth A, Lum C, Sinclair J, Marshall S, Dowlatshahi D, Boutin A, Pagliarello G, McIntyre LA (2016) Aneurysmal SubArachnoid Hemorrhage-Red Blood Cell Transfusion And Outcome (SAHaRA): a pilot randomised controlled trial protocol. BMJ Open 6:e012623
Taccone FS, Badenes R, Rynkowski CB, Bouzat P, Caricato A, Kurtz P, Moller K, Diaz MQ, Van Der Jagt M, Videtta W, Vincent J-L (2023) TRansfusion strategies in Acute brain INjured patients (TRAIN): a prospective multicenter randomized interventional trial protocol. Trials 24:20
Blackburn SL, Grande AW, Swisher CB, Hauck EF, Jagadeesan B, Provencio JJ (2019) Prospective trial of cerebrospinal fluid filtration after aneurysmal subarachnoid hemorrhage via lumbar catheter (PILLAR). Stroke 50:2558–2561
Roelz R, Schubach F, Coenen VA, Jenkner C, Scheiwe C, Grauvogel J, Niesen WD, Urbach H, Taschner C, Seufert J, Kätzler J, Beck J, Reinacher PC (2021) Stereotactic cisternal lavage in patients with aneurysmal subarachnoid hemorrhage with urokinase and nimodipine for the prevention of secondary brain injury (SPLASH): study protocol for a randomized controlled trial. Trials 22:285
Liu J, He J, Chen X, Feng Y, Wang C, Awil MA, Wang Y, Tian Y, Hou D (2022) Cilostazol for aneurysmal subarachnoid hemorrhage: an updated systematic review and meta-analysis. Cerebrovasc Dis 51:138–148
Shibuya M, Suzuki Y, Enomoto H, Okada T, Ogura K, Sugita K (1994) Effects of prophylactic intrathecal administrations of nicardipine on vasospasm in patients with severe aneurysmal subarachnoid haemorrhage. Acta Neurochir (Wien) 131:19–25
Sadan O, Waddel H, Moore R, Feng C, Mei Y, Pearce D, Kraft J, Pimentel C, Mathew S, Akbik F, Ameli P, Taylor A, Danyluk L, Martin KS, Garner K, Kolenda J, Pujari A, Asbury W, Jaja BNR, Macdonald RL, Cawley CM, Barrow DL, Samuels O (2022) Does intrathecal nicardipine for cerebral vasospasm following subarachnoid hemorrhage correlate with reduced delayed cerebral ischemia? A retrospective propensity score-based analysis. J Neurosurg 136:115–124
Barth M, Capelle HH, Weidauer S, Weiss C, Münch E, Thomé C, Luecke T, Schmiedek P, Kasuya H, Vajkoczy P (2007) Effect of nicardipine prolonged-release implants on cerebral vasospasm and clinical outcome after severe aneurysmal subarachnoid hemorrhage: a prospective, randomized, double-blind phase IIa study. Stroke 38:330–336
Pileggi M, Mosimann PJ, Isalberti M, Piechowiak EI, Merlani P, Reinert M, Cianfoni A (2021) Stellate ganglion block combined with intra-arterial treatment: a “one-stop shop” for cerebral vasospasm after aneurysmal subarachnoid hemorrhage-a pilot study. Neuroradiology 63:1701–1708
Zhang J, Nie Y, Pang Q, Zhang X, Wang Q, Tang J (2021) Effects of stellate ganglion block on early brain injury in patients with subarachnoid hemorrhage: a randomised control trial. BMC Anesthesiol 21:23
Zanaty M, Allan L, Samaniego EA, Piscopo A, Ryan E, Torner JC, Hasan D (2021) Phase 1/2a trial of ISPASM. Stroke 52:3750–3758
Dawley T, Claus CF, Tong D, Rajamand S, Sigler D, Bahoura M, Garmo L, Soo TM, Kelkar P, Richards B (2020) Efficacy and safety of cilostazol-nimodipine combined therapy on delayed cerebral ischaemia after aneurysmal subarachnoid haemorrhage: a prospective, randomised, double-blinded, placebo-controlled trial protocol. BMJ Open 10:e036217
Jing L, Wu Y, Liang F, Jian M, Bai Y, Wang Y, Liu H, Wang A, Chen X, Han R (2022) Effect of early stellate ganglion block in cerebral vasospasm after aneurysmal subarachnoid hemorrhage (BLOCK-CVS): study protocol for a randomized controlled trial. Trials 23:922
Macdonald RL, Hänggi D, Ko NU, Darsaut TE, Carlson AP, Wong GK, Etminan N, Mayer SA, Aldrich EF, Diringer MN, Ng D, Strange P, Bleck T, Grubb R, Suarez JI (2020) NEWTON-2 cisternal (nimodipine microparticles to enhance recovery while reducing toxicity after subarachnoid hemorrhage): a phase 2, multicenter, randomized, open-label safety study of intracisternal EG-1962 in aneurysmal subarachnoid hemorrhage. Neurosurgery 88:E13-e26
Funding
Open access funding provided by Università degli Studi di Genova within the CRUI-CARE Agreement. MDIV is supported by a Clinical Established Investigator grant from the Dutch Heart Foundation (2018T076). KMB is supported by the National Institute Of Neurological Disorders And Stroke of the National Institutes of Health (U01NS124613). JC is supported by Neurological Disorders And Stroke of the National Institutes of Health (R01NS106014, R03NS112760, R21NS128326) and the James S. McDonnell Foundation. SP is supported by the National Institutes of Health (R01NS131606, R01NS129760). GC participated in the manuscript preparation during his personal involvement in the Italian Minister of University MUR Dipartimenti di Eccellenza 2023–2027 (l. 232/2016, art. 1, commi 314–337).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
CR received fees as a speaker from Masimo and Edwards Lifesciences, Nihon Kohden and BD, all outside the submitted work. GC reports grants and personal fees as a speakers' bureau member and advisory board member from Integra Neurosciences, NeurOptics, Biogen, Invex Ltd and Idorsia, all outside the submitted work. KMB has received honoraria from the American Academy of Neurology for speaking, editing and course directing and is an Associate Editor for Critical Care Medicine. JC is a minority shareholder at iCE Neurosystems.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Robba, C., Busl, K.M., Claassen, J. et al. Contemporary management of aneurysmal subarachnoid haemorrhage. An update for the intensivist. Intensive Care Med 50, 646–664 (2024). https://doi.org/10.1007/s00134-024-07387-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-024-07387-7