Abstract
Purpose
Acute kidney disease (AKD) is a significant health care burden worldwide. However, little is known about this complication after major surgery.
Methods
We conducted an international prospective, observational, multi-center study among patients undergoing major surgery. The primary study endpoint was the incidence of AKD (defined as new onset of estimated glomerular filtration rate (eCFR) < 60 ml/min/1.73 m2 present on day 7 or later) among survivors. Secondary endpoints included the relationship between early postoperative acute kidney injury (AKI) (within 72 h after major surgery) and subsequent AKD, the identification of risk factors for AKD, and the rate of chronic kidney disease (CKD) progression in patients with pre-existing CKD.
Results
We studied 9510 patients without pre-existing CKD. Of these, 940 (9.9%) developed AKD after 7 days of whom 34.1% experiencing an episode of early postoperative-AKI. Rates of AKD after 7 days significantly increased with the severity (19.1% Kidney Disease Improving Global Outcomes [KDIGO] 1, 24.5% KDIGO2, 34.3% KDIGO3; P < 0.001) and duration (15.5% transient vs 38.3% persistent AKI; P < 0.001) of early postoperative-AKI. Independent risk factors for AKD included early postoperative-AKI, exposure to perioperative nephrotoxic agents, and postoperative pneumonia. Early postoperative-AKI carried an independent odds ratio for AKD of 2.64 (95% confidence interval [CI] 2.21–3.15). Of 663 patients with pre-existing CKD, 42 (6.3%) had worsening CKD at day 90. In patients with CKD and an episode of early AKI, CKD progression occurred in 11.6%.
Conclusion
One in ten major surgery patients developed AKD beyond 7 days after surgery, in most cases without an episode of early postoperative-AKI. However, early postoperative-AKI severity and duration were associated with an increased rate of AKD and early postoperative-AKI was strongly associated with AKD independent of all other potential risk factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
One in ten patients develop acute kidney disease (AKD) beyond 7 days after major surgery, most of them without a prior episode of acute kidney injury (AKI). However, early postoperative AKI is a major risk factor for subsequent AKD. These findings have important implications for the management and prognosis of surgical patients. |
Introduction
Each year, more than 310 million patients undergo major surgery worldwide and postoperative complications are associated with increased morbidity and mortality [1, 2]. Acute kidney injury (AKI) is an important and common complication after major surgery and is independently associated with morbidity and mortality in a wide range of surgical settings [3,4,5,6]. Moreover, one in five patients will develop an episode of postoperative-AKI occurring within 72 h after major surgery (early postoperative-AKI) [7]. Furthermore, the development of AKI has been associated with an increased risk for subsequent chronic kidney disease (CKD) [8, 9]. Such CKD is logically likely to be preceded by postoperative acute kidney disease (AKD). Surprisingly, the epidemiology of AKD after major surgery has not been investigated in depth and the contribution of early postoperative-AKI as well as AKI characteristics, duration, and development of AKD have not been studied in detail. Finally, additional factors occurring in the perioperative period which may be associated with AKD development remain unknown.
We aimed to test the primary hypothesis that AKD after 7 days is common after major surgery and determine whether early postoperative-AKI and its characteristics (duration, severity, and specific diagnostic criteria), patient characteristics and perioperative factors influence the development of AKD after 7 days. Additionally, we assessed the incidence of CKD progression in terms of KDIGO staging in those with pre-existing CKD and its relationship with early postoperative-AKI.
Methods
Study design and ethics
This is an a priori planned secondary analysis of the Epidemiology of Surgery-Associated Acute Kidney Injury (EPIS-AKI) study, an international prospective, observational, multicenter, cohort study, which has been described in detail elsewhere [10]. Briefly, 10,568 patients from 30 countries and 148 centers were enrolled from June 2020 to December 2021. All patients (age ≥ 18 years) undergoing major elective and emergency surgery (operative time ≥ 2 h) with subsequent intensive care unit (ICU) or high dependency unit admission were included regardless of surgical subspecialty. Exclusion criteria included pre-existing AKI, AKI within the last 3 months, end-stage renal disease with dialysis dependency, and kidney transplant.
The EPIS-AKI study was approved by the Research Ethics Committee of the Chamber of Physicians Westfalen-Lippe and the Westphalian Wilhelms-University Münster (2019-424-f-S). Country-specific requirements, including local ethics approval and/or study registration were fulfilled according to the local requirements and prior to patient enrollment. The study was registered at clinicaltrials.gov (NCT04165369, November 18th 2019). The manuscript follows the principles of “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) and the Declaration of Helsinki (Fortaleza 2013).
Outcomes
The primary endpoint of this study was the development of AKD present on 7 days or later after surgery. AKD was defined as surviving to 90 days with an observed decrease in estimated glomerular filtration rate (eGFR) to < 60 ml/min/1.73 m2, in patients with a eGFR > 60 ml/min/1.73 m2 prior to surgery.
Secondary endpoints were the association of early postoperative-AKI and AKI characteristics (duration, severity (as defined by Kidney Disease Improving Global Outcomes [KDIGO] stages), and specific diagnostic criteria (serum creatinine and/or urine output)) with the development of AKD after 7 days. We concentrated on early postoperative-AKI, because the majority of AKI occurs within 72 h after surgery and because this time frame reflects changes directly related to surgery and perioperative interventions [11,12,13]. In a subset of patients with preoperative CKD (defined by eGFR), we determined the incidence of CKD progression (progression of at least one stage of CKD) and the relationship of such progression to postoperative-AKI.
Statistical analysis
Frequencies, percentages, medians, quartiles were calculated for the baseline variables, primary and secondary endpoints as applicable and P-values for primary and secondary endpoints.
Fisher’s exact test and Pearson’s Chi-squared test were used to compare categorical variables between groups. Continuous variables were compared using Welch’s t-test or Mann–Whitney U test depending on whether the target variable was normally distributed in both groups or not.
Confidence intervals (CIs) for binomial proportion estimates, e.g., the development of AKD after 7 days, were calculated using the Clopper-Pearson exact method with a 95% confidence level. For multinomial proportion estimates, e.g. KDIGO stages (1/2/3) in postoperative-AKI patients, simultaneous 95% CIs were calculated using Goodman’s methods [14].
To examine the association of postoperative-AKI and AKD development beyond 7 days in detail, two univariate logistic regression models were fitted, each including a combined variable. The categories of the first variable were composed of all possible combinations of postoperative-AKI (yes/no), KDIGO stage (1/2/3), and AKI duration, defined as transient (< 48 h duration) or persistent (> 48 h duration) [15]. The categories of the second variable were composed of all possible combinations of postoperative-AKI (yes/no), KDIGO stage (1/2/3), and the criteria used to define postoperative-AKI (serum creatinine and/or urine output).
To identify and assess the association of potential risk factors for the development of AKD after 7 days, multivariable logistic regression analyses were performed to develop a prediction model. Firstly, we selected variables that have been proposed as being associated with the development of AKD after 7 days including gender, age, body mass index (BMI), UN-geoscheme, health expenditure, hypertension, atrial fibrillation, previous myocardial infarction, congestive cardiac failure, diabetes, chronic obstructive pulmonary disease (COPD), peripheral vascular disease, stroke, American Society of Anesthesiologists (ASA) score, urgency of procedure, surgery duration, type of surgery, as well as intra- and postoperative transfusion, fluid balance, blood loss, complications, intra- and postoperative use of nephrotoxic agents, use of vasopressors, and early postoperative development of AKI. We included all these variables in a logistic regression model and then performed a fast-backward variable selection based on Akaike’s Information Criterion (AIC) to identify a reasonable set of potential risk factors for development of AKD after 7 days. In each iteration, the influencing variable whose exclusion caused the greatest reduction of the AIC compared to the current model was excluded from the current model until no omission of a single variable resulted in a further reduction of the AIC. The variable selection and parameter estimation procedures were validated jointly using 500 bootstrap samples using the methods described by Harrell [16]. Further statiscal analyses are presented in supplementary eMethods.
All p-values and confidence limits were two-sided. Only the confidence interval of the primary endpoint, the incidence of AKD after 7 days, is to be interpreted confirmatory. All other analyses are to be interpreted in an exploratory sense and were not adjusted for multiple testing. P-values are therefore considered statistically noticeable (“significant”) in case P ≤ 0.05. An overall significance level across all statistical analyses was not determined and cannot be calculated. In all analyses, only the complete cases were considered, i.e., missing values were not imputed. Statistical analyses were conducted using R (Version R-4.1.2).
Results
Patients
The primary analysis included 9510/10,568 (90%) evaluable patients without any pre-existing CKD who survived at least 90 days (Fig. 1). Among the survivors, pre-operative CKD was present in 663 (6.5%) patients. Demographic and baseline characteristics according to the development of postoperative AKD beyond 7 days are presented in Table 1 and supplementary eFigure 1. Surgical and postoperative details are presented in supplementary eTable 1.
The mean age in the primary analysis cohort was 59 (standard deviation [SD] 15) years, 61% were men, median serum creatinine was 0.8 (Q1, Q3, 0.7–1) mg/dl. Overall, 9129 (96.3%) patients had elective procedures and the median duration of surgery was 230 (Q1, Q3, 172, 294) minutes.
Development of AKD after 7 days
In total, 940/9,510 (9.9% [95% CI 9.3–10.5%]) patients developed AKD beyond 7 days (supplementary eTable 2). Vascular and cardiac surgery had similar rates of AKD after 7 days at 13.8% and 12.8%, respectively. In contrast, in patients undergoing orthopaedic procedures the AKD rate after 7 days was 8.4% (supplementary eTable 1). The highest rate of AKD after 7 days was seen in patients undergoing urological procedures. Patients who developed AKD after 7 days had a higher baseline serum creatinine, were older, showed higher rates of comorbidities (hypertension, diabetes, congestive heart failure, previous myocardial infarction, peripheral vascular disease, and atrial flutter/fibrillation), received more nephrotoxic agents including contrast agents and nonsteroidal anti-inflammatory drugs (NSAIDs), and were more often treated with vasopressors (supplementary eTable 1).
AKI and AKD after 7 days
Overall, only 34.1% of patients who developed AKD after 7 days had a documented episode of early postoperative-AKI (Table 1 and supplementary eTable 3). However, when compared to patients who did not develop AKD after 7 days, patients with AKD after 7 days had higher rates of moderate and severe early postoperative-AKI (stage 2 and 3) (Table 1).
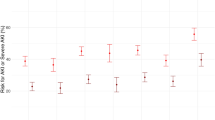
Rates of AKD after 7 days increased significantly with severity of early postoperative-AKI with 19.1% of patients reaching KDIGO stage 1 developing AKD after 7 days, increasing to 24.5% for KDIGO 2, and 34.3% for KDIGO stage 3 (P < 0.001). Similarly, duration of AKI was associated with increased rates of AKD after 7 days with 15.5% of patients who had transient AKI and 38.3% in patients with persistent AKI developing AKD after 7 days (P < 0.001) (Figs. 2 and 3) and showing increased odds ratio (ORs) for AKD after 7 days (supplementary eTable 4a). Regarding diagnostic criteria, early postoperative-AKI patients diagnosed by serum creatinine or both serum creatinine and urine output KDIGO criteria had the highest rate of AKD after 7 days (urine output: 10.7%, serum creatinine: 27.2%, both criteria: 26.8%; P < 0.001) and highest ORs for AKD after 7 days (supplementary eTable 4b).
Development of AKD beyond 7 days with 95% confidence intervals according to KDIGO stage and diagnostic criteria in postoperative-AKI patients without CKD prior to surgery. The data in gray present patients according to the criteria used to diagnose early PO-AKI with the darker gray indicating the proportion of such patients with AKD after 7 days and the numbers indicating the actual number of patients affected. In the first thinner color panels, patients are then subdivided according to whether they had stage 1 or 2 or 3 in the first 72 h (maximum stage). In the second larger color panels, the proportion of patients in each stage as measured in the first 72 h is also presented. For example, among all patients with early postoperative-AKI in KDIGO stage 1 diagnosed by serum creatinine alone, 12.5% developed AKD after 7 days
Development of AKD after 7 days with 95% confidence intervals according to KDIGO stage and duration of postoperative-AKI in postoperative-AKI patients without CKD prior to surgery. The data in gray present patients according to transient or persistent early PO-AKI with the darker gray indicating the proportion of such patients with AKD after 7 days and the numbers indicating the actual number of patients affected. In the first thinner color panels, patients are then subdivided according to whether they had stage 1 or 2 or 3 in the first 72 h. In the second larger color panels, the proportion of patients in each stage as measured in the first 72 h is also presented. For example, among all patients with a transient postoperative-AKI in KDIGO stage 1, 14.9% developed AKD after 7 days
Perioperative risk factors for AKD after 7 days
In multivariable regression analyses, early postoperative-AKI, female sex, age, comorbidities (hypertension, atrial fibrillation, myocardial infarction, peripheral vascular disease), emergency procedures, intraoperative nephrotoxic agents (vancomycin and cyclosporine or tacrolimus), postoperative nephrotoxic agents (e.g., aminoglycosides) and postoperative complications (pneumonia) were associated with subsequent AKD after 7 days (Table 2). Bootstrap validation of the model fitting process yielded a bias corrected area under the curve (AUC) of 0.768 and pseudo-R2 of 0.172 for the final model (supplementary eTable 5a and b). Of note, postoperative treatment with NSAIDs and vancomycin was associated with a lower development of AKD after 7 days. Early postoperative-AKI carried an independent OR of 2.64 (2.21, 3.15) for AKD after 7 days, the third highest OR after urological surgery and treatment with cyclosporine A or tacrolimus.
Some identified risk factors for AKD after 7 days differed between patients with and without early postoperative-AKI. However, age, gender, and the administration of nephrotoxins (cyclosporine) remained significant in both groups (supplementary eTables 6 and 7).
Combining AKD after 7 days and death at day 90 did not change the prediction model except for sepsis (supplementary eTables 8, 9 and 10).
Progression of CKD stage
Of 663 patients with pre-existing CKD, 42 (6.3% [95% CI 4.6–8.5%]) patients had progression of CKD; 29/251 patients with postoperative-AKI (11.6% [95% CI 7.9–16.2%]) and 13/412 patients without postoperative-AKI (3.2% [95% CI 1.7–5.3%]; P < 0.001) (supplementary eTable 11). Of these, most patients progressed by one stage of CKD (supplementary eTable 12). Progression of CKD was similar in patients with persistent and transient postoperative-AKI (14.5% vs. 9%, respectively; P = 0.168) (supplementary eFigure 2). Postoperative-AKI patients diagnosed by serum creatinine or both KDIGO criteria showed the highest rates of CKD progression (urine output 1.6%, serum creatinine 15%, both criteria 14.7%; P = 0.017) (supplementary eFigure 3).
Discussion
Among patients undergoing major cardiac and non-cardiac surgery, one in ten patients developed AKD after 7 days and only one-third of patients developing AKD after 7 days had a prior episode of early postoperative-AKI. However, when early postoperative-AKI was associated with AKD after 7 days, AKD rates increased significantly with both greater severity and duration of postoperative-AKI. Patients diagnosed with early post-operative AKI by both urine output and creatinine KDIGO criteria, had the highest AKD rates. Moreover, early postoperative-AKI was strongly associated with subsequent AKD after 7 days, independent of all other potential risk factors. Other risk factors for AKD after 7 days were female sex, age, comorbidities (hypertension, atrial fibrillation, myocardial infarction, peripheral vascular disease), emergency procedures, perioperative nephrotoxic agents (intraoperative vancomycin and cyclosporine/tacrolimus, postoperative aminoglycosides), and postoperative complications (pneumonia). Finally, among patients with preoperative CKD, progression of CKD was also more frequent in patients with early postoperative-AKI.
The findings of the EPIS-AKI study align with previous findings among hospitalized patients where CKD occurred in 11% of patients (CKD stages 3–5)[17] and confirm the relationship between postoperative-AKI and AKD [18,19,20,21,22]. However, a small recent study of general hospitalized CKD patients, rather than patients solely undergoing surgery, showed that the association between mild and moderate AKI and worsening subsequent kidney function was small [23]. Our observations are consistent with the notion that AKI is a key driver for the development of AKD and that AKI and AKD are two interconnected syndromes in the perioperative setting [8]. However, although early postoperative-AKI might be a significant risk factor for AKD after 7 days, our study also shows that patients undergoing major surgery are at risk for AKD after 7 days even if they did not have an episode of early postoperative-AKI.
Among early postoperative-AKI associated AKD patients, the duration and severity of early postoperative-AKI were two key risk factors. Aligned with our findings, one retrospective study among elderly patients showed that AKI duration of more than seven days increased the risk of CKD [24]. Another retrospective study found an OR of 23.7 for CKD in patients with an AKI duration of more than seven days [25]. However, the Acute Dialysis Quality Initiative (ADQI) proposed a standardized definition of persistent AKI based on the recovery of kidney function within 48 h, which is consistent with the definition used in the EPIS-AKI study [15]. Even using such a shorter 48 h cut off point, our findings align with previous studies.
Among all surgical subspecialties, urological procedures were associated with the highest risk of AKD after 7 days; however, this is partly confounded because it included urological procedures, which resulted in nephron loss (100 patients received a nephrectomy).
Nearly 8% of patients without early postoperative-AKI developed AKD after 7 days. It is conceivable that AKI might have occurred after 72 h postoperatively, as such data was not collected in the EPIS-AKI study. Subclinical (stage 1 s) AKI, which is defined by kidney damage without a functional loss (functional biomarkers serum creatinine and urine output are normal but damage biomarkers are elevated) [26], might have also occurred and affected the development of AKD as suggested by previous studies [27,28,29,30]. Nonetheless, the data suggests that this patient population has an increased susceptibility to developing kidney functional loss in the post-operative period and AKD after 7 days. Based on this increased risk, in these patients, it might be advisable to limit the use of nephrotoxic drugs whenever possible, use therapeutic drug monitoring where available, and try to optimize hemodynamic and volume status. Further trials have to address the question whether a specific drug treatment (e.g., early treatment with angiotensin converting enzyme [ACE] and/or sodium-glucose cotransporter-2 [SGLT2] inhibitors) can affect the development of AKD.
In other studies, age, female sex, and hypertension have been associated with AKD [17]. These risk factors were also found in the EPIS-AKI study, but additionally perioperative modifiable risk factors were detected. Nephrotoxic agents are known risk factors for AKI [31, 32] but, as shown here, probably also for AKD after 7 days even in patients without early AKI. As such, intraoperative use of immunosuppressive agents was associated with a higher risk of AKD after 7 days. One could question whether this is caused by the drug itself or whether this might be related to kidney transplantation. However, kidney transplantation was an exclusion criterion for the EPIS-AKI study. Immunosuppressive agents have different indications apart from kidney transplantation (e.g., other transplantations, rheumatoid arthritis, psoriasis). Consequently, it is likely the toxicity of the drug itself was related to the increased risk of AKD after 7 days. Thus, treatment with nephrotoxic medications must be carefully considered and, if possible, avoided. The implementation of a nephrotoxic drug stewardship could help prevent AKI and maybe also prevent AKD and CKD [33]. The fact that the postoperative application of NSAIDs and vancomycin was associated with lower rates of AKI may appear counterintuitive. However, it might be explained by selection bias or that, in such cases, clinicians were alerted to the risks associated with these drugs and therefore modify their treatment.
Strengths and limitations
The strengths of this study are the largest cohort of patients studied to date examining progression of AKD beyond 7 days postoperatively and the influence of early postoperative-AKI, the multinational setting, the multiple types of surgeries included, the detailed collection of data, the close monitoring for early post-operative AKI and the protocolized follow-up to determine the development of AKD after 7 days. As such, this is the first international multicentre study of the epidemiology of postoperative AKD and of its association with early postoperative-AKI and it provides novel insights.
We acknowledge several limitations. First, as AKI is part of the AKD definition and we wanted to distinguish between AKI and persistent renal dysfunction, we used the term “AKD beyond day 7”. The definition of AKD was based on a single assessment of kidney function beyond 7 days after surgery (mostly at day 90). In addition, we only used serum creatinine to estimate the GFR but did not consider other markers of CKD such as proteinuria. For these reasons, the term CKD could not be used [34]. It remains uncertain whether such prolonged reduced kidney function was reflective of a steady state and how it might relate to CKD. In addition, it remains unknown what factors that might have occurred between hospital discharge and day 90 may have impacted the incidence of AKD. Second, some surgical procedures were underrepresented, potentially resulting in a selection bias nor did we collect the granularity of all surgical procedures within speciality. Third, we only assessed patients for early postoperative-AKI and are unable to comment on whether AKI, or indeed AKD, developed thereafter at any time between day 3 and day 90. Furthermore, we did not measure biomarkers other than urine output and serum creatinine which may have been elevated in the 72 h post-surgery and which could have been associated with prolonged kidney dysfunction. Finally, the findings of this study can only be generalized to survivors to 90-day but may vary between the different countries, healthcare systems, and types and complexity of surgery. Some findings, such as the lower risk of AKD after 7 days with the use of intraoperative NSAIDs, are surprising and may be related to unknown confounding factors and selection bias.
Conclusion
In a large multicentre international study among patients undergoing major cardiac and non-cardiac surgery, one in ten patients developed AKD after 7 days. Although most patients developed AKD without a prior episode of early postoperative-AKI, early postoperative-AKI was a major risk factor for subsequent AKD after 7 days. Nephrotoxic drugs were also significant modifiable risk factors for AKD after 7 days. Moreover, among patients with preoperative CKD, progression of CKD was higher in patients with early postoperative-AKI. These findings have important implications not only for the management and prognosis of surgical patients and early postoperative-AKI but also for the design of future interventional clinical trials.
Data availability
The data used in the present study are available from the corresponding author upon reasonable request.
References
Weiser TG, Haynes AB, Molina G, Lipsitz SR, Esquivel MM, Uribe-Leitz T, Fu R, Azad T, Chao TE, Berry WR, Gawande AA (2015) Estimate of the global volume of surgery in 2012: an assessment supporting improved health outcomes. Lancet 385(Suppl 2):S11
Pearse RM, Moreno RP, Bauer P, Pelosi P, Metnitz P, Spies C, Vallet B, Vincent JL, Hoeft A, Rhodes A, European Surgical Outcomes Study group for the Trials groups of the European Society of Intensive Care M, the European Society of A (2012) Mortality after surgery in Europe: a 7 day cohort study. Lancet 380:1059–1065
Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW (2005) Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 16:3365–3370
Uchino S, Kellum JA, Bellomo R, Doig GS, Morimatsu H, Morgera S, Schetz M, Tan I, Bouman C, Macedo E, Gibney N, Tolwani A, Ronco C, Beginning, Ending Supportive Therapy for the Kidney I (2005) Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA 294:813–818
Barrantes F, Tian J, Vazquez R, Amoateng-Adjepong Y, Manthous CA (2008) Acute kidney injury criteria predict outcomes of critically ill patients. Crit Care Med 36:1397–1403
Longo WE, Virgo KS, Johnson FE, Oprian CA, Vernava AM, Wade TP, Phelan MA, Henderson WG, Daley J, Khuri SF (2000) Risk factors for morbidity and mortality after colectomy for colon cancer. Dis Colon Rectum 43:83–91
Zarbock A, Weiss R, Albert F, Rutledge K, Kellum JA, Bellomo R, Grigoryev E, Candela-Toha AM, Demir ZA, Legros V, Rosenberger P, Galan Menendez P, Garcia Alvarez M, Peng K, Leger M, Khalel W, Orhan-Sungur M, Meersch M, The E-AKII (2023) Epidemiology of surgery associated acute kidney injury (EPIS-AKI): a prospective international observational multi-center clinical study. Intensive Care Med 49(12):1441–1455
Chawla LS, Eggers PW, Star RA, Kimmel PL (2014) Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med 371:58–66
Bihorac A, Yavas S, Subbiah S, Hobson CE, Schold JD, Gabrielli A, Layon AJ, Segal MS (2009) Long-term risk of mortality and acute kidney injury during hospitalization after major surgery. Ann Surg 249:851–858
Weiss R, Saadat-Gilani K, Kerschke L, Wempe C, Meersch M, Zarbock A, Investigators E-A (2021) EPIdemiology of Surgery-Associated Acute Kidney Injury (EPIS-AKI): study protocol for a multicentre, observational trial. BMJ Open 11:e055705
Lombardi R, Nin N, Penuelas O, Ferreiro A, Rios F, Marin MC, Raymondos K, Lorente JA, Koh Y, Hurtado J, Gonzalez M, Abroug F, Jibaja M, Arabi Y, Moreno R, Matamis D, Anzueto A, Esteban A, Group V (2017) Acute kidney injury in mechanically ventilated patients: the risk factor profile depends on the timing of Aki Onset. Shock 48:411–417
Priyanka P, Zarbock A, Izawa J, Gleason TG, Renfurm RW, Kellum JA (2021) The impact of acute kidney injury by serum creatinine or urine output criteria on major adverse kidney events in cardiac surgery patients. J Thorac Cardiovasc Surg 162(143–151):e147
Myles PS, Bellomo R, Corcoran T, Forbes A, Peyton P, Story D, Christophi C, Leslie K, McGuinness S, Parke R, Serpell J, Chan MTV, Painter T, McCluskey S, Minto G, Wallace S, Australian, New Zealand College of Anaesthetists Clinical Trials N, the A, New Zealand Intensive Care Society Clinical Trials G (2018) Restrictive versus liberal fluid therapy for major abdominal surgery. N Engl J Med 378:2263–2274
Goodman LA (1965) On simultaneous confidence intervals for multinomial proportions. Technometrics 7:247–254
Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, Bittleman D, Cruz D, Endre Z, Fitzgerald RL, Forni L, Kane-Gill SL, Hoste E, Koyner J, Liu KD, Macedo E, Mehta R, Murray P, Nadim M, Ostermann M, Palevsky PM, Pannu N, Rosner M, Wald R, Zarbock A, Ronco C, Kellum JA, Acute Disease Quality Initiative W (2017) Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol 13:241–257
Harrell FE Jr (2016) Regression modeling strategies. Springer International Publishing
Hill NR, Fatoba ST, Oke JL, Hirst JA, O’Callaghan CA, Lasserson DS, Hobbs FD (2016) Global prevalence of chronic kidney disease—a systematic review and meta-analysis. PLoS ONE 11:e0158765
See EJ, Jayasinghe K, Glassford N, Bailey M, Johnson DW, Polkinghorne KR, Toussaint ND, Bellomo R (2019) Long-term risk of adverse outcomes after acute kidney injury: a systematic review and meta-analysis of cohort studies using consensus definitions of exposure. Kidney Int 95:160–172
Gameiro J, Marques F, Lopes JA (2021) Long-term consequences of acute kidney injury: a narrative review. Clin Kidney J 14:789–804
Forni LG, Darmon M, Ostermann M, Oudemans-van Straaten HM, Pettila V, Prowle JR, Schetz M, Joannidis M (2017) Renal recovery after acute kidney injury. Intensive Care Med 43:855–866
Hobson C, Ozrazgat-Baslanti T, Kuxhausen A, Thottakkara P, Efron PA, Moore FA, Moldawer LL, Segal MS, Bihorac A (2015) Cost and mortality associated with postoperative acute kidney injury. Ann Surg 261:1207–1214
Petaja L, Vaara S, Liuhanen S, Suojaranta-Ylinen R, Mildh L, Nisula S, Korhonen AM, Kaukonen KM, Salmenpera M, Pettila V (2017) Acute kidney injury after cardiac surgery by complete KDIGO criteria predicts increased mortality. J Cardiothorac Vasc Anesth 31:827–836
Muiru AN, Hsu JY, Zhang X, Appel LJ, Chen J, Cohen DL, Drawz PE, Freedman BI, Go AS, He J, Horwitz EJ, Hsu RK, Lash JP, Liu KD, McCoy IE, Porter A, Rao P, Ricardo AC, Rincon-Choles H, Sondheimer J, Taliercio J, Unruh M, Hsu CY, Investigators CS (2023) Risk for chronic kidney disease progression after acute kidney injury: findings from the chronic renal insufficiency cohort study. Ann Intern Med 176:961–968
Li Q, Li Y, Zhou F (2022) Duration of acute kidney injury predicts 90-day mortality and chronic kidney disease progression in elderly patients. J Intensive Med 2:110–117
Gameiro J, Duarte I, Marques F, Fonseca JA, Jorge S, Rosa R, Lopes JA (2020) Transient and persistent AKI and outcomes in patients undergoing major abdominal surgery. Nephron 144:236–244
Ostermann M, Zarbock A, Goldstein S, Kashani K, Macedo E, Murugan R, Bell M, Forni L, Guzzi L, Joannidis M, Kane-Gill SL, Legrand M, Mehta R, Murray PT, Pickkers P, Plebani M, Prowle J, Ricci Z, Rimmele T, Rosner M, Shaw AD, Kellum JA, Ronco C (2020) Recommendations on acute kidney injury biomarkers from the acute disease quality initiative consensus conference: a consensus statement. JAMA Netw Open 3:e2019209
Menez S, Moledina DG, Garg AX, Thiessen-Philbrook H, McArthur E, Jia Y, Liu C, Obeid W, Mansour SG, Koyner JL, Shlipak MG, Wilson FP, Coca SG, Parikh CR (2021) Results from the TRIBE-AKI Study found associations between post-operative blood biomarkers and risk of chronic kidney disease after cardiac surgery. Kidney Int 99:716–724
Ronco C, Kellum JA, Haase M (2012) Subclinical AKI is still AKI. Crit Care 16:313
Joannidis M, Forni LG, Haase M, Koyner J, Shi J, Kashani K, Chawla LS, Kellum JA, Sapphire I (2019) Use of cell cycle arrest biomarkers in conjunction with classical markers of acute kidney injury. Crit Care Med 47:e820–e826
Haase M, Kellum JA, Ronco C (2012) Subclinical AKI–an emerging syndrome with important consequences. Nat Rev Nephrol 8:735–739
Perazella MA (2012) Drug use and nephrotoxicity in the intensive care unit. Kidney Int 81:1172–1178
Goldstein SL (2016) Medication-induced acute kidney injury. Curr Opin Crit Care 22:542–545
Gray MP, Barreto EF, Schreier DJ, Kellum JA, Suh K, Kashani KB, Rule AD, Kane-Gill SL (2022) Consensus obtained for the nephrotoxic potential of 167 drugs in adult critically ill patients using a modified Delphi method. Drug Saf 45:389–398
(2013) Summary of recommendation statements. Kidney Int Suppl (2011) 3:5–14
Acknowledgements
The European Society of Anesthesiology and Intensive Care (ESAIC), among the following medical societies endorsed this study but had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript. However, we would like to thank these societies for their support on the promotion of EPIS-AKI: Colegio Mexicano de Medicina Crítica (COMMEC), Colombian Association of Surgery (CAS), Czech Society of Anesthesiology and Intensive Care (CSARIM), Deutsche Gesellschaft für Anästhesiologie und Intensivmedizin (DGAI), European Society of Anaesthesiology and Intensive Care (ESAIC), Korean Society of Anesthesiologists (KSA), Società Italiana di Anestesia Analgesia Rianimazione e Terapia Intensiva (SIAARTI), Société Française d’Anesthésie et de Réanimation (SFAR), South African Society of Anaesthesiologists (SASA), Spanish Perioperative Audit and Research Network (REDGERM), Turkish Anaesthesiology and Reanimation Society (TARD).
The EPIS AKI investigators: Algeria: Constantine Centre Hospitalier Universitaire (Hichem Makhloufi), Etablissement Hospitalier Spécialisé Salim Zemirli El Harrach (Rachida Sakhraoui), Saadna Abdenour Teaching Hospital (Amel Ouyahia, Mounira Rais, Aya Tinhinane Kouicem), Sabratha Libya National Cancer Institute (Khawla Derwish), University Hospital of Sétif (Meriem Abdoun, Ilhem Ouahab, Souad Bouaoud), University Hospital 1st November 1954 (Anisse Tidjane). Colombia: Universidad El Bosque, Fundación Cardioinfantil (Carlos Jose Pérez Rivera, Juan Pablo García). China: First Affiliated Hospital of Soochow University (Ke Peng, Fu-hai Ji, Zheng-min Ma). Egypt: Tanta University Faculty of Medicine (Mohamed Gamal Elbahnasawy, Shady Elsalhawy), Alexandria University Main Hospital (Ahmed Mahmoud Nafea), Alexandria University (Nermin A. Osman), Mansoura University, Faculty of Medicine, Gastrointestinal Surgical Center (Moataz Maher Emara, Mohamed Mamdouh Bonna), Zagazig University Hospital (Ibrahim Abdelmonaem Abdehaleem), Assiut University Hospital (Ahmed Mohamed Abbas, Mostafa Samy Abbas, Hany Mostafa Esmaeil). France: Centre Hospitalier Universitaire de Bordeaux (Oliver Joannes-Boyau), Centre Hospitalier Universitaire de Reims (Vincent Legros, Thierry Floch, Salvatore Muccio, Lison Menage-Innocenti, Benjamin Brochet, Marion Leclercq-Rouget), Centre Hospitalier Universitaire de Orléans (Claire Geneve, Bernardita Valenzuela Mocarquer), Hôpital Privé Sévigné (Christophe Aveline, Pierre Vautier), Hôpital Robert Schuman-Groupe UNEOS (Julien Nadaud), Hôpital Edouard Herriot (Thomas Rimmelé, Valérie Cerro), Gustave Roussy (Stéphanie Suria, Jamie Elmawieh , Rita El-Jawiche), Centre Hospitalier Universitaire de Lille (Cédric Cirenei, Gilles Lebuffe), Polyclinique de Limoges, Clinique François Chénieux (Sébastien Ponsonnard), Centre Hospitalier des Pays de Morlaix (Pierre-Yves Egreteau), Centre Hospitalier Universitaire de Nice, Université Côte d’Azur (Carole Ichai), Centre Hospitalier Dron Tourcoing (Vanessa Jean-Michel), Centre Hospitalier Universitaire d'Angers (Maxime Léger, Sigismond Lasocki, Charline Masson, Emmanuel Rineau , Viviane Cassisa), Centre François Baclesse (Pierre Verrier), Hôpital Bichat (Enora Atchade), Centre Hospitalier Simone Veil de Blois (Charles-Edouard Rochon), Centre Hospitalier de Bigorre (Vidal Quentin), Centre Hospitalier Universitaire de Bordeaux, Site Pellegrin (Nina Queixalos), Clinique Sainte Anne (Thierry Braun), Centre Hospitalier Libourne (Hubert Grand), Clinique Pasteur (Nicolas Mayeur, Marie Pasquie), Centre Hospitalier de Marne-La-Vallée (Pierre Garçon), Centre Hospitalier Universitaire de Nice (Vincent Bruckert), Centre Hospitalier Henri Mondor d'Aurillac (Gaël Pradel), Centre Hospitalier Mont de Marsan (Andersen Ramorasata), Centre Hospitalier de Dax (Céline Ravry), Clinique de la Sauvegarde (Nicolas Mottard). Germany: University Hospital Münster (Alexander Zarbock, Melanie Meersch, Raphael Weiss, Thilo von Groote, Christian Dörr, Mira Küllmar, Christina Massoth, Arash Motekallemi, Khaschayar Saadat-Gilani, Felix Albert, Laura Kerschke, Michael Storck, Julian Varghese, Carola Wempe), University Hospital RWTH Aachen (Linda Grüßer, Ana Kowark), University Hospital Düsseldorf (Timo Brandenburger), Kliniken Maria Hilf Mönchengladbach (Andreas Hohn), University Hospital Tübingen (Peter Rosenberger, Helene Häberle, Pascal Hofmann, Jonathan Kuhle, Stefanie Calov, Alice Marie Bernard, Valbona Mirakaj, Kathrin Weber, Kathrin Pfister, Lena Stetz), University Hospital Leipzig (Sarah Dorothea Müller), Herz-Jesu-Hospital Münster (Stephan Klaus, Marco Sadlo), St. Josef-Stift Sendenhorst (Christian Sengelhoff), Sankt Franziskus Hospital (Carina-Kristin Stenger, Ulrich Göbel) Hosspital Karlsburg (Matthias Heringlake). Greece: University Hospital Larissa (Eleni Arnaoutoglou), Laiko General Hospital of Athens (Panagiota Stratigopoulou), University Hospital Ioannina (Pantazi Danai), G. Gennimatas’ General Hospital of Athens (Antonia Dimakopoulou), General Hospital of Thessaloniki "George Papanikolaou" (Apostolos-Alkiviadis Menis), Aristotle University of Thessaloniki, General Hospital “George Papanikolaou” (Orestis Ioannidis). Iraq: Al-Diwaniyah Hospital (Humam Jalaawiy), Bagdad Medical City Hospital (Aeshah Anwar), Al-Nassiryah Teaching Hospital (Hashim Talib Hashim), Khanaqeen General Hospital (Hogir Imad Rasheed Aldawoody). Italy: Policlinico Paolo Giaccone (Andrea Cortegiani, Mariachiara Ippolito, Claudia Marino, Gabriele Presti, Dario Calogero Fricano), San Bortolo Hospital (Silvia De Rosa), Montebelluna City Hospital (Andrea Bianchin), San Carlo Regional Hospital (Gianluca Paternoster, Umberto Fasciano), Fondazione Policlinico Universitario A. Gemelli IRCCS and Catholic University of the Sacred Heart (Salvatore Lucio Cutuli), University of Ferrara (Spadaro Savino, Bussolati Enrico, Palmieri Marco, Volta Carlo Alberto), Great Metropolitan Hospital "Bianchi-Melacrino-Morelli" (Vincenzo Francesco Tripodi), Sant'Eugenio Hospital (Diego Fiume), Frangipane Hospital (Angela Iuorio), General Hospital Vallecamonica (Clemente Santorsola). Jordan: Al-Basheer Hospital Amman (Bilal Abu-Hussein, Khaled Hasanein). (South) Korea: Yonsei University College of Medicine (Seokyung Shin), University Hospital Yeungnam, Yeungnam School of Medicine (Jongyoon Baek, Sehui Kim). Libya: University of Tripoli (Muhammed Elhadi), Benghazi Medical Center (Wafa Aldressi, Issa A. Abuzeid, Mohammed N. Albaraesi, Mohamed Aziz Moftah, Sarah Aldressi), Tripoli Central Hospital (Wegdan Khalel, Eman Abdulwahed, Entisar Ahmed Ali Alshareea, Akram Abdulhamid Ashur Abujrad Reem Ghmagh, Marwa Isa Biala), National Cancer Institute Sabratha (Khawla Derwish), National Heart Center Tripoli (Rayet Al Islam Benjouira, Mohamed Aliwa), University Hospital Tripoli (Ahmed Msherghi, Ahmed Tuwaib, Tahani Mustafa, Haifa Zriba), AboSleem Hospital (Hamza Mahmoud Agilla), El-Khadra Hospital (Bahaeddin Taher Sadek Ben Hamida, Rema Hassan Mohamed Otman). Macedonia: University Hospital Skopje Mother Theresa (Maja Mojsova Mijovska). Malta: Mater Dei Hospital (Anne Marie Camilleri Podesta). Mexico: Regional Hospital of High Specialty of Ixtapaluca (Gilberto Adrián Gasca López). Palestine: Princess Alia Governmental Hospital (Sarah Amro). Portugal: Hospital São Bernardo, Central Hosptial of Setúbal (Rita de Freitas Regufe). Russia: Kemerovo Cardiology Centre (Evgeny Grigoryev, Artem Ivkin, Dmitriy Balakhnin, Dmitriy Shukevich), A.N. Bakulev National Medical Research Center of Cardiovascular Surgery (Michael Yaroustovsky). Saudi Arabia: Almana General Hospital Al-Khobar (Abdulnaser Barmou). Switzerland: University Hospital Zurich (Alexander Kaserer, Clara Castellucci, Samira Akbas). Slovenia: University Medical Centre Maribor (Andreja Möller Petrun, Irena Gregorcic, Vesna Sok). South Africa: Stellenbosch University, Faculty of Medicine and Health Sciences (Andre Links). Spain: University Hospital Virgen de las Nieves(Elizabeth Bárcena Barreto), University Hospital Infanta Leonor (Javier Ripollés Melchor), University Hospital Gran Canaria Doctor Negrín (Ángel Becerra-Bolaños, Aurelio Rodríguez-Pérez), University Hospital Son Llàtzer (Javier Mata Estévez, Juan Mulet Matas, Sara Pérez Palao), Hospital de Sant Pau (Mercedes García Álvarez, Albert Bainac Albadalejo, Astrid Batalla González, Ana María Gómez Caro, Ignacio Hinojal Blanco, Diego Toral Fernandez, Gracia Herranz Perez), Txagorritxu Hospital Álava (Margarita Logroño Ejea, Noelia de la Rosa Ruiz, María Gastaca Abasolo), San Pedro Hospital (Lourdes Ferreira, Félix Lobato, Marta Aguado Sevilla), Fundación Puigvert, (Andres Erazo), University Hospital Donostia (Berta Castellano Paulis), Hospital 12 de Octubre (Isabel de la Calle Gil), University Hospital Bellvitge (Peter Adamove, Francho Miguel Blasco Blasco), University Hospital Fundacion Alcorcón (Jose Ignacio García-Sánchez, Sara García Zamorano, Natalia Gijón Herreros), Navarra University Clinic (Raquel Callejas), University Hospital Ciudad Real (Mercedes Estaire Gómez), University Hospital Ramón y Cajal (Angel M. Candela-Toha, Elisabeth Claros-Llamas, Pilar Cobeta-Orduña, Pascual Crespo-Aliseda, Trinidad Dorado-Díaz, María Gómez-Rojo, M. Nuria Mané-Ruiz, M. Carmen Martín-González, Adolfo Martínez-Pérez, Carlos Tiscar), University Hospital Vall d’Hebron (Patricia Galán Menéndez, Verónica Estepa Calvo, Laura Llinares Espí, Yuri Santiago Loaiza Aldeán, Víctor Morales Ariza, Laura Villarino Vila), La Gerencia de Asistencia Sanitaria de Segovia (Francisco Javier García-Miguel). Sudan: Sharg Alneel Hospital & Omdurman Military Hospital (Elfayadh S. M. Suliman), Gadarif Teaching Hospital (Ahmed Mohamed Ibrahim), Ahmed Gasim Hospital (Hammad Ali. Fadlalmola). Syria: Aleppo, Syria Faculty of Medicine (Sarya Swed). Taiwan: National Taiwan University Hospital and College of Medicine (Vin-Cent Wu). Turkey: Istanbul University Istanbul Faculty of Medicine (Mukadder Orhan-Sungur, Demet Altun, Nur Canbolat, Müşerref Beril Dinçer), Acibadem Mehmet Ali Aydinlar University School of Medicine (Serap Aktas Yildirim), Acibadem Mehmet Ali Aydinar University School of Medicine, Atakent Hospital (Muzeyyen Iyigun), Mersin University (Davud Yapıcı, Levent Özdemir, Aslınur Sagün), Akdeniz University (Neval Boztug, Emel Gündüz,), Cukurova University (Demet Lafli-Tunay), Ondokuz Mayis University (Deniz Karakaya, Burhan Dost, Ozgur Komurcu), Istanbul University-Cerrahpasa (Ozlem Korkmaz Dilmen, Eren Fatma Akcil, Yusuf Tunali), Manisa Celal Bayar University (Gulay Ok, Eda Tok-Alsina), Prof. Dr. Cemil Taşcıoğlu City Hospital (Cengiz Polat), Yeditepe University (Nurcan Kızılcık), University of Health Sciences Haseki Education and Training Hospital (Öznur Şen), Koç University (Kamil Darçın, Semra Uğur , Yavuz Gürkan), Health Sciences University Kartal Dr Lutfi Kirdar City Hospital (Kemal Tolga Saracoglu, Özge Yıldız-Koyuncu), Ankara Bilkent City Hospital (Z. Aslı Demir, N. Aysun Postacı, Ayşegül Özgök, Ümit Karadeniz, Hülya Yiğit Özay, Eda Balcı, Nevriye Salman, Behiç Girgin), Balikesir University (Ozlem Sagir, Hafize Fisun Demir, Fatih Ugun), Inonu University (Hüseyin İlksen Toprak), Süleyman Demirel University ( Mustafa Soner Özcan, Filiz Alkaya-Solmaz), Derince Training And Research Hospital (Mehmet Yilmaz), Bursa Yüksek Ihtisas Training and Research Hospital (Umran Karaca), Trakya University (Sevtap Hekimoglu Şahin), Ankara University İbni Sina Hospital (Süheyla Karadağ Erkoç, Neslihan Alkış), Cebeci Hospital (Volkan Baytaş), Karadeniz Technical University (Engin Erturk, Sedat Saylan, Ali Akdogan), Gazi University School of Medicine (Beyza Büyükgebiz Yeşil), Kahramanmaraş Sütçü İmam University Faculty of Medicine (Omer Faruk Boran, Yavuz Orak, Feyza Çalişir), Dokuz Eylül University (Sibel Büyükçoban, Bahar Kuvaki), Bursa Uludag University (Seda Cansabuncu, Selcan Akesen, Suna Gören), Baltalimani Metin Sabanci Bone and Joint Diseases Education and Research Hospital (Tugce Yeniocak, Osman Orman), Duzce University (Özlem Ersoy Karka), Kocaeli University (Tulay Sahin). Ukraine: Zaporizhzhia State Medical University Hospital (Natalia Momot, Anna Panchenko). USA: University of Alabama (Jean-Francois Pittet, Kristen Rutledge).
Funding
Open Access funding enabled and organized by Projekt DEAL. The study was supported by the German Research Foundation (KFO342-1, ZA428/18-1, and ZA428/21-1 to AZ and ME5413/1-1 and ME5413/1-2 to MM) and by an unrestricted research grant from Baxter (to AZ).
Author information
Authors and Affiliations
Consortia
Contributions
MM and AZ conceived and designed the study; FA and MM performed statistical analysis; MM, RW, CR, HB, JFP, PR, AZ acquired data; MM, FA, RM, RB, JAK, AZ drafted the manuscript; RW, CS, HB, JFP, MR, PR, LF made critical revision of the manuscript for key intellectual component. All authors provided final approval of the final version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
MM received lecture fees from Fresenius Medical Care and Baxter unrelated to current study. JAK is a paid consultant for BioMerieux, and a fulltime employee of Spectral Medical. AZ received lecture fees from Biomerieux, Fresenius Medical Care and Baxter unrelated to current study and an unrestricted research grant from Baxter related to the current study. All other authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
EPIS-AKI investigators are listed in the Acknowledgement section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Meersch, M., Weiss, R., Strauß, C. et al. Acute kidney disease beyond day 7 after major surgery: a secondary analysis of the EPIS-AKI trial. Intensive Care Med 50, 247–257 (2024). https://doi.org/10.1007/s00134-023-07314-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-023-07314-2