Abstract
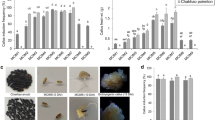
We investigated the frequency of callus induction from mature-seed scutella of 39 varieties of Tongil rice(Oryza sativa japonica x indica). These were divided into two groups according to condition of the callus at the time of induction. Members of the first group, which developed brown calli, were further classified into two types (B1 or B2), based on the extent of their color change and callus condition. Tissues in the second group developed yellow calli, with varieties being placed into one of three types (Y1, Y2, or Y3) according to solidity. Type-Y1 seeds generated suitably soft calli that were healthy and grew rapidly. These calli were transformed with binary vector pGA2722 viaAgrobacterium- mediated co-cultivation. The vector contained the hygromycin-resistance selectable marker and agus reporter gene with an intron that is functional in plant cells but not inAgrobacterium. Most varieties that formed brown calli failed to generate transformants. Likewise, several varieties with suitably soft calli also failed. Among the 16 transformable varieties, the final regeneration frequencies ranged from 1.0 to 10.7%. The highest frequency was achieved by the variety Tongil, which also had the most rapidly growing calli. DNA gel-blot analysis revealed one to three copies of the introducedgus gene.
Similar content being viewed by others
Literature cited
Abe T, Futsuhara Y (1986) Cenotypic variability for callus formation and plant regeneration in rice. Theor Appl Genet72: 5–10
Aldemita RR, Hodges TK (1996)Agrobacterium tumefaciens-mediated transformation of japonica and indica rice varieties. Planta199: 612–617
Anonymous (1995) Food and Agriculture Organization of the United Nations. FAO Quart Bull Stat8: 1–2
Bajaj S, Rajam MV (1995) Efficient plant regeneration from long-term callus cultures of rice by spermidine. Plant Cell Rep14: 717–720
Bajaj S, Rajam MV (1996) Polyamine accumulation and near-loss of morphogenesis in long-term callus cultures of rice: Restoration of plant regeneration by manipulation of cellular polyamine levels. Plant Physiol112: 1343–1348
Binns AN, Thomashow MF (1988) Cell biology ofAgrobacterium infection and transformation of plants. Annu Rev Microbiol42: 575–606
Chan MT, Chang HH, Ho SL, Tong WF, Yu SM (1993) Agrobacterium-mediated production of transgenic rice plants expressing a chimaeric α-amylase-promoter/ß- glucuronidase gene. Plant Mol Biol22: 491–506
Chan MT, Lee TM, Chang HH (1992) Transformation of indica rice (Oryza sativa L.) mediated byAgrobacterium tumefaciens. Plant Cell Physiol33: 577–583
Chen DH, Ronald PC (1999) A rapid DNA minipreparation method suitable for AFLP and other PCR applications. Plant Mol Biol Rep17: 53–57
Cho SK, Chung YS, Park SJ, Shin JS, Kwon HJ, Kang KH (1998) Efficient transformation of Korea rice cultivars (Oryzasativa L.) mediated byAgrobacterium tumefaciens. J Plant Biol41: 262–268
Donaldson PA, Simmonds DH (2000) Susceptibility toAgrobacterium tumefaciens and cotyledonary node transformation in short-season soybean. Plant Cell Rep19: 478–484
Goff S, Riche A, Lan D (2002) Draft sequence of the rice genome (Oryzasativa L.) ssp. japonica Science296: 92–100
Hiei Y, Komari T, Kubo T (1997) Transformation of rice mediated byAgrobacterium tumefaciens. Plant Mol Biol35:205–218
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice(Oryza sativa L.) mediated byAgrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J6: 271–282
Hoekema A, Hirsch PR, Hooykaas P], Schilperoort RA (1983) A binary plant vector strategy based on separation ofvir- and T-region of theAgrobacterium tumefaciens Ti-plasmid. Nature303: 179–180
lida A, Yamashita Y, Yamada Y, Morikawa H (1991) Efficiency of particle-bombardment-mediated transformation is influenced by cell stage in synchronized cultured cells of tobacco. Plant Physiol97: 1585–1587
Jefferson RA (1987) Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol Biol Rep5: 387–405
Kudirka DT, Colburn SM, Hinchee MA, Wright MS (1986) Interactions ofAgrobacterium tumefaciens with soybean(Glycine max L, Merr.) leaf expiants in tissue culture. Can J Genet Cytol28: 808–817
Kwon YS, Sohn JK (2000) Varietal difference and inheritance of plant regenerability in anther culture of rice. Kor J Plant Tiss Cult27: 163–167
Lee S, Jeon JS, Jung KH, An G (1999) Binary vectors for efficient transformation of rice. J Plant Biol42: 310–316
Li XQ, Liu CN, Ritchie SW, Pang JY, Gelvin SB, Hodges TK (1992) Factors influencing Agrobacterium-mediated transient expression ofgusA in rice. Plant Mol Biol20: 1037–1048
Mohanty A, Sarma NP, Tyagi AK (1999)Agrobacterium-mediated high frequency transformation of an elite indica rice variety Pusa Basmati 1 and transmission of the transgenes to R2 progeny. Plant Sci147: 127–137
Raineri DM, Bottino P, Gordon MR Nester EW (1990)Agrobacterium mediated transformation of rice(Oryza sativa L). Bio/Technology8: 33–38
Rashid H, Yokoi S, Toriyama K, Hinata K (1996) Transgenic plant production mediated byAgrobacterium in indica rice. Plant Cell Rep15: 727–730
Ryu S, Chung KC, Chi YT, Cheong H (2001) Expression of chicken cystatin for improving insect resistance in rice. J Plant Biol44: 205–212
Shoeb F, Yadav JS, Bajaj S, Rajam MV (2001) Polyamines as biomarkers for plant regeneration capacity: Improvement of regeneration by modulation of polyamine metabolism in different genotypes of indica rice. Plant Sci160: 1229–1235
Sohn JK, Kim KM, Kim JS (1995) Plant regeneration and somatic embryo formation from root-derived callus of rice. KorJ Plant Tiss Cult22: 143–147
Toki S (1997) Rapid and efficient Agrobacterium-mediated transformation in rice. Plant Mol Biol Rep15: 16–21
Valvekens D, van Montagu M, van Lijsebettens M (1988)Agrobacterium tumefaciens-mediated transformation ofArabidopsis thaliana root expiants by using kanamycin selection. Proc Natl Acad Sci USA85: 5536–5540
Vijayachandra K, Palanichelvam K, Veluthambi K (1995) Rice scutellum inducesAgrobacterium tumefaciens vir genes and T-strand generation. Plant Mol Biol29: 125–133
Wullems GJ, Molendijk L, Ooms G, Schilperoort RA (1981) Differential expression of crown gall tumor markers in transformants obtained afterin vitro Agrobacterium tumefaciens-induced transformation of cell wall-regeneration protoplasts derived fromNicotiana tabacum. Proc Natl Acad Sci USA78: 4344–4348
Yu J, Hu S, Wang J (2002) A draft sequence of the rice genome(Oryza sativa L. ssp. indica Science296: 79–92
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, M.C., Shin, J., Kim, N. et al. High-frequencyAgrobacterium-med’iated genetic transformation of tongil rice varieties. J. Plant Biol. 46, 23–30 (2003). https://doi.org/10.1007/BF03030297
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03030297