Abstract
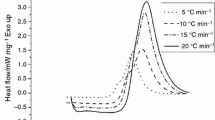
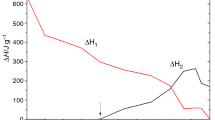
Differential scanning calorimetry (DSC) was applied to study the cure kinetics of an epoxy system containing both tetraglycidyl 4,4′-diaminodiphenylmethane (TGDDM) and a multifunctional Novolac glycidyl ether resin, cured with 4,4′-diaminodiphenylsulfone (DDS). The experimental data were analyzed in terms of a mechanistic model proposed by Cole, which includes the etherification reaction. The kinetics can be completely described in terms of three rate constants, which obey the Arrhenius relationship. This model gives a good description of the cure kinetics up to the onset of vitrification. The effect of diffusion control was incorporated to describe the cure in the later stages. By combining the model and a diffusion factor, it was possible to predict the cure kinetics over the whole range of conversion, including an analysis of the evolution of different chemical species during the curing process. Good agreement with the experimental DSC data was achieved with this mechanistic model over the whole range of cure when the etherification reaction was assumed to be of first order with respect to the concentrations of epoxide groups, hydroxy groups, and the tertiary amine groups formed in the epoxide amine reaction.
Similar content being viewed by others
References
J. Mijovic, J. Kim and J. Slaby, J. Appl. Polym. Sci., 29 (1984) 1449.
J. M. Barton, Br. Polym. J., 18 (1986) 37.
R. J. Morgan and E. T. Mones, J. Appl. Polym. Sci., 33 (1987) 999.
L. Chiao, Macromolecules, 23 (1990) 1286.
J. D. Keenan, J. C. Seferis and J. T. Quinlivan, J. Appl. Polym. Sci., 24 (1979) 2375.
E. B. Stark, A. M. Ibrahim and J. C. Seferis, Interrelations between Processing Structure and Properties of Polymeric Materials, J. C. Seferis and P. S. Theocaris (eds), Elsevier, Amsterdam 1984.
K. C. Cole, J. J. Hechler and D. Noel, Macromolecules., 24 (1991) 3098.
K. C. Cole, Macromolecules, 24 (1991) 3093.
L. Matejka and K. Dusek, Macromolecules, 22 (1989) 2902.
J. Mijovic, A. Fishbain and J. Wijana, Macromolecules, 25 (1992) 979.
J. Mijovic and J. Wijana, Polym. Commun, 35 (1994) 2683.
J. M. Barton, Adv. Polym. Sci., 72 (1985) 111.
K. Horie, H. Hiura, M. Sawada, L. Mita and H. Kambe, J. Polym. Sci. Part A-l, 8 (1970) 1357.
H. Jahn and P. Goetzky, Epoxy Resins. Chemistry and Technology. 2nd ed, C. A. May Ed, Marcel Dekker, 1988. Chap 13.
W. I. Lee, A. C. Loos and G. S. Springer, J. Compos. Mater. 16 (1982) 510.
C. S. Chern and G. W. Poehlein, Polym. Eng. Sci., 27 (1987) 782.
S. E. Rabinowitch, Trans Faraday Soc., 33 (1937) 1225.
U. Khanna and M. Chanda, J. Appl. Polym. Sci., 49 (1993) 319.
L. Barrai, J. Cano, A. J. López, J. López, P. Nogueira and C. Ramírez, J. Appl. Polym. Sci., 56 (1995) 1029.
L. Barrai, J. Cano, J. López, P. Nogueira, C. Ramírez and M. J. Abad, Polym. Int., 42 (1997) 301.
Author information
Authors and Affiliations
Additional information
This work was supported by the Xunta de Galicia through grant XUGA-17201A95. The authors wish to thank Ciba-Geigy for supplying the MY 720 and EPN 1138 prepolymers.
Rights and permissions
About this article
Cite this article
Barral, L., Cano, J., López, J. et al. Cure kinetics of an epoxy system containing tetraglycidyl-4,4′-diaminodiphenylmethane (TGDDM) and a multifunctional Novolac glycidyl ether cured with 4,4′-diaminodiphenylsulfone (DDS). Journal of Thermal Analysis 50, 409–423 (1997). https://doi.org/10.1007/BF01980501
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01980501