Summary
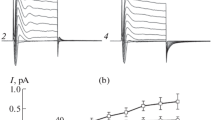
Patch-clamp techniques have been applied to characterize the channels in the basolateral membrane of resting (cimetidine-treated, nonacid secreting) oxyntic cells isolated from the gastric mucosa ofNecturus maculosa. In cell-attached patches with pipette solution containing 100mm KCl, four major classes of K+ channels can be distinguished on the basis of their kinetic behavior and conductance: (1) 40% of the patches contained either voltage-independent (a) or hyperpolarization-activated (b), inward-rectifying channels with short mean open times (16 msec fora, and 8 msec forb). Some channels showed subconductance levels. The maximal inward conductanceg max was 31±5 pS (n=13) and the reversal potentialE rev was atV p=−34±6 mV (n=9). (2) 10% of the patches contained depolarization-activated and inward-rectifying channels withg max=40 ±18 pS (n=3) andE rev was atV p=−31±5 mV (n=3). With hyperpolarization, the channels open in bursts with rapid flickerings within bursts. Addition of carbachol (1mm) to the bath solution in cell-attached patches increased the open probabilityP o of these channels. (3) 10% of the patches contained voltage-independent inward-rectifying channels withg max=21±3 pS (n=4) andE rev was atV p=−24±9 mV (n=4). These channels exhibited very high open probability (P o=0.9) and long mean open time (1.6 sec) at the resting potential. (4) 20% of the patches contained voltage-independent channels with limiting inward conductance of 26±2 pS (n=3) andE rev atV p=−33±3 mV (n=3). The channels opened in bursts consisting of sequential activation of multiple channels with very brief mean open times (10 msec). In addition, channels with conductances less than 6 pS were observed in 20% of the patches. In all nine experiments with K+ in the pipette solution replaced by Na+, unitary currents were outward, and inward currents were observed only for large hyperpolarizing potentials. This indicates that the channels are more selective for K+ over Na+ and Cl−. A variety of K+ channels contributes to the basolateral K+ conductance of resting oxyntic cells.
Similar content being viewed by others
References
Barrett, J.N., Magleby, K.L., Pallotta, B.S. 1982. Properties of single calcium-activated potassium channels in cultured rat muscle.J. Physiol. 331:221–230
Berglindh, T. 1978. The effects of K+ and Na+ on acid formation in isolated gastric glands.Acta Physiol. Scand. Spec. Suppl.: 55–68
Blum, A.L., Shah, G.T., Wiebelhaus, V.D., Brennan, F.T., Helander, H.F., Ceballos, R., Sachs, G. 1971. Pronase method for isolation of viable cells fromNecturus gastric mucosa.Gastroenterology 61:189–200
Butt, A.G., Clapp, W.L., Frizzell, R.A. 1990. Potassium conductances in tracheal epithelium activated by secretion and cell swelling.Am. J. Physiol. 258:C630-C638
Colquhoun, D., Sigworth, F.J. 1983. Fitting and statistical analysis of single-channel records.In: Single-Channel Recording. B. Sakmann and E. Neher, editor. pp. 191–263 Plenum, New York
Demarest, J.R. 1991. Potassium channel-dependent membrane potential oscillations in oxyntic cells.Biophys. J. 59:649(abstr.)
Demarest, J.R., Loo, D.D.F., Sachs, G. 1989. Activation of apical chloride channels in the gastric oxyntic cell.Science 245:402–404
Demarest, J.R., Machen, T.E. 1985. Microelectrode measurements from oxyntic cells in intactNecturus gastric mucosa.Am. J. Physiol. 23:C535-C540
Ganser, A.L., Forte, J.G. 1973. K+-stimulated ATPase in purified microsomes of bullfrog oxyntic cells.Biochim. Biophys. Acta 307:169–180
Germann, W.J., Ernst, S.A., Dawson, D.C. 1986. Differentiation of two distinct K conductances in the basolateral membrane of turtle colon.J. Gen. Physiol. 88:237–252
Harris, J.B., Edelman, I.S. 1960. Transport of potassium by the gastric mucosa of the frog.Am. J. Physiol. 198:280–284
Hille, B. 1984. Ionic channels of excitable membranes. Sinauer, Sunderland (MA)
Latorre, R., Oberhauser, A., Labarca, P., Alvarez, O. 1989. Varieties of calcium-activated potassium channels.Annu. Rev. Physiol. 51:385–399
Lau, K.R., Hudson, R.L., Schultz, S.G. 1984. Cell swelling increases a barium-inhibitable potassium conductance in the basolateral membrane ofNecturus small intestine.Proc. Natl. Acad. Sci. USA 81:3591–3594
Makhlouf, G.M. 1981. Electrolyte composition of gastric secretion.In: Physiology of the Gastrointestinal Tract. L.R. Johnson, editor. pp. 551–566. Raven, New York
Mieno, H., Shirakawa, T., Inoue, M., Kajiyama, G. 1990. Patchclamp studies of K+ channels in frog oxyntic cells.Gastroenterology 98:A548(abstr.)
Moczydlowski, E., Latorre, R. 1983. Gating kinetics of Ca2+-activated K+ channels from rat muscle incorporated into planar lipid bilayers. Evidence for two voltage-dependent Ca2+-binding reactions.J. Gen. Physiol. 82:511–542
Muallem, S., Burnham, C., Blissard, D., Berglindh, T., Sachs, G. 1985. Electrolyte transport across the basolateral membrane of the parietal cell.J. Biol. Chem. 260:6641–6653
Muallem, S., Sachs, G. 1985. Ca2+ metabolism during cholinergic stimulation of acid secretion.Am. J. Physiol. 248:G216-G228
Negulescu, P.A., Machen, T.E. 1988. Intracellular Ca regulation during secretagogue stimulation of the parietal cell.Am. J. Physiol. 254:C130-C140
Rae, J.L., Levis, R.A., Eisenberg, R.S. 1988. Ionic channels in ocular epithelia.In: Ion Channels. Vol 1, pp. 283–327. T. Narahashi, editor Plenum, New York
Sachs, G., Chang, H.H., Rabon, E., Schackmann, R., Lewin, M., Saccomani, G. 1976. A nonelectrogenic H+ pump in plasma membrane of hog stomachs.J. Biol. Chem. 251:7690–7698
Sakai, H., Okada, Y., Morii, M., Takeguchi, N. 1989. Anion and cation channels in the basolateral membrane of rabbit parietal cells.Pfluegers Arch. 414:185–192
Ueda, S., Loo, D.D.F., Sachs, G. 1987. Regulation of K+ channels in the basolateral membrane ofNecturus oxyntic cells.J. Membrane Biol. 97:31–41
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Supplisson, S., Loo, D.D.F. & Sachs, G. Diversity of K+ channels in the basolateral membrane of restingnecturus oxyntic cells. J. Membrain Biol. 123, 209–221 (1991). https://doi.org/10.1007/BF01870404
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01870404