Summary
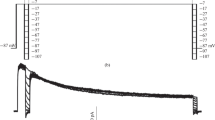
We have used single electrode voltage clamp in the intact animal and whole-cell recording from dissociated cell bodies to investigate the properties of potassium conductances in large monopolar cells (LMCs) of the first optic ganglion of the blowfly Calliphora vicina. Two classes of voltage gated potassium conductances were found: a delayed rectifier current (Kd) with slow inactivation (τinac = 1–3 sec), and an A current (Ka) showing both faster inactivation (τinac = 21 ms) and also more rapid activation. The reversal potential of both currents is ca. -90 mV with 2 mM [Ko] and 140 mM [Ki], and follows the Nernst slope with increasing [Ko]. The voltage operating range of Ka is unusually negative, with the mid point of the steady-state inactivation curve (V50) at- 101 mV. V50 for Kd is - 84 mV. Although no inward currents were detected, for technical reasons their presence cannot be excluded.
In inside-out patches from LMC soma membranes the single channels underlying the currents both have a conductance of ca. 20 pS in symmetrical 140 mM K solutions and channel densities may be as high as 10/μm2. Less frequently, inside-out patches contained a large conductance (110 pS) calcium-activated potassium channel which existed almost exclusively in a rapidly flickering mode. Open probability increased with depolarization and Ca concentrations greater than 40 nM.
In whole-cell recordings, dissociated LMC cell bodies fall into two classes with respect to their voltage sensitive currents: 37 % of cells only showed Kd; the remainder (63%) were dominated by Ka with a variable (0–30%) contribution from Kd. In the intact animal, intracellular recordings from LMCs, combined with dye-marking, indicate that cells expressing only Kd are type L3 cells, whilst L1 and L2 express predominantly Ka. Since L1 and L2 have resting potentials of ca. - 40 mV and maximum hyperpolarizations reaching -90 mV only transiently, inactivation of Ka is unlikely to be removed under most physiological conditions. In contrast, L3 cells have a more negative resting potential (−60 mV) and Kd should play a significant role in signal-shaping, in particular contributing to the falling phase of a prominent spike-like transient in response to dimming.
Similar content being viewed by others
Abbreviations
- Ka :
-
A current
- Kd :
-
delayed rectifier
- LMC :
-
large monopolar cell
- L1-L3 :
-
classes thereof
- TTX :
-
tetrodotoxin
References
Ammermüller J, Zettler F (1986) Time- and voltage-dependent currents in locust ocellar L-neurones. J Comp Physiol A 159:363–376
Atkins PT, Surmeier DJ, Kitai ST (1990) Muscarinic modulation of a transient K+ conductance in rat neostriatal neurons. Nature 344:240–242
Autrum H, Zettler F, Järvilehto M (1970) Postsynaptic potentials from a single neuron of the ganglion opticum I of the blowfly Calliphora erythrocephala. Z Vergl Physiol 48:357–384
Byerly L, Leung H-T (1988) Ionic current in Drosophila neurons in embryonic cultures. J Neurosci 8:4379–4393
Cull-Candy SG, Marshall CG, Ogden D (1989) Voltage-activated membrane currents in rat cerebellar granule neurones. J Physiol 414:179–199
Fenwick EM, Marty A, Neher E (1982) A patch-clamp study of bovine chromaffin cells and their sensitivity to acetylcholine. J Physiol 331:577–597
Finkel AS, Redman SJ (1984) Theory and operation of a single microelectrode voltage clamp. J Neurosci Methods 11:101–127
Ganetzky B, Wu C-F (1985) Genes and membrane excitability in Drosophila. Trends Neurosci 8:322–326
Guy RG, Srinivasan MV (1988) Integrative properties of second-order visual neurons: a study of large monopolar cells in the dronefly Eristalls. J Comp Physiol A 162:317–332
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth F (1981) Improved patch clamp techniques for high resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391:85–100
Hardie RC (1987) Is histamine a neurotransmitter in insect photoreceptors? J Comp Physiol A 161:201–213
Hardie RC (1989) A histamine-gated chloride channel involved in neurotransmission at a photoreceptor synapse. Nature 339:704–707
Hardie RC, Laughlin SB, Osorio D (1989) Early visual processing in the compound eye: physiology and pharmacology of the retina-lamina projection in the fly. In: Singh RN, Strausfeld NJ (eds) Neurobiology of sensory systems. Plenum Press, New York London, pp 23–42
Hateren JH van, Laughlin SB (1990) Membrane parameters, signal transmission, and the design of a graded potential neuron. J Comp Physiol A 166:437–448
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117:500–544
Hoshi T, Aldrich RW (1988) Voltage-dependent K+ currents and underlying single K+ channels in Pheochromocytoma cells. J Gen Physiol 91:73–106
Järvilehto M (1985) The eye: vision and perception. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology. Pergamon, Oxford, pp 355–429
Järvilehto M, Zettler F (1971) Localized intracellular potential from pre- and postsynaptic components in the external plexiform layer of an insect retina. Z Vergl Physiol 75:422–440
Kaneko A, Tachibana M (1985) A voltage-clamp analysis of membrane currents in solitary bipolar cells dissociated from Carassius auratus. J Physiol 358:131–152
Kasai H, Kameyama M, Yamaguchi K, Fukida J (1986) Single transient K+ channels in mammalian sensory neurones. Biophys J 49:1243–1247
Lasater EM (1988) Membrane currents of retinal bipolar cells in culture. J Neurophysiol 60:1460–1480
Latorre R, Oberhauser A, Labarca P, Alvarez O (1989) Varieties of calcium-activated potassium channels. Ann Rev Physiol 51:385–399
Laughlin SB (1981) Neural principles in the peripheral visual systems of invertebrates In: Autrum H (ed) Handbook of sensory physiology Vol VII/6b. Springer, Berlin Heidelberg New York, pp 133–280
Laughlin SB (1987) Form and function in retinal processing. Trends Neurosci 10:478–483
Laughlin SB (1989) Coding efficiency and design in visual processing. In: Stavenga DG, Hardie RC (eds) Facets of vision. Springer, Berlin Heidelberg New York, pp 212–234
Laughlin SB, Hardie RC (1978) Common strategies for light adaptation in the peripheral visual systems of fly and dragonfly. J Comp Physiol 128:319–340
Laughlin SB, Osorio D (1989) Mechanisms for neural signal enhancement in the blowfly compound eye. J Exp Biol 144:113–146
Laurent G (1990) Voltage-dependent non-linearities in the membrane of locust nonspiking local interneurones, and their significance for synaptic integration. J Neurosci (in press)
Mackie GO, Meech RW (1989) Potassium channel family in the axons of jellyfish Aglantha digitale. J Physiol 418:14P
Marty A, Neher E (1985) Potassium channels in cultured bovine adrenal chromaffin cells. J Physiol 367:117–141
Mizunami M, Yamashita S, Tateda H (1987) Calcium-dependent action potentials in the second-order neurones of cockroach ocelli. J Exp Biol 130:259–274
Nicol D, Meinertzhagen IA (1982) An analysis of the number and composition of the synaptic populations formed by photoreceptors of the fly. J Comp Neurol 207:29–44
Nightingale WD, Pitman RM (1989) Ionic currents in the soma of an identified cockroach motorneuron recorded under voltage clamp. Comp Biochem Physiol 93A:85–93
O'Sullivan AJ, Cheek TR, Moreton RB, Berridge MJ, Burgoyne RD (1989) Localization and heterogeneity of agonist-induced changes in cytosolic calcium concentration in single bovine adrenal chromaffin cells from video imaging of fura-2. EMBO J 8:401–411
Pinnock RD, Sattelle DB (1987) Dissociation and maintenance in vitro of neurones from adult cockroach (Periplaneta americana) and housefly (Musca domestica). J Neurosci Methods 20:195–202
Rudy B (1988) Diversity and ubiquity of K channels. Neuroscience 25:729–749
Shaw SR (1984) Early visual processing in insects. J Exp Biol. 112:225–251
Shaw SR (1989) The retina-lamina pathway in insects, particularly Diptera, viewed from an evolutionary perspective. In: Stavenga DG, Hardie RC (eds) Facets of Vision. Springer, Berlin Heidelberg New York, pp 186–212
Solc CK, Aldrich RW (1988) Voltage-gated potassium channels in larval CNS neurons of Drosophila. J Neurosci 8:2256–2570
Stewart WW (1978) Functional connections between cells as revealed by dye-coupling with a highly fluorescent naphthalimide tracer. Cell 14:741–759
Strausfeld NJ (1971) The organisation of the insect visual system (light microscopy). I. Projection and arrangements of neurons in the lamina ganglionaris of Diptera. Z Zellforsch 121:377–441
Strausfeld NJ (1976) Atlas of an insect brain. Springer, Berlin Heidelberg New York
Strausfeld NJ (1984) Functional neuroanatomy of the blowfly's visual system. In: Ali MA (ed) Photoreception and vision in invertebrates. Plenum Press, New York London, pp 483–522
Strausfeld NJ (1989) Insect vision and olfaction: common design principles of neuronal organization. In: Singh RN, Strausfeld NJ (eds) Neurobiology of sensory systems. Plenum Press, New York London, pp 319–353
Tessier-Lavigne M, Attwell D, Mobbs P, Wilson M (1988) Membrane currents in retinal bipolar cells of the axolotl. J Gen Physiol 91:49–72
Thomas MV (1982) Techniques in calcium research. Academic Press, London
Thomas MV (1984) Voltage clamp analysis of a calcium-mediated potassium conductance in cockroach (Periplaneta americana) central neurones. J Physiol 350:159–178
Weckström M, Kouvalainen E, Djupsund K, Järvilehto M (1989) More than one type of conductance is activated during responses of blowfly monopolar neurones. J Exp Biol. 144:147–154
Wilson W, Goldner MM (1975) Voltage clamping with a single microelectrode. J Neurobiol 6:411–422
Yamamoto D, Pinnock RD, Sattelle DB (1989) Switching between two types of bursting activity of single Ca++ dependent K+ channels in dissociated neurones. J Neuroendocrinol 1:89–94
Zagotta WN, Brainard MS, Aldrich RW (1988) Single-channel analysis of four distinct classes of potassium channels in Drosophila muscle. J Neurosci 8:4765–4779
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hardie, R.C., Weckström, M. Three classes of potassium channels in large monopolar cells of the blowfly Calliphora vicina . J Comp Physiol A 167, 723–736 (1990). https://doi.org/10.1007/BF00189763
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00189763