Abstract
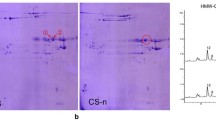
In this study, we successfully expressed the active 1Ay subunit of Triticum urartu in barley by Agrobacterium-mediated transformation with a transformation efficiency of 19.9%. The results of SDS–PAGE revealed that the expressed proteins of 1Ay subunit were present at some grains of each of 46 original T0 plants, showing identical mobility to those of positive standards of T. urartu grain protein and bacteria expressional proteins. In the T2 generation, three homozygous lines, 2–28, 3–11, and 5–6, were identified. The results of scanning electron microscopy showed an increased amount of protein bodies in these transgenic lines. The main effects in the expression of the 1Ay subunits was a considerable increase in the glutenin content, but a decrease in the contents of gliadins while there were no effects in the contents of albumin, globulin and the total protein. We found that the gluten could not be washed out from the flour obtained from transgenic barley lines when using a Gluten index analyzer and a Farinograph indicating that the transgenic barley lines could not form dough. The lack of x-type HMW-GS and the reduction in number of subunit were inferred as the possible reasons for the inability to form gluten polymer.




Similar content being viewed by others
References
Bartlett JG, Alves SC, Smedley M, Snape JW, Harwood WA (2008) High-throughput Agrobacterium-mediated barley transformation. Plant Methods 4:22
Blake TK, Ullrich SE, Nilan RA (1982) Mapping of the Hor-3 locus encoding D hordein in barley. Theor Appl Genet 63:367–371
Chen J, Liu C, Shi B, Chai Y, Han N, Zhu M, Bian H (2017) Overexpression of HvHGGT enhances tocotrienol levels and antioxidant activity in barley. J Agric Food Chem 65:5181–5187
Cheng M, Fry JE, Pang S, Zhou H, Hironaka CM, Duncan DR, Conner TW, Wan Y (1997) Genetic transformation of wheat mediated by Agrobacterium tumefaciens. Plant Physiol 115:971
Éva C, Téglás F, Zelenyánszki H, Tamás C, Juhász A, Mészáros K, Tamás L (2018) Cold inducible promoter driven Cre-lox system proved to be highly efficient for marker gene excision in transgenic barley. J Biotechnol 265:15–24
Ferdous J, Whitford R, Nguyen M, Brien C, Langridge P, Tricker P (2017) Drought-inducible expression of Hv-miR827 enhances drought tolerance in transgenic barley. Funct Integr Genom 17:279–292
Gilhumanes J, Pistón F, Shewry PR, Tosi P, Barro F (2011) Suppression of gliadins results in altered protein body morphology in wheat. J Exp Bot 62:4203
Gu YQ, Anderson OD, Londeorë CF, Kong X, Chibbar RN, Lazo GR (2003) Structural organization of the barley D-hordein locus in comparison with its orthologous regions of wheat genomes. Genome 46:1084–1097
Halford NG, Field JM, Blair H, Urwin P, Moore K, Robert L, Thompson R, Flavell RB, Tatham AS, Shewry PR (1992) Analysis of HMW glutenin subunits encoded by chromosome 1A of bread wheat (Triticum aestivum L.) indicates quantitative effects on grain quality. Theor Appl Genet 83:373–378
Harwood W (2014) A protocol for high-throughput Agrobacterium-mediated barley transformation. Methods Mol Biol 1099:251–260
Hisano H, Sato K (2016) Genomic regions responsible for amenability to Agrobacterium-mediated transformation in barley. Sci Rep 6:37505
Hu T, Metz S, Chay C, Zhou HP, Biest N, Chen G, Cheng M, Feng X, Radionenko M, Lu F (2003) Agrobacterium-mediated large-scale transformation of wheat (Triticum aestivum L.) using glyphosate selection. Plant Cell Rep 21:1010
Jiang Q, Wei Y, Wang F, Wang J, Yan Z, Zheng Y (2009) Characterization and comparative analysis of HMW glutenin 1Ay alleles with differential expressions. BMC Plant Biol 9:16
Jiang Q, Ma J, Wei Y, Liu Y, Zheng Y (2012) Novel variants of HMW glutenin subunits from Aegilops section Sitopsis species in relation to evolution and wheat breeding. BMC Plant Biol 12:73
Jones HD (2005) Wheat transformation: current technology and applications to grain development and composition. J Cereal Sci 41:137–147
Komari T, Hiei Y, Saito Y, Murai N, Kumashiro T (1996) Vectors carrying two separate T-DNAs for co-transformation of higher plants mediated by Agrobacterium tumefaciens and segregation of transformants free from selection markers. Plant J 10:165–174
Lafiandra D, D’Ovidio R, Porceddu E, Margiotta B, Colaprico G (1993) New data supporting high Mr glutenin subunit 5 as the determinant of quality differences among the pairs 5 + 10 vs. 2 + 12. J Cereal Sci 18:197–205
Lamacchia C, Shewry PR, Fonzo ND, Forsyth JL, Harris N, Lazzeri PA, Napier JA, Halford NG, Barcelo P (2001) Endosperm-specific activity of a storage protein gene promoter in transgenic wheat seed. J Exp Bot 52:243–250
Lawrenson T, Shorinola O, Stacey N, Li C, Østergaard L, Patron N, Uauy C, Harwood W (2015) Induction of targeted, heritable mutations in barley and Brassica oleracea using RNA-guided Cas9 nuclease. Genome Biol 16:258
Mackie AM, Lagudah ES, Sharp PJ, Lafiandra D (1996) Molecular and biochemical characterisation of HMW glutenin subunits from T. tauschii and the D genome of hexaploid wheat. J Cereal Sci 23:213–225
Mrizova K, Jiskrova E, Vyroubalova S, Novak O, Ohnoutkova L, Pospisilova H, Frebort I, Harwood W, Galuszka P (2013) Overexpression of cytokinin dehydrogenase genes in barley (Hordeum vulgare cv. Golden Promise) fundamentally affects morphology and fertility. PLoS ONE 8:e79029
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Osborne TB (1907) The proteins of the wheat kernel. Carnegie Institution of Washington, Washington
Payne PI (1983) Catalogue of alleles for the complex gene loci, GluA1, Glu-B1 and Glu-D1 which code for high-molecular-weight subunits of glutenin in hexaploid wheat. Cereal Res Commun 11:29–35
Payne PI (1987) Genetics of wheat storage proteins and the effect of allelic variation on bread-making quality. Annu Rev Plant Biol 38:141–153
Payne P, Corfield K, Blackman J (1979) Identification of a high-molecular-weight subunit of glutenin whose presence correlates with bread-making quality in wheats of related pedigree. Theor Appl Genet 55:153–159
Payne PI, Corfield KG, Holt LM, Blackman JA (1981) Correlations between the inheritance of certain high molecular weight subunits of glutenin and breadmaking quality in progencies of six crosses of bread wheat. J Sci Food Agric 32:51–60
Payne PI, Nightingale MA, Krattiger AF, Holt LM (2010) The relationship between HMW glutenin subunit composition and the bread-making quality of British-grown wheat varieties. J Sci Food Agric 40:51–65
Piston F, Shewry PR, Barro F (2007) D hordeins of Hordeum chilense: a novel source of variation for improvement of wheat. Theor Appl Genet 115:77–86
Sallaud C, Meynard D, Van BJ, Gay C, Bès M, Brizard JP, Larmande P, Ortega D, Raynal M, Portefaix M (2003) Highly efficient production and characterization of T-DNA plants for rice (Oryza sativa L.) functional genomics. Theor Appl Genet 106:1396–1408
Santamaria M, Diaz-Mendoza M, Perez-Herguedas D, Hensel G, Kumlehn J, Diaz I, Martinez M (2018) Overexpression of HvIcy6 in barley enhances resistance against Tetranychus urticae and entails partial transcriptomic reprogramming. Int J Mol Sci 19:697
Shewry P, Halford N (2002) Cereal seed storage proteins: structures, properties and role in grain utilization. J Exp Bot 53:947–958
Shewry PR, Jones HD (2005) Transgenic wheat: where do we stand after the first 12 years? Ann Appl Biol 147:1–14
Shewry PR, Halford NG, Tatham AS (1992) High molecular weight subunits of wheat glutenin. J Cereal Sci 15:105–120
Shewry PR, Tatham AS, Barro F, Barcelo P, Lazzeri P (1995) Biotechnology of breadmaking: unraveling and manipulating the multi-protein gluten complex. Bio/Technology 13:1185
Shewry PR, Halford NG, Lafiandra D (2003a) Genetics of wheat gluten proteins. Adv Genet 49:111–184
Shewry PR, Halford NG, Tatham AS, Popineau Y, Lafiandra D, Belton PS (2003b) The high molecular weight subunits of wheat glutenin and their role in determining wheat processing properties. Adv Food Nutr Res 45:219–302
Shewry PR, Halford NG, Tatham AS, Popineau Y, Lafiandra D, Belton PS (2003c) The high molecular weight subunits of wheat glutenin and their role in determining wheat processing properties. Adv Food Nutr Res 45:219–302
Shibata D, Liu YG (2000) Technical focus-Agrobacterium-mediated plant transformation with large DNA fragments. Trends Plant Sci 5:354–357
Shou H, Frame BR, Whitham SA, Kan W (2004) Assessment of transgenic maize events produced by particle bombardment or Agrobacterium-mediated transformation. Mol Breed 13:201–208
Tang Y-L, Yang W-Y, Tian J-C, Chen F (2008) Effect of HMW-GS 6 + 8 and 1.5 + 10 from synthetic hexaploid wheat on wheat quality traits. Agric Sci China 7:1161–1171
Travella S, Ross S, Harden J, Everett C, Snape J, Harwood W (2005) A comparison of transgenic barley lines produced by particle bombardment and Agrobacterium-mediated techniques. Plant Cell Rep 23:780–789
Waines JG, Payne PI (1987) Electrophoretic analysis of the high-molecular-weight glutenin subunits of Triticum monococcum, T. urartu, and the A genome of bread wheat (T. aestivum). Theor Appl Genet 74:71–76
Wan Y, Wang D, Shewry P, Halford N (2002) Isolation and characterization of five novel high molecular weight subunit of glutenin genes from Triticum timopheevi and Aegilops cylindrica. Theor Appl Genet 104:828–839
Wieser H (2007) Chemistry of gluten proteins. Food Microbiol 24:115
Xia L, Geng H, Chen X, He Z, Lillemo M, Morris CF (2008) Silencing of puroindoline a alters the kernel texture in transgenic bread wheat. J Cereal Sci 47:331–338
Yang Q, Deng M, Zhang LL, Zhang XW, Wang LN, Chen H, Ma J, Qi PF, Jiang QT, Lan XJ (2016) A super twin T-DNA vector that allows independent gene expression during Agrobacterium-mediated transformation. Plasmid 87–88:58–64
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2016YFD0100100), the Key Research and Development Program of Sichuan Province (2018NZDZX0002), the International Science and Technology Cooperation project of Sichuan Province, China (2016HH0057).
Author information
Authors and Affiliations
Contributions
YQ carried out the experiments and wrote the draft; LSY did the barley transformation; LXY and MJ finished the SDS–PAGE detection; WJR and QPF designed the plant expression vector; CGY, PZE and LW extracted DNA for the experiment and finished the PCR detection; Wendy Harwood guided the barley transformation; LZY conducted data analysis; DM did the qRT-PCR; LXJ and LZX contributed to Southern blotting; WYM reviewed the manuscript; ZYL contributed to the improvement of the research program; JQT revised the manuscript and conducted data analysis. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig.
1. Barley transformation. a The immature grains for embryo isolation. b Isolated immature embryo. c Induction of embryogenic callus. d Regeneration of transgenic shoots. e Rooting of transgenic plants. f A T0 plant in soil. g Transgenic barley growing in greenhouse. h Gus staining of transgenic lines (right) compared to non-transgenic controls (left). (TIFF 879 kb)
Supplementary Fig.
2. PCR analysis of the transgenic barley T0 plants. Lanes 1 to 9 represent some of the transgenic barley lines; the CK- indicates the genomic DNA of non-transgenic barley ‘Golden promise’, and the CK + indicates the plasmid DNA of pCAMBIA1302-Glu-D1-1Ay. (TIFF 213 kb)
Supplementary Fig.
3. Scanning electron microscopy images of dried barley grains. a donor barley ‘Golden promise’; b transgenic barley line 2-28; c transgenic barley line 3-11; d transgenic barley line 5-6. Yellow arrows: large starch granule A type. Blue arrows: B type starch granule. Red arrows: protein matrices. Bars indicating 20 um are shown on each micrograph. (TIFF 758 kb)
Rights and permissions
About this article
Cite this article
Yang, Q., Li, S., Li, X. et al. Expression of the high molecular weight glutenin 1Ay gene from Triticum urartu in barley. Transgenic Res 28, 225–235 (2019). https://doi.org/10.1007/s11248-019-00117-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-019-00117-6