Abstract
Laccase-based biocatalysts are promising agents for the remediation of environmental pollutants. However, stability, reusability, and cost-effectiveness remain a challenge for biochemical engineering. Herein, Trametes versicolor IBL-04 laccase was immobilized on poly(vinyl alcohol) (PVA)-alginate beads. The maximum immobilization efficiency was observed using a 2.5 mm bead size with 12% (w/v) PVA and 2% (w/v) alginate concentration. The native enzyme displayed optimal activity at pH 5.0, and 45 °C, which was improved to pH 6 and 70 °C in PVA-alginate immobilized biocatalyst. PVA-alginate immobilized laccase presented comparatively high Vmax (from 720 to 962 U/mL) and low Km (from 70 to 12 µM) values from the native enzyme. Immobilized laccase was applied to catalyze the decolorization of various textile dye pollutants, including Rhodamine B, Reactive Black 5 (RB5), Drimaren Red and Drimaren Turquoise. Results revealed almost complete decolorization and decomposition of all dye’s solution by the laccase-assisted biocatalytic reaction. Prominent peaks of untreated dyes observed in the visible region disappeared following enzymatic treatment, and the appearance of any new peaks was not detected during the enzyme-mediated degradation process. In conclusion, immobilizing laccase on PVA-alginate matrix offers a facile and cost-efficient approach to remediate dyes and exhibit considerable potential for numerous biochemical engineering applications.
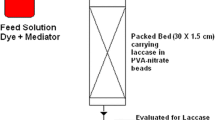
Graphic Abstract







Similar content being viewed by others
References
Asgher M, Noreen S, Bilal M (2017) Enhancement of catalytic, reusability, and long-term stability features of Trametes versicolor IBL-04 laccase immobilized on different polymers. Int J Biol Macromol 95:54–62
Singh AK, Bilal M, Iqbal HM, Raj A (2021) Lignin peroxidase in focus for catalytic elimination of contaminants—A critical review on recent progress and perspectives. Int J Biol Macromol 179:58–82
Noreen S, Asgher M, Hussain F, Iqbal A (2016) Performance improvement of Ca-alginate bead cross-linked laccase from Trametes versicolor IBL-04. BioResources 11(1):558–572
Olajuyigbe FM, Adetuyi OY, Fatokun CO (2019) Characterization of free and immobilized laccase from Cyberlindnera fabianii and application in degradation of bisphenol A. Int J Biol Macromol 125:856–864
Singh AK, Bilal M, Iqbal HM, Meyer AS, Raj A (2021) Bioremediation of lignin derivatives and phenolics in wastewater with lignin modifying enzymes: Status, opportunities and challenges. Sci Total Env 777:145988
Guan ZB, Shui Y, Song CM, Zhang N, Cai YJ, Liao XR (2015) Efficient secretory production of CotA-laccase and its application in the decolorization and detoxification of industrial textile wastewater. Environ Sci Pollut Res 22(12):9515–9523
Mojtabavi S, Khoshayand MR, Fazeli MR, Samadi N, Faramarzi MA (2020) Combination of thermal and biological treatments for bio-removal and detoxification of some recalcitrant synthetic dyes by betaine-induced thermostabilized laccase. Environ Technol Innov 20:10104
Boruah P, Sarmah P, Das PK, Goswami T (2019) Exploring the lignolytic potential of a new laccase producing strain Kocuria sp PBS-1 and its application in bamboo pulp bleaching. Int Biodeter Biodegradation 143:104726
Bagewadi ZK, Mulla SI, Ninnekar HZ (2017) Purification and immobilization of laccase from Trichoderma harzianum strain HZN10 and its application in dye decolorization. J Gene Eng Biotechnol 15(1):139–150
Bilal M, Ashraf SS, Iqbal HM (2020) Laccase-mediated bioremediation of dye-based hazardous pollutants. In: Inamuddin MI, Ahamed EL, Asiri AM (eds) Methods for Bioremediation of Water and Wastewater Pollution. Springer, Cham, pp 137–160
Bilal M, Rasheed T, Nabeel F, Iqbal HM, Zhao Y (2019) Hazardous contaminants in the environment and their laccase-assisted degradation–a review. J Environ Manage 234:253–264
Ji C, Nguyen LN, Hou J, Hai FI, Chen V (2017) Direct immobilization of laccase on titania nanoparticles from crude enzyme extracts of P. ostreatus culture for micro-pollutant degradation. Sep Purif Technol 178:215–223
Stevens JC, Shi J (2019) Biocatalysis in ionic liquids for lignin valorization: Opportunities and recent developments. Biotechnol Adv 37(8):107418
Narnoliya LK, Agarwal N, Patel SN, Singh SP (2019) Kinetic characterization of laccase from Bacillus atrophaeus, and its potential in juice clarification in free and immobilized forms. J Microbiol 57(10):900–909
Wang F, Owusu-Fordjour M, Xu L, Ding Z, Gu Z (2020) Immobilization of laccase on magnetic chelator nanoparticles for apple juice clarification in magnetically stabilized fluidized bed. Front Bioeng Biotechnol 8:589
Mogharabi M, Faramarzi MA (2014) Laccase and laccase-mediated systems in the synthesis of organic compounds. Adv Synth Catal 356(5):897–927
Kudanga T, Nemadziva B, Le Roes-Hill M (2017) Laccase catalysis for the synthesis of bioactive compounds. Appl Microbiol Biotechnol 101(1):13–33
Hanefeld U, Gardossi L, Magner E (2009) Understanding enzyme immobilisation. Chem Soc Rev 38(2):453–468
Al-Maqdi KA, Bilal M, Alzamly A, Iqbal H, Shah I, Ashraf SS (2021) Enzyme-loaded flower-shaped nanomaterials: a versatile platform with biosensing, biocatalytic, and environmental promise. Nanomaterials 11(6):1460
Qamar SA, Qamar M, Bilal M, Bharagava RN, Ferreira LFR, Sher F, Iqbal HM (2021) Cellulose-deconstruction potential of nano-biocatalytic systems: A strategic drive from designing to sustainable applications of immobilized cellulases. Int J Biol Macromol 185:1–19
Morsi R, Al-Maqdi KA, Bilal M, Iqbal H, Khaleel A, Shah I, Ashraf SS (2021) Immobilized soybean peroxidase hybrid biocatalysts for efficient degradation of various emerging pollutants. Biomolecules 11(6):904
Sheldon RA (2016) Engineering a more sustainable world through catalysis and green chemistry. J R Soc Interface 13(116):20160087
Almulaiky, Y. Q., & Al-Harbi, S. A. (2021). Preparation of a calcium alginate-coated polypyrrole/silver nanocomposite for site-specific immobilization of polygalacturonase with high reusability and enhanced stability. Catalysis Letters, 1–15.
Sheldon RA (2007) Enzyme immobilization: the quest for optimum performance. Adv Synth Catal 349(8–9):1289–1307
Gan, J., Bagheri, A. R., Aramesh, N., Gul, I., Franco, M., Almulaiky, Y. Q., & Bilal, M. (2020). Covalent organic frameworks as emerging host platforms for enzyme immobilization and robust biocatalysis–A review. International Journal of Biological Macromolecules.
Weiser D, Nagy F, Bánóczi G, Oláh M, Farkas A, Szilágyi A, Poppe L (2017) Immobilization engineering–How to design advanced sol–gel systems for biocatalysis? Green Chem 19(16):3927–3937
Bilal M, Iqbal HM (2019) Naturally-derived biopolymers: Potential platforms for enzyme immobilization. Int J Biol Macromol 130:462–482
Bilal M, Qamar SA, Ashraf SS, Rodríguez-Couto S, Iqbal HM (2021) Robust nanocarriers to engineer nanobiocatalysts for bioprocessing applications. Adv Coll Interface Sci 293:102438
Almulaiky YQ, Al-Harbi SA (2019) A novel peroxidase from Arabian balsam (Commiphora gileadensis) stems: Its purification, characterization and immobilization on a carboxymethylcellulose/Fe3O4 magnetic hybrid material. Int J Biol Macromol 133:767–774
Al-Harbi SA, Almulaiky YQ (2020) Purification and biochemical characterization of Arabian balsam α-amylase and enhancing the retention and reusability via encapsulation onto calcium alginate/Fe2O3 nanocomposite beads. Int J Biol Macromol 160:944–952
Piao M, Zou D, Yang Y, Ren X, Qin C, Piao Y (2019) Multi-functional laccase immobilized hydrogel microparticles for efficient removal of bisphenol A. Materials 12(5):704
Qamar SA, Asgher M, Bilal M (2020) Immobilization of alkaline protease from Bacillus brevis using Ca-alginate entrapment strategy for improved catalytic stability, silver recovery, and dehairing potentialities. Catal Lett 150:3572–3583
Bilal M, Asgher M (2015) Sandal reactive dyes decolorization and cytotoxicity reduction using manganese peroxidase immobilized onto polyvinyl alcohol-alginate beads. Chem Cent J 9(1):1–14
Arca-Ramos A, Ammann EM, Gasser CA, Nastold P, Eibes G, Feijoo G, Corvini PX (2016) Assessing the use of nanoimmobilized laccases to remove micropollutants from wastewater. Environ Sci Pollut Res 23(4):3217–3228
Zhang Y, Ge J, Liu Z (2015) Enhanced activity of immobilized or chemically modified enzymes. ACS Catal 5(8):4503–4513
Yinghui D, Qiuling W, Shiyu F (2002) Laccase stabilization by covalent binding immobilization on activated polyvinyl alcohol carrier. Lett Appl Microbiol 35(6):451–456
Zhou, W., Zhang, W., & Cai, Y. (2020). Laccase immobilization for water purification: A comprehensive review. Chemical Engineering Journal, 126272.
Tien M, Kirk TK (1988) Lignin peroxidase of Phanerochaete chrysosporium. Methods Enzymol 161:238–249
Kay-Shoemake JL, Watwood ME (1996) Limitations of the lignin peroxidase system of the white-rot fungus. Phanerochaete chrysosporium Appl Microbiol Biotechnol 46(4):438–442
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Wolfenden BS, Willson RL (1982) Radical-cations as reference chromogens in kinetic studies of ono-electron transfer reactions: pulse radiolysis studies of 2, 2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). J Chem Soc Perkin Trans 2(7):805–812
Zhan JF, Jiang ST, Pan LJ (2013) Immobilization of phospholipase A1 using a polyvinyl alcohol-alginate matrix and evaluation of the effects of immobilization. Braz J Chem Eng 30(4):721–728
Idris A, Zain NAM, Suhaimi MS (2008) Immobilization of Baker’s yeast invertase in PVA–alginate matrix using innovative immobilization technique. Process Biochem 43(4):331–338
Nunes MA, Vila-Real H, Fernandes PC, Ribeiro MH (2010) Immobilization of naringinase in PVA–alginate matrix using an innovative technique. Appl Biochem Biotechnol 160(7):2129–2147
Takei T, Ikeda K, Ijima H, Kawakami K (2011) Fabrication of poly (vinyl alcohol) hydrogel beads crosslinked using sodium sulfate for microorganism immobilization. Process Biochem 46(2):566–571
Qin X, Wang J, Zheng G (2010) Enantioselective resolution of γ-lactam by a whole cell of microbacterium hydrocarbonoxydans (L29–9) immobilized in polymer of PVA–alginate–boric acid. Appl Biochem Biotechnol 162(8):2345–2354
Embaby, Ahmed S., ""An Investigation into the Potential Of Immobilized Nitrifies In Wastewater Treatment"" (2005). Theses. 448. https://scholarworks.uaeu.ac.ae/all_theses/448.
Grosová Z, Rosenberg M, Gdovin M, Sláviková L, Rebroš M (2009) Production of D-galactose using β-galactosidase and Saccharomyces cerevisiae entrapped in poly (vinylalcohol) hydrogel. Food Chem 116(1):96–100
Stanescu MD, Fogorasi M, Shaskolskiy BL, Gavrilas S, Lozinsky VI (2010) New potential biocatalysts by laccase immobilization in PVA cryogel type carrier. Appl Biochem Biotechnol 160(7):1947–1954
Freitas FF, Marquez LDS, Ribeiro GP, Brandão GC, Cardoso VL, Ribeiro EJ (2012) Optimization of the immobilization process of β-galatosidade by combined entrapment-cross-linking and the kinetics of lactose hydrolysis. Braz J Chem Eng 29(1):15–24
Vujčić Z, Miloradović Z, Milovanović A, Božić N (2011) Cell wall invertase immobilisation within gelatin gel. Food Chem 126(1):236–240
Upadhyay LSB, Verma N (2014) Highly efficient production of inverted syrup in an analytical column with immobilized invertase. J Food Sci Technol 51(12):4120–4125
El-Tanash AB, Sherief AA, Nour A (2011) Catalytic properties of immobilized tannase produced from Aspergillus aculeatus compared with the free enzyme. Braz J Chem Eng 28(3):381–391
Alaoui MS, Ghanam J, Merzouki M, Penninckx MJ, Benlemlih M (2013) Immobilisation of Pycnoporus coccineus laccase in ca alginate beads for use in the degradation of aromatic compounds present in olive oil mill wastewaters. J Biotechnol Lett 4(2):91
Kharkrang, K., & Ambasht, P. K. (2013). Immobilization of?-amylase from pennisetum typhoides inside gelatin beads and its characterization. Journal of Proteins & Proteomics, 4(3).
Nicolucci C, Rossi S, Menale C, Godjevargova T, Ivanov Y, Bianco M, Diano N (2011) Biodegradation of bisphenols with immobilized laccase or tyrosinase on polyacrylonitrile beads. Biodegradation 22(3):673–683
Srivastava PK, Anand A (2014) Immobilization of acid phosphatase from Vigna aconitifolia seeds on chitosan beads and its characterization. Int J Biol Macromol 64:150–154
De Queiroz AA, Passos ED, De Brito Alves S, Silva GS, Higa OZ, Vítolo M (2006) Alginate–poly (vinyl alcohol) core–shell microspheres for lipase immobilization. J Appl Polym Sci 102(2):1553–1560
Asgher M, Shahid M, Kamal S, Iqbal HMN (2014) Recent trends and valorization of immobilization strategies and ligninolytic enzymes by industrial biotechnology. J Mol Catal B Enzym 101:56–66
Tanriseven A, Doğan Ş (2001) Immobilization of invertase within calcium alginate gel capsules. Process Biochem 36(11):1081–1083
Emregul E, Sungur S, Akbulut U (2006) Polyacrylamide–gelatine carrier system used for invertase immobilization. Food Chem 97(4):591–597
Zheng F, Cui BK, Wu XJ, Meng G, Liu HX, Si J (2016) Immobilization of laccase onto chitosan beads to enhance its capability to degrade synthetic dyes. Int Biodeterior Biodegradation 110:69–78
Ma HF, Meng G, Cui BK, Si J, Dai YC (2018) Chitosan crosslinked with genipin as supporting matrix for biodegradation of synthetic dyes: Laccase immobilization and characterization. Chem Eng Res Des 132:664–676
Peng J, Wu E, Lou X, Deng Q, Hou X, Lv C, Hu Q (2021) Anthraquinone removal by a metal-organic framework/polyvinyl alcohol cryogel-immobilized laccase: Effect and mechanism exploration. Chem Eng J 418:129473
Mogharabi, M., Nassiri-Koopaei, N., Bozorgi-Koushalshahi, M., Nafissi-Varcheh, N., Bagherzadeh, G., & Faramarzi, M. A. (2012). Immobilization of laccase in alginate-gelatin mixed gel and decolorization of synthetic dyes. Bioinorganic Chemistry and Applications, 2012.
Uygun M (2013) Preparation of laccase immobilized cryogels and usage for decolorization. J Chem 2013:1–7
Kunamneni A, Ghazi I, Camarero S, Ballesteros A, Plou FJ, Alcalde M (2008) Decolorization of synthetic dyes by laccase immobilized on epoxy-activated carriers. Process Biochem 43(2):169–178
Tallur PN, Megadi VB, Ninnekar HZ (2009) Biodegradation of p-cresol by immobilized cells of Bacillus sp. strain PHN 1. Biodegradation 20(1):79–83
Asgher M, Kamal S, Iqbal HMN (2012) Improvement of catalytic efficiency, thermo-stability and dye decolorization capability of Pleurotus ostreatus IBL-02 laccase by hydrophobic sol gel entrapment. Chem Cent J 6(1):1–10
Campos R, Kandelbauer A, Robra KH, Cavaco-Paulo A, Gübitz GM (2001) Indigo degradation with purified laccases from Trametes hirsuta and Sclerotium rolfsii. J Biotechnol 89(2–3):131–139
Soares GM, de Amorim MP, Costa-Ferreira M (2001) Use of laccase together with redox mediators to decolourize Remazol Brilliant Blue R. J Biotechnol 89(2–3):123–129
Claus H, Faber G, König HJAM (2002) Redox-mediated decolorization of synthetic dyes by fungal laccases. Appl Microbiol Biotechnol 59(6):672–678
Daassi D, Rodríguez-Couto S, Nasri M, Mechichi T (2014) Biodegradation of textile dyes by immobilized laccase from Coriolopsis gallica into Ca-alginate beads. Int Biodeterior Biodegradation 90:71–78
Bilal M, Asgher M, Shahid M, Bhatti HN (2016) Characteristic features and dye degrading capability of agar-agar gel immobilized manganese peroxidase. Int J Biol Macromol 86:728–740
Bilal M, Jing Z, Zhao Y, Iqbal HM (2019) Immobilization of fungal laccase on glutaraldehyde cross-linked chitosan beads and its bio-catalytic potential to degrade bisphenol A. Biocatal Agri Biotechnol 19:101174
Asgher M, Wahab A, Bilal M, Iqbal HM (2018) Delignification of lignocellulose biomasses by alginate–chitosan immobilized laccase produced from Trametes versicolor IBL-04. Waste and Biomass Valorization 9(11):2071–2079
Acknowledgements
Financial support from Higher Education Commission (HEC), Islamabad, Pakistan is gratefully acknowledged. Consejo Nacional de Ciencia y Tecnología (CONACYT) is thankfully acknowledged for partially supporting this work under Sistema Nacional de Investigadores (SNI) program awarded to Hafiz M.N. Iqbal (CVU: 735,340).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Noreen, S., Asgher, M., Qamar, S.A. et al. Poly(vinyl Alcohol)-Alginate Immobilized Trametes versicolor IBL-04 Laccase as Eco-friendly Biocatalyst for Dyes Degradation. Catal Lett 152, 1869–1879 (2022). https://doi.org/10.1007/s10562-021-03778-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-021-03778-3