Abstract
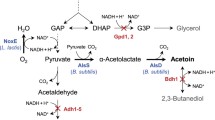
Acetaldehyde is synthesized by yeast during the main fermentation period of beer production, which causes an unpleasant off-flavor. Therefore, there has been extensive effort toward reducing acetaldehyde to obtain a beer product with better flavor and anti-staling ability. In this study, we discovered that acetaldehyde production in beer brewing is closely related with the intracellular NADH equivalent regulated by the citric acid cycle. However, there was no significant relationship between acetaldehyde production and amino acid metabolism. A reverse engineering strategy to increase the intracellular NADH/NAD+ ratio reduced the final acetaldehyde production level, and vice versa. This work offers new insight into acetaldehyde metabolism and further provides efficient strategies for reducing acetaldehyde production by the regulating the intracellular NADH/NAD+ ratio through cofactor engineering.






Similar content being viewed by others
References
Albers E, Larsson C, Liden G, Niklasson C, Gustafsson L (1996) Influence of the nitrogen source on Saccharomyces cerevisiae anaerobic growth and product formation. Appl Environ Microbiol 62(9):3187–3195
Anders S, Pyl PT, Huber W (2015) HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31(2):166–169. https://doi.org/10.1093/bioinformatics/btu638
Aranda A, del Olmo M (2003) Response to acetaldehyde stress in the yeast Saccharomyces cerevisiae involves a strain-dependent regulation of several ALD genes and is mediated by the general stress response pathway. Yeast 20(8):747–759. https://doi.org/10.1002/yea.991
Bakker BM, Bro C, Kotter P, Luttik MA, van Dijken JP, Pronk JT (2000) The mitochondrial alcohol dehydrogenase Adh3p is involved in a redox shuttle in Saccharomyces cerevisiae. J Bacteriol 182(17):4730–4737
Bakker BM, Overkamp KM, van Maris AJ, Kotter P, Luttik MA, van Dijken JP, Pronk JT (2001) Stoichiometry and compartmentation of NADH metabolism in Saccharomyces cerevisiae. FEMS Microbiol Rev 25(1):15–37
Balagurunathan B, Tan L, Zhao H (2018) Metabolic engineering of Escherichia coli for acetaldehyde overproduction using pyruvate decarboxylase from Zymomonas mobilis. Enzym Microb Technol 109:58–65. https://doi.org/10.1016/j.enzmictec.2017.09.012
Bisson LF, Fraenkel DG (1983) Involvement of kinases in glucose and fructose uptake by Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 80(6):1730–1734
Blieck L, Toye G, Dumortier F, Verstrepen KJ, Delvaux FR, Thevelein JM, Van Dijck P (2007) Isolation and characterization of brewer’s yeast variants with improved fermentation performance under high-gravity conditions. Appl Environ Microbiol 73(3):815–824. https://doi.org/10.1128/aem.02109-06
Byrne KP, Wolfe KH (2005) The Yeast Gene Order Browser: combining curated homology and syntenic context reveals gene fate in polyploid species. Genome Res 15(10):1456–1461. https://doi.org/10.1101/gr.3672305
Camarasa C, Grivet JP, Dequin S (2003) Investigation by 13C-NMR and tricarboxylic acid (TCA) deletion mutant analysis of pathways for succinate formation in Saccharomyces cerevisiae during anaerobic fermentation. Microbiology 149(Pt 9):2669–2678. https://doi.org/10.1099/mic.0.26007-0
Camarasa C, Faucet V, Dequin S (2007) Role in anaerobiosis of the isoenzymes for Saccharomyces cerevisiae fumarate reductase encoded by OSM1 and FRDS1. Yeast 24(5):391–401. https://doi.org/10.1002/yea.1467
Celton M, Goelzer A, Camarasa C, Fromion V, Dequin S (2012) A constraint-based model analysis of the metabolic consequences of increased NADPH oxidation in Saccharomyces cerevisiae. Metab Eng 14(4):366–379. https://doi.org/10.1016/j.ymben.2012.03.008
Chen X, Li S, Liu (2014) Engineering redox balance through cofactor systems. Trends Biotechnol 32(6):337–343
Cheraiti N, Guezenec S, Salmon JM (2005) Redox interactions between Saccharomyces cerevisiae and Saccharomyces uvarum in mixed culture under enological conditions. Appl Environ Microbiol 71(1):255–260
Cheraiti N, Sauvage FX, Salmon JM (2008) Acetaldehyde addition throughout the growth phase alleviates the phenotypic effect of zinc deficiency in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 77(5):1093–1109. https://doi.org/10.1007/s00253-007-1234-z
de Winde H (2003) Functional genetics of industrial yeasts; of ancient skills and modern applications. In: de Winde JH (ed) Functional genetics of industrial yeasts, Topics in Current Genetics, vol 2. Springer, Berlin, pp 1–16
Duong CT, Strack L, Futschik M, Katou Y, Nakao Y, Fujimura T, Shirahige K, Kodama Y, Nevoigt E (2011) Identification of Sc-type ILV6 as a target to reduce diacetyl formation in lager brewers' yeast. Metab Eng 13(6):638–647. https://doi.org/10.1016/j.ymben.2011.07.005
Dzialo MC, Park R, Steensels J, Lievens B, Verstrepen KJ (2017) Physiology, ecology and industrial applications of aroma formation in yeast. FEMS Microbiol Rev 41(Supp_1):S95–s128. https://doi.org/10.1093/femsre/fux031
Ehsani M, Fernández MR, Biosca JA, Julien A, Dequin S (2009) Engineering of 2,3-butanediol dehydrogenase to reduce acetoin formation by glycerol-overproducing, low-alcohol Saccharomyces cerevisiae. Appl Environ Microbiol 75(10):3196–3205
Heinrikson RL, Meredith SC (1984) Amino acid analysis by reverse-phase high-performance liquid chromatography: Precolumn derivatization with phenylisothiocyanate. Anal Biochem 136(1):65–74. https://doi.org/10.1016/0003-2697(84)90307-5
Heux S, Cachon R, Dequin S (2006) Cofactor engineering in Saccharomyces cerevisiae: expression of a H2O-forming NADH oxidase and impact on redox metabolism. Metab Eng 8(4):303–314
Hill JE, Myers AM, Koerner TJ, Tzagoloff A (1986) Yeast/E. coli shuttle vectors with multiple unique restriction sites. Yeast 2(3):163–167. https://doi.org/10.1002/yea.320020304
Hong KK, Vongsangnak W, Vemuri GN, Nielsen J (2011) Unravelling evolutionary strategies of yeast for improving galactose utilization through integrated systems level analysis. Proc Natl Acad Sci U S A 108(29):12179–12184. https://doi.org/10.1073/pnas.1103219108
Hou J, Scalcinati G, Oldiges M, Vemuri GN (2010) Metabolic impact of increased NADH availability in Saccharomyces cerevisiae. Appl Environ Microbiol 76(3):851–859
Hubmann G, Foulquie-Moreno MR, Nevoigt E, Duitama J, Meurens N, Pais TM, Mathe L, Saerens S, Nguyen HT, Swinnen S, Verstrepen KJ, Concilio L, de Troostembergh JC, Thevelein JM (2013) Quantitative trait analysis of yeast biodiversity yields novel gene tools for metabolic engineering. Metab Eng 17:68–81. https://doi.org/10.1016/j.ymben.2013.02.006
Kim KS, Rosenkrantz MS, Guarente L (1986) Saccharomyces cerevisiae contains two functional citrate synthase genes. Mol Cell Biol 6(6):1936–1942
Kosugi S, Kiyoshi K, Oba T, Kusumoto K, Kadokura T, Nakazato A, Nakayama S (2014) Isolation of a high malic and low acetic acid-producing sake yeast Saccharomyces cerevisiae strain screened from respiratory inhibitor 2,4-dinitrophenol (DNP)-resistant strains. J Biosci Bioeng 117(1):39–44. https://doi.org/10.1016/j.jbiosc.2013.06.016
Li E, Mira de Orduña (2011) Evaluation of the acetaldehyde production and degradation potential of 26 enological Saccharomyces and non-Saccharomyces yeast strains in a resting cell model system. J Ind Microbiol Biotechnol 38(9):1391–1398. https://doi.org/10.1007/s10295-010-0924-1
Li E, Mira de Orduña (2017) Acetaldehyde kinetics of enological yeast during alcoholic fermentation in grape must. J Ind Microbiol Biotechnol 44(2):229–236. https://doi.org/10.1007/s10295-016-1879-7
Li J, Li Y, Cui Z, Liang Q, Qi Q (2017) Enhancement of succinate yield by manipulating NADH/NAD(+) ratio and ATP generation. Appl Microbiol Biotechnol 101(8):3153–3161. https://doi.org/10.1007/s00253-017-8127-6
Li F, Li Y, Sun LM, Chen X, An X, Yin C, Cao Y, Wu H, Song H (2018) Modular engineering intracellular NADH regeneration boosts extracellular electron transfer of Shewanella oneidensis MR-1. ACS Synth Biol 7(3):885–895. https://doi.org/10.1021/acssynbio.7b00390
Liu L, Li Y, Du G, Chen J (2006a) Redirection of the NADH oxidation pathway in Torulopsis glabrata leads to an enhanced pyruvate production. Appl Microbiol Biotechnol 72(2):377–385. https://doi.org/10.1007/s00253-005-0284-3
Liu L, Li Y, Shi Z, Du G, Chen J (2006b) Enhancement of pyruvate productivity in Torulopsis glabrata: increase of NAD+ availability. J Biotechnol 126(2):173–185. https://doi.org/10.1016/j.jbiotec.2006.04.014
Livak KJ, Schmittgen TD (2001) Analysis of relative rene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Nevoigt E, Pilger R, Mast-Gerlach E, Schmidt U, Freihammer S, Eschenbrenner M, Garbe L, Stahl U (2002) Genetic engineering of brewing yeast to reduce the content of ethanol in beer. FEMS Yeast Res 2(2):225–232
Pires EJ, Teixeira JA, Branyik T, Vicente AA (2014) Yeast: the soul of beer's aroma--a review of flavour-active esters and higher alcohols produced by the brewing yeast. Appl Microbiol Biotechnol 98(5):1937–1949. https://doi.org/10.1007/s00253-013-5470-0
Qin Y, Johnson CH, Liu L, Chen J (2011) Introduction of heterogeneous NADH reoxidation pathways into Torulopsis glabrata significantly increases pyruvate production efficiency. Korean J Chem Eng 28(4):1078–1084
Remize F, Andrieu E, Dequin S (2000) Engineering of the pyruvate dehydrogenase bypass in Saccharomyces cerevisiae: role of the cytosolic Mg(2+) and mitochondrial K(+) acetaldehyde dehydrogenases Ald6p and Ald4p in acetate formation during alcoholic fermentation. Appl Environ Microbiol 66(8):3151–3159
Roustan JL, Sablayrolles JM (2002) Modification of the acetaldehyde concentration during alcoholic fermentation and effects on fermentation kinetics. J Biosci Bioeng 93(4):367–375
Saerens SM, Duong CT, Nevoigt E (2010) Genetic improvement of brewer’s yeast: current state, perspectives and limits. Appl Microbiol Biotechnol 86(5):1195–1212. https://doi.org/10.1007/s00253-010-2486-6
Saison D, De Schutter DP, Uyttenhove B, Delvaux F, Delvaux FR (2009) Contribution of staling compounds to the aged flavour of lager beer by studying their flavour thresholds. Food Chem 114(4):1206–1215. https://doi.org/10.1016/j.foodchem.2008.10.078
Shi XC, Zou YN, Chen Y, Zheng C, Li BB, Xu JH, Shen XN, Ying HJ (2016) A water-forming NADH oxidase regulates metabolism in anaerobic fermentation. Biotechnol Biofuels 9:103. https://doi.org/10.1186/s13068-016-0517-y
Vuoristo KS, Mars AE, Sanders JP, Eggink G, Weusthuis RA (2015) Metabolic engineering of TCA cycle for production of chemicals. Trends Biotechnol 34(3):191–197. https://doi.org/10.1016/j.tibtech.2015.11.002
Walsh CT, Tu BP, Tang Y (2017) Eight kinetically stable but thermodynamically activated molecules that power cell metabolism. Chem Rev 118(4):1460–1494. https://doi.org/10.1021/acs.chemrev.7b00510
Wang ZY, Wang JJ, Liu XF, He XP, Zhang BR (2009) Recombinant industrial brewing yeast strains with ADH2 interruption using self-cloning GSH1+CUP1 cassette. FEMS Yeast Res 9(4):574–581. https://doi.org/10.1111/j.1567-1364.2009.00502.x
Wang Y, Halls C, Zhang J, Matsuno M, Zhang Y, Yu O (2011) Stepwise increase of resveratrol biosynthesis in yeast Saccharomyces cerevisiae by metabolic engineering. Metab Eng 13(5):455–463. https://doi.org/10.1016/j.ymben.2011.04.005
Wang J, Shen N, Yin H, Liu C, Li Y, Li Q (2013) Development of industrial brewing yeast with low acetaldehyde production and improved flavor stability. Appl Biochem Biotechnol 169(3):1016–1025
Wang P, Yang X, Lin B, Huang J, Tao Y (2017) Cofactor self-sufficient whole-cell biocatalysts for the production of 2-phenylethanol. Metab Eng 44:143–149. https://doi.org/10.1016/j.ymben.2017.09.013
Xu X, Liu C, Niu C, Wang J, Zheng F, Li Y, Li Q (2018) Rationally designed perturbation factor drives evolution in Saccharomyces cerevisiae for industrial application. J Ind Microbiol Biotechnol 45:869–880. https://doi.org/10.1007/s10295-018-2057-x
Yan D, Wang C, Zhou J, Liu Y, Yang M, Xing J (2014) Construction of reductive pathway in Saccharomyces cerevisiae for effective succinic acid fermentation at low pH value. Bioresour Technol 156:232–239. https://doi.org/10.1016/j.biortech.2014.01.053
Yoshida S, Imoto J, Minato T, Oouchi R, Sugihara M, Imai T, Ishiguro T, Mizutani S, Tomita M, Soga T, Yoshimoto H (2008) Development of bottom-fermenting Saccharomyces strains that produce high SO2 levels, using integrated metabolome and transcriptome analysis. Appl Environ Microbiol 74(9):2787–2796. https://doi.org/10.1128/aem.01781-07
Acknowledgements
This study was financially supported by the National Science Foundation (No. 31571942, No. 31771963, No. 31601558), Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), Program of Introducing Talents of Discipline to Universities (No. 111-2-06), Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX18_1790), and the Fundamental Research Funds for the Central Universities (JUSRP51306A, JUSRP51402A, JUDCF13008).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 123 kb)
Rights and permissions
About this article
Cite this article
Xu, X., Wang, J., Bao, M. et al. Reverse metabolic engineering in lager yeast: impact of the NADH/NAD+ ratio on acetaldehyde production during the brewing process. Appl Microbiol Biotechnol 103, 869–880 (2019). https://doi.org/10.1007/s00253-018-9517-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9517-0