Abstract
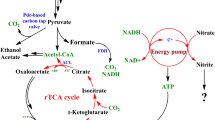
This study aimed at increasing the pyruvate productivity of a multi-vitamin auxotrophic yeast Torulopsis glabrata by redirecting NADH oxidation from adenosine triphosphate (ATP)-production pathway (oxidative phosphorylation pathway) to non-ATP production pathway (fermentative pathway). Two respiratory-deficient mutants, RD-17 and RD-18, were screened and selected after ethidium bromide (EtBr) mutagenesis of the parent strain T. glabrata CCTCC M202019. Compared with the parent strain, cytochrome aa 3 and b in electron transfer chain (ETC) of RD-18 and cytochrome b in RD-17 were disrupted. As a consequence, the activities of key ETC enzymes of the mutant RD-18, including F0F1-ATP synthase, complex I, complex I + III, complex II + III, and complex IV, decreased by 22.2, 41.6, 53.1, 23.6, and 84.7%, respectively. With the deficiency of cytochromes in ETC, a large amount of excessive cytosolic NADH was accumulated, which hampered the further increase of the glycolytic flux. An exogenous electron acceptor, acetaldehyde, was added to the strain RD-18 culture to oxidize the excessive NADH. Compared with the parent strain, the concentration of pyruvate and the glucose consumption rate of strain RD-18 were increased by 26.5 and 17.6%, respectively, upon addition of 2.1 mM of acetaldehyde. The strategy for increasing the glycolytic flux in T. glabrata by redirecting the NADH oxidation pathway may provide an alternative approach to enhance the glycolytic flux in yeast.



Similar content being viewed by others
References
Aust A, Yun SL, Suelter CH (1975) Pyruvate kinase from yeast (Saccharomyces cerevisiae). Methods Enzymol 42:176–182
Bakker BM, Bro C, Kötter P, Luttik MAH, van Dijken JP, Pronk JT (2000) The mitochondrial alcohol dehydrogenase Adh3p is involved in a redox shuttle in Saccharomyces cerevisiae. J Bacteriol 182:4730–4737
Barber A, Henningsson M, Pamment N (2002) Acceleration of high gravity yeast fermentations by acetaldehyde addition. Biotechnol Lett 24(11):891–895
Barnard EA (1975) Hexokinase from yeast. Methods Enzymol 42:6–20
Beauvoit B, Rigoulet M, Bunoust O, Raffard G, Canioni P, Guerin B (1993) Interactions between glucose metabolism and oxidative phosphorylations on respiratory-competent Saccharomyces cerevisiae cells. J Biochem 329:51–54
Boumans H, Grivell LA, Berden JA (1998) The respiratory chain in yeast behaves as a single functional unit. J Biol Chem 273:4872–4877
Bulder CJ (1964) Induction of petite mutation and inhibition of synthesis of respiratory enzymes in various yeast. J Microbiol 30:1–9
Davies SE, Brindle KM (1992) Effects of overexpression of phosphofructokinase on glycolysis in the yeast Saccharomyces cerevisiae. Biochemistry 31:4729–4735
De V, Marres CA (1987) The mitochondrial respiratory chain of yeast: structure and biosynthesis and the role in cellar metabolism. Biochim Biophys Acta 895:205–239
Deken De (1966) The crabtree effect and its relation to the petite mutation. J Gen Microbiol 44:157–165
Dietzler DN, Leckie MP, Magnani JL (1975) Evidence for the coordinate control of glycogen synthesis, glucose utilization, and glycolysis in Escherichia coli: II. Quantitative correlation of the inhibition of glycogen synthesis and the stimulation of glucose utilization by 2,4-dinitrophenol with the effects on the cellular levels of glucose 6-phosphate, fructose, 1,6-diphosphate, and total adenylates. J Biol Chem 250(18):7195–7203
Flikweert MT (1999) Physiological roles of pyruvate decarboxylase in Saccharomyces cerevisiae. Ph.D. thesis, Delft University of Technology, Delft
Huang M, Biggs DR, Linnane AW (1966) Chloramphenicol inhibition of the formation of particulate mitochondrial enzymes of Saccharomyces cerevisiae. Biochim Biophys Acta 114:434–436
Hynne F, Dano S, Sorensen PG (2001) Full-scale model of glycolysis in Saccharomyces cerevisiae. Biophys Chem 94:121–163
Jensen PR, Michelsen O (1992) Carbon and energy metabolism of ATP synthase mutants of Escherichia coli. J Bacteriol 174:7635–7641
Joseph-Horne T, Hollomon D, Wood P (2001) Fungal respiration: a fusion of standard and alternative components. Biochim Biophys Acta Bioenerg 1504:179–195
Kerschner SJ, Okun JG, Brandt U (1999) A single external enzyme confers alternative NADH:ubiquinone oxidoreductase activity in Yarrowia lipolytica. J Cell Sci 112:2347–2354
Koebmann BJ, Westerhoff HV, Snoep JL, Nilsson D, Jensen PR (2002) The glycolytic flux in Escherichia coli is controlled by the demand for ATP. J Bacteriol 184:3909–3916
Krebs A, Bridger W (1976) On the monomeric structure and proposed regulatory properties of phosphoenolpyruvate carboxykinase of Escherichia coli. Can J Biochem 54:22–26
Larsson C, Pahlman I, Gustafsson L (2000) The importance of ATP as a regulator of glycolytic flux in Saccharomyces cerevisiae. Yeast 16:797–809
Li Y, Chen J, Lun SY, Rui XS (2001) Efficient pyruvate production by a multi-vitamin auxotroph of Torulopsis glabrata: key role and optimization of vitamin levels. Appl Microbiol Biotechnol 55(6):680–685
Liu LM, Li Y, Li HZ, Chen J (2004) Manipulating the pyruvate dehydrogenase bypass of a multi-vitamin auxotrophic yeast Torulopsis glabrata enhanced pyruvate production. Lett Appl Microbiol 39(2):199–206
Liu LM, Li Y, Li HZ, Chen J (2005) Effect of oxidative phosphorylation inhibitors on the glycolytic flux in Torulopsis glabrata. Prog Biochem Biophys 32(3):251–257
Maiorella B, Blanch H, Wilke C (1983) By-product inhibition effects on ethanolic fermentation by Saccharomyces cerevisiae. Biotechnol Bioeng 25:103–121
Miseta A, Tokes-Fuzesi M, Aiello D, Bedwell D (2003) A Saccharomyces cerevisiae mutant unable to convert glucose to glucose-6-phosphate accumulates excessive glucose in the endoplasmic reticulum due to core oligosaccharide trimming. Eukaryot Cell 2(3):534–541
O’connor RM, Mcarthur CR (1976) Respiratory-deficient mutants of Torulopsis glabrata, a yeast with circular mitochondrial deoxyribonucleic acid of 6 mum. J Bacteriol 126(2):959–968
Overkamp K, Bakker B, Kotter P, van Tuijl A, de Vries S, van Dijken J, JT P (2000) In vivo analysis of the mechanisms for oxidation of cytosolic NADH by Saccharomyces cerevisiae mitochondria. J Bacteriol 182:2823–2830
Santana M, Ionescu MS, Vertes A, Longin R, Kunst F, Danchin A, Glaser P (1994) Bacillus subtilis F0F1-ATP synthase: DNA sequence of the atp operon and characterization of atp mutants. J Bacteriol 176(22):6802–6811
Sato K, Yoshida Y, Hirahata T (2000) On-line measurement of intracellular ATP of Saccharomyces cerevisiae and pyruvate during sake mashing. J Biosci Bioeng 90:294–301
Schmid A, Dordick JS, Hauer B, Kiener A, Wubbolts M, Witholt B (2001) Industrial biocatalysis today and tomorrow. Nature 409(6817):258–268
Sekine H, Shimada T, Hayashi C (2001) H+–ATP synthase defect in Corynebacterium glutamicum abolishes glutamic acid production with enhancement of glucose consumption rate. Appl Microbiol Biotechnol 57:534–540
Senior AE (1988) ATP synthesis by oxidative phosphorylation. Physiol Rev 68(1):177–231
Small WC, McAlister-Henn H (1998) Identification of a cytosolically directed NADH dehydrogenase in mitochondria of Saccharomyces cerevisiae. J Bacteriol 180:4051–4055
Stanley PE (1986) Extraction of adenosine triphosphate from microbial and somatic acid. Methods Enzymol 133:14–22
Stanley G, Douglas E, Every E, Tzanatos T, Pamment N (1993) Inhibition and stimulation of yeast growth by acetaldehyde. Biotechnol Lett 15:1199–1204
Stanley G, Hobley T, Pamment N (1997) Effect of acetaldehyde on Saccharomyces cerevisiae and Zymomonas mobilis subjected to environmental shocks. Biotechnol Bioeng 53:71–78
Van Dijken J, Scheffers W (1986) Redox balances in the metabolism of sugars by yeast. FEMS Microbiol Rev 32:199–224
Wills C, Martin T (1980) Alteration in the redox balance of yeast leads to allyl alcohol resistance. FEBS Lett 119:105–108
Yokota A, Terasawa Y, Takaoka N, Shimizu H, Tomita F (1994) Pyruvic acid production by an F1-ATP synthase-defective mutant of Escherichia coli W1485lip2. Biosci Biotechnol Biochem 58(12):2164–2167
Yokota A, Henmi M, Takaoka N (1997) Enhancement of glucose metabolism in pyruvic acid-hyperproducing Escherichia coli mutant defective in F1-ATP synthase activity. J Ferment Bioeng 83:132–138
Acknowledgements
The authors thank the financial support by the Natural Science Foundation of Jiangsu Province of China (contract No. BK2002072); the Scientific Research Foundation for the Returned Overseas Chinese Scholars, Ministry of Education of China; the Ph.D. Programs Foundation of Ministry of Education of China (No. 20040294003); and the Post-graduate Innovation Program of Jiangsu Province, China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, L., Li, Y., Du, G. et al. Redirection of the NADH oxidation pathway in Torulopsis glabrata leads to an enhanced pyruvate production. Appl Microbiol Biotechnol 72, 377–385 (2006). https://doi.org/10.1007/s00253-005-0284-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-005-0284-3