Abstract
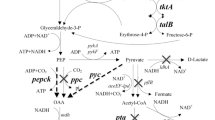
We previously engineered Escherichia coli YL104 to efficiently produce succinate from glucose. In this study, we investigated the relationships between the NADH/NAD+ ratio, ATP level, and overall yield of succinate production by using glucose as the carbon source in YL104. First, the use of sole NADH dehydrogenases increased the overall yield of succinate by 7% and substantially decreased the NADH/NAD+ ratio. Second, the soluble fumarate reductase from Saccharomyces cerevisiae was overexpressed to manipulate the anaerobic NADH/NAD+ ratio and ATP level. Third, another strategy for reducing the ATP level was applied by introducing ATP futile cycling for improving succinate production. Finally, a combination of these methods exerted a synergistic effect on improving the overall yield of succinate, which was 39% higher than that of the previously engineered strain YL104. The study results indicated that regulation of the NADH/NAD+ ratio and ATP level is an efficient strategy for succinate production.





Similar content being viewed by others
References
Ahn JH, Jang YS, Lee SY (2016) Production of succinic acid by metabolically engineered microorganisms. Curr Opin Biotechnol 42:54–66
Becker S, Vlad D, Schuster S, Pfeiffer P, Unden G (1997) Regulatory O2 tensions for the synthesis of fermentation products in Escherichia coli and relation to aerobic respiration. Arch Microbiol 168(4):290–296
Bekker M, de Vries S, Ter Beek A, Hellingwerf K, de Mattos MT (2009) Respiration of Escherichia coli can be fully uncoupled via the nonelectrogenic terminal cytochrome bd-II oxidase. J Bacteriol 191(17):5510–5517
Besteiro S, Biran M, Biteau N, Coustou V, Baltz T, Canioni P, Bringaud F (2002) Succinate secreted by Trypanosoma brucei is produced by a novel and unique glycosomal enzyme, NADH-dependent fumarate reductase. J Biol Chem 277(41):38001–38012
Borisov VB, Murali R, Verkhovskaya ML, Bloch DA, Han H, Gennis RB, Verkhovsky MI (2011) Aerobic respiratory chain of Escherichia coli is not allowed to work in fully uncoupled mode. Proc Natl Acad Sci U S A 108(42):17320–17324
Cabrera R, Baez M, Pereira HM, Caniuguir A, Garratt RC, Babul J (2011) The crystal complex of phosphofructokinase-2 of Escherichia coli with fructose-6-phosphate: kinetic and structural analysis of the allosteric ATP inhibition. J Biol Chem 286(7):5774–5783
Calhoun MW, Oden KL, Gennis RB, de Mattos MJ, Neijssel OM (1993) Energetic efficiency of Escherichia coli: effects of mutations in components of the aerobic respiratory chain. J Bacteriol 175(10):3020–3025
Camarasa C, Faucet V, Dequin S (2007) Role in anaerobiosis of the isoenzymes for Saccharomyces cerevisiae fumarate reductase encoded by OSM1 and FRDS1. Yeast 24(5):391–401
Chao YP, Liao JC (1994) Metabolic responses to substrate futile cycling in Escherichia coli. J Biol Chem 269(7):5122–5126
Coustou V, Besteiro S, Riviere L, Biran M, Biteau N, Franconi JM, Boshart M, Baltz T, Bringaud F (2005) A mitochondrial NADH-dependent fumarate reductase involved in the production of succinate excreted by procyclic Trypanosoma brucei. J Biol Chem 280(17):16559–16570
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. PNAS 97(12):6640–6645
de Graef MR, Alexeeva S, Snoep JL, Teixeira de Mattos MJ (1999) The steady-state internal redox state (NADH/NAD) reflects the external redox state and is correlated with catabolic adaptation in Escherichia coli. J Bacteriol 181(8):2351–2357
de Jongh WA, Nielsen J (2008) Enhanced citrate production through gene insertion in Aspergillus niger. Metab Eng 10(2):87–96
Enomoto K, Arikawa Y, Muratsubaki H (2002) Physiological role of soluble fumarate reductase in redox balancing during anaerobiosis in Saccharomyces cerevisiae. FEMS Microbiol Lett 215(1):103–108
Enomoto K, Ohki R, Muratsubaki H (1996) Cloning and sequencing of the gene encoding the soluble fumarate reductase from Saccharomyces cerevisiae. DNA Res 3(4):263–267
Forster AH, Gescher J (2014) Metabolic engineering of Escherichia coli for production of mixed-acid fermentation end products. Frontiers Biotech Bioeng 2:16
Goldberg I, Lonberg-Holm K, Bagley EA, Stieglitz B (1983) Improved conversion of fumarate to succinate by Escherichia coli strains amplified for fumarate reductase. Appl Environ Microbiol 45(6):1838–1847
Gordon EH, Pealing SL, Chapman SK, Ward FB, Reid GA (1998) Physiological function and regulation of flavocytochrome c3, the soluble fumarate reductase from Shewanella putrefaciens NCIMB 400. Microbiology 144:937–945
Hadicke O, Bettenbrock K, Klamt S (2015) Enforced ATP futile cycling increases specific productivity and yield of anaerobic lactate production in Escherichia coli. Biotechnol Bioeng 112(10):2195–2199
Kang Z, Gao C, Wang Q, Liu H, Qi Q (2010) A novel strategy for succinate and polyhydroxybutyrate co-production in Escherichia coli. Bioresour Technol 101(19):7675–7678
Kihira C, Hayashi Y, Azuma N, Noda S, Maeda S, Fukiya S, Wada M, Matsushita K, Yokota A (2012) Alterations of glucose metabolism in Escherichia coli mutants defective in respiratory-chain enzymes. J Bacteriol 158(4):215–223
Kim Y, Ingram LO, Shanmugam KT (2008) Dihydrolipoamide dehydrogenase mutation alters the NADH sensitivity of pyruvate dehydrogenase complex of Escherichia coli K-12. J Bacteriol 190(11):3851–3858
Kuhlman TE, Cox EC (2010) Site-specific chromosomal integration of large synthetic constructs. Nucleic Acids Res:gkp1193
Leonardo MR, Dailly Y, Clark DP (1996) Role of NAD in regulating the adhE gene of Escherichia coli. J Bacteriol 178(20):6013–6018
Li Y, Huang B, Wu H, Li Z, Ye Q, Zhang YP (2016) Production of succinate from acetate by metabolically engineered Escherichia coli. ACS Synth Biol 5(11):1299–1307
Li Y, Li M, Zhang X, Yang P, Liang Q, Qi Q (2013) A novel whole-phase succinate fermentation strategy with high volumetric productivity in engineered Escherichia coli. Bioresour Technol 149:333–340
Liang L, Liu R, Wang G, Gou D, Ma J, Chen K, Jiang M, Wei P, Ouyang P (2012) Regulation of NAD(H) pool and NADH/NAD+ ratio by overexpression of nicotinic acid phosphoribosyltransferase for succinic acid production in Escherichia coli NZN111. Enzym Microb Technol 51(5):286–293
Liang LY, Liu RM, Ma JF, Chen KQ, Jiang M, Wei P (2011) Increased production of succinic acid in Escherichia coli by overexpression of malate dehydrogenase. Biotechnol Lett 33(12):2439–2444
Liang Q, Qi Q (2014) From a co-production design to an integrated single-cell biorefinery. Biotechnol Adv 32(7):1328–1335
Liang Q, Zhang F, Li Y, Zhang X, Li J, Yang P, Qi Q (2015) Comparison of individual component deletions in a glucose-specific phosphotransferase system revealed their different applications. Sci Rep 5:13200
Liu Q, Lin Z, Zhang Y, Li Y, Wang Z, Chen T (2014) Improved poly(3-hydroxybutyrate) production in Escherichia coli by inactivation of cytochrome bd-II oxidase or/and NDH-II dehydrogenase in low efficient respiratory chains. J Bacteriol 192:170–176
Liu R, Liang L, Li F, Wu M, Chen K, Ma J, Jiang M, Wei P, Ouyang P (2013) Efficient succinic acid production from lignocellulosic biomass by simultaneous utilization of glucose and xylose in engineered Escherichia coli. Bioresour Technol 149:84–91
Meng J, Wang B, Liu D, Chen T, Wang Z, Zhao X (2016) High-yield anaerobic succinate production by strategically regulating multiple metabolic pathways based on stoichiometric maximum in Escherichia coli. Microb Cell Factories 15(1):141
Millard CS, Chao YP, Liao JC, Donnelly MI (1996) Enhanced production of succinic acid by overexpression of phosphoenolpyruvate carboxylase in Escherichia coli. Appl Environ Microbiol 62(5):1808–1810
Muratsubaki H, Enomoto K (1998) One of the fumarate reductase isoenzymes from Saccharomyces cerevisiae is encoded by the OSM1 gene. Arch Biochem Biophys 352(2):175–181
Olajuyin AM, Yang M, Liu Y, Mu T, Tian J, Adaramoye OA, Xing J (2016) Efficient production of succinic acid from Palmaria palmata hydrolysate by metabolically engineered Escherichia coli. Bioresour Technol 214:653–659
Patnaik R, Roof WD, Young RF, Liao JC (1992) Stimulation of glucose catabolism in Escherichia coli by a potential futile cycle. J Bacteriol 174(23):7527–7532
Portnoy VA, Herrgard MJ, Palsson BO (2008) Aerobic fermentation of D-glucose by an evolved cytochrome oxidase-deficient Escherichia coli strain. Appl Environ Microbiol 74(24):7561–7569
Salusjarvi L, Kaunisto S, Holmstrom S, Vehkomaki ML, Koivuranta K, Pitkanen JP, Ruohonen L (2013) Overexpression of NADH-dependent fumarate reductase improves D-xylose fermentation in recombinant Saccharomyces cerevisiae. J Ind Microbiol Biotechnol 40(12):1383–1392
Singh A, Cher Soh K, Hatzimanikatis V, Gill RT (2011) Manipulating redox and ATP balancing for improved production of succinate in E. coli. Metab Eng 13(1):76–81
Singh A, Lynch MD, Gill RT (2009) Genes restoring redox balance in fermentation-deficient E. coli NZN111. Metab Eng 11(6):347–354
Steinsiek S, Stagge S, Bettenbrock K (2014) Analysis of Escherichia coli mutants with a linear respiratory chain. PLoS One 9(1):e87307
Stols L, Donnelly MI (1997) Production of succinic acid through overexpression of NAD(+)-dependent malic enzyme in an Escherichia coli mutant. Appl Environ Microbiol 63(7):2695–2701
Stouthamer A (1979) The search for correlation between theoretical and experimental growth yields. Int Rev Biochem 21(1):1–47
Tielens AG, Van Hellemond JJ (1998) The electron transport chain in anaerobically functioning eukaryotes. Biochim Biophys Acta 1365(1–2):71–78
Van der Werf MJ, Guettler MV, Jain MK, Zeikus JG (1997) Environmental and physiological factors affecting the succinate product ratio during carbohydrate fermentation by Actinobacillus sp. 130Z. Arch Microbiol 167(6):332–342
Van Hellemond JJ, Tielens AG (1994) Expression and functional properties of fumarate reductase. The Biochem J 304:321–331
Vemuri GN, Altman E, Sangurdekar DP, Khodursky AB, Eiteman MA (2006) Overflow metabolism in Escherichia coli during steady-state growth: transcriptional regulation and effect of the redox ratio. Appl Environ Microbiol 72(5):3653–3661
Vemuri GN, Eiteman MA, Altman E (2002) Effects of growth mode and pyruvate carboxylase on succinic acid production by metabolically engineered strains of Escherichia coli. Appl Environ Microbiol 68(4):1715–1727
Verkhovskaya ML, Belevich N, Euro L, Wikstrom M, Verkhovsky MI (2008) Real-time electron transfer in respiratory complex I. Proc Natl Acad Sci U S A 105(10):3763–3767
Vuoristo KS, Mars AE, Sanders JP, Eggink G, Weusthuis RA (2016) Metabolic engineering of TCA cycle for production of chemicals. Trends Biotechnol 34(3):191–197
Wang C, Yan D, Li Q, Sun W, Xing J (2014) Ionic liquid pretreatment to increase succinic acid production from lignocellulosic biomass. Bioresour Technol 172:283–289
Weitzman PD (1981) Unity and diversity in some bacterial citric acid-cycle enzymes. Adv Microb Physiol 22:185–244
Werpy T, Petersen G (2004) Volume 1: results of screening for potential candidates from sugars and synthetic gas. Oak Ridge, TN, US Department of Energy
Wu H, Tuli L, Bennett GN, San KY (2015) Metabolic transistor strategy for controlling electron transfer chain activity in Escherichia coli. Metab Eng 28:159–168
Zhang F, Li J, Liu H, Liang Q, Qi Q (2016) ATP-based ratio regulation of glucose and xylose improved succinate production. PLoS One 11(6):e0157775
Zhu X, Tan Z, Xu H, Chen J, Tang J, Zhang X (2014) Metabolic evolution of two reducing equivalent-conserving pathways for high-yield succinate production in Escherichia coli. Metab Eng 24:87–96
Acknowledgements
This work was financially supported by a grant from the National Basic Research Program of China (2012CB725202), a grant from the National Natural Science Foundation of China (31170097, 31370085), and a grant from the Shandong Science and Technology Development Plan (2015GSF121042).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
.
ESM 1
(PDF 182 kb)
Rights and permissions
About this article
Cite this article
Li, J., Li, Y., Cui, Z. et al. Enhancement of succinate yield by manipulating NADH/NAD+ ratio and ATP generation. Appl Microbiol Biotechnol 101, 3153–3161 (2017). https://doi.org/10.1007/s00253-017-8127-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8127-6