Abstract
Background
Although clozapine is an effective option for treatment-resistant schizophrenia (TRS), there are still 1/3 to 1/2 of TRS patients who do not respond to clozapine. The main purpose of this randomized, double-blind, placebo-controlled trial was to explore the amisulpride augmentation efficacy on the psychopathological symptoms and cognitive function of clozapine-resistant treatment-refractory schizophrenia (CTRS) patients.
Methods
A total of 80 patients were recruited and randomly assigned to receive initial clozapine plus amisulpride (amisulpride group) or clozapine plus placebo (placebo group). Positive and Negative Syndrome Scale (PANSS), Scale for the Assessment of Negative Symptoms (SANS), Clinical Global Impression (CGI) scale scores, Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), Treatment Emergent Symptom Scale (TESS), laboratory measurements, and electrocardiograms (ECG) were performed at baseline, at week 6, and week 12.
Results
Compared with the placebo group, amisulpride group had a lower PANSS total score, positive subscore, and general psychopathology subscore at week 6 and week 12 (PBonferroni < 0.01). Furthermore, compared with the placebo group, the amisulpride group showed an improved RBANS language score at week 12 (PBonferroni < 0.001). Amisulpride group had a higher treatment response rate (P = 0.04), lower scores of CGI severity and CGI efficacy at week 6 and week 12 than placebo group (PBonferroni < 0.05). There were no differences between the groups in body mass index (BMI), corrected QT (QTc) intervals, and laboratory measurements. This study demonstrates that amisulpride augmentation therapy can safely improve the psychiatric symptoms and cognitive performance of CTRS patients.
Conclusion
This study indicates that amisulpride augmentation therapy has important clinical significance for treating CTRS to improve clinical symptoms and cognitive function with tolerability and safety.
Trial registration Clinicaltrials.gov identifier- NCT03652974. Registered August 31, 2018, https://clinicaltrials.gov/ct2/show/NCT03652974
Similar content being viewed by others
Background
Schizophrenia is a severe psychiatric disorder characterized by positive symptoms, negative symptoms, and cognitive deficits [1, 2]. Despite a wide variety of available antipsychotic drugs, there are still many schizophrenia patients (about 1/5 to 1/3) who are resistant to two or more antipsychotic treatments, defined as “treatment-resistant schizophrenia (TRS)” or “treatment-refractory schizophrenia” [3, 4].
Clozapine is the only evidence-based antipsychotic drug for treating TRS patients [5, 6]. However, even with sufficient clozapine levels in the blood, about 1/3 to 1/2 of TRS patients are still resistant to clozapine [7, 8]. According to the definition of TRS proposed by Kane [9] and Honer et al. [10], TRS patients who do not respond well to clozapine monotherapy are known as having clozapine-resistant treatment-refractory schizophrenia (CTRS). According to the National Institute for Health and Care Excellence guidelines for treating TRS [11], augmentation therapies may have potential benefits for TRS patients who do not respond to clozapine monotherapy [12]. Previous studies have shown that when dopamine D2 receptors are 70% or more occupied, antipsychotics achieve their maximum efficacy [13, 14]. Clozapine is an antipsychotic drug with multi-receptor blocking effects, and its affinity for dopamine D2 receptors is low [15, 16]. Amisulpride has highly selective blocking effects on dopamine D2 and dopamine D3 receptors [17]. The unique dopamine receptor blocking effects of amisulpride can selectively enhance the limited dopamine D2 receptor blocking effects of clozapine [18, 19], making it a suitable drug for combination with clozapine [19]. In addition, a previous meta-analysis has shown that the efficacy of amisulpride is second only to clozapine and that its treatment interruption rate is the lowest among the 15 antipsychotics commonly used for schizophrenia treatment [20]. Previous randomized, double-blind, placebo-controlled trials using amisulpride augmented with clozapine in TRS patients showed no statistical advantages [19, 21]. However, these studies had relatively small sample sizes, and their patients may not have met CTRS criteria for the following reasons: 1) patients participating in the study may not have actually received two antipsychotic agents with different mechanisms of action in the past five years, or patients may not have taken the appropriate dose for a sufficient period of time before clozapine treatment; and 2) the studies stipulated that clozapine monotherapy should be administered for at least 3 months instead of 6 months. Previous studies have shown that some patients may have a delayed response to clozapine. Among these patients, 30% respond after 6 weeks, 20% respond after 3 months, and 10–20% respond after 6 months [22, 23], suggesting that clozapine resistance should ideally be measured after 6 months. The evidence for amisulpride combined with clozapine in the treatment of CTRS patients remains poor. Most importantly, to the best of our knowledge, few studies have investigated the effects of amisulpride augmentation on cognitive function of CTRS patients. It has been reported that 98% of patients with schizophrenia have cognitive impairment, including first-onset or chronic episode patients [24, 25]. The recovery of cognitive function is considered to be one of the main goals of clinical treatment of schizophrenia [26, 27].
Thus, this 12-week, randomized, double-blind, placebo-controlled study aims to investigate the efficacy and safety of amisulpride augmentation therapy in CTRS patients who have received at least two appropriate doses of antipsychotics with different chemical structures within a sufficient period of time and have recently received a stable dose of clozapine (i.e., at least 400 mg or more per day) for at least 6 months. The main purpose of this study was to investigate whether amisulpride augmentation therapy improved the psychopathological symptoms and cognitive performance of these CTRS patients.
Methods
The study is a randomized, double-blind, placebo-controlled trial. Regulatory approvals for this study were obtained from the Institutional Review Board of Shanghai Pudong New Area Mental Health Center (No. 2018008), and each written informed consent was signed. The protocol was registered before participant enrolment on clincialtrials.gov (ID: NCT03652974).
Participants
All participants were recruited from the Shanghai Pudong New Area Mental Health Center between September 6, 2018, and August 1, 2021. The inclusion criteria were: (1) Han Chinese ethnicity; (2) between 18 and 65 years old; (3) satisfied the diagnostic criteria for schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV), using the Structured Clinical Interview for DSM-IV (SCID-I/P); (4) had received at least two antipsychotic agents with different mechanisms of action, at appropriate doses for a sufficient course of treatment, and had recently received a stable dose of clozapine (i.e., at least 400 mg/d or more for at least 6 months) in order to ensure a reasonable response to clozapine monotherapy; (5) a review of the patient’s past medical history revealed that the patient had stubborn psychotic symptoms and had never been effectively controlled; and (6) the patient had a baseline PANSS score > 60 before entering the study [28]. The exclusion criteria were: (1) any other major Axis I disorder; (2) serious physical diseases; (3) substance abuse/dependence; or (4) pregnant women.
Intervention procedures
After the enrollment was completed, all eligible CTRS patients continued to take clozapine (doses from 400 to 550 mg) and were randomly assigned to receive clozapine plus amisulpride or clozapine plus placebo on a 1:1 basis. Randomization was carried out according to computer-generated random identification. The titration started with amisulpride 200 mg/d or 1 placebo tablet in the first week, amisulpride 400 mg/d or 2 placebo tablets in the second week, and up to 800 mg of amisulpride or 4 placebo tablets for the remaining 10 weeks.
Patients suffering from severe anxiety or insomnia were treated with benzodiazepines over a short time. Diphenylethyl hydrochloride was applied for a limited time in patients with extrapyramidal symptoms (EPS). No other antipsychotics and antidepressants were allowed during this study. The amisulpride and placebo tablets were identical in appearance. All researchers and participants were masked for treatment randomization and assessments.
The assessments of Positive and Negative Syndrome Scale (PANSS), Scale for the Assessment of Negative Symptoms (SANS), Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), Clinical Global Impression (CGI), and Treatment Emergent Symptom Scale (TESS) were evaluated at baseline, week 6, and week 12. The primary outcome was the PANSS scores at week 6 and week 12. The secondary outcome was the responder rate, SANS, RBANS, and CGI scores at week 6 and week 12. The treatment response was determined by a more than 25% reduction in the PANSS total score [29, 30]. The PANSS reduction rate was calculated using the following formula: (baseline PANSS total score − follow-up PANSS total score)/(baseline PANSS total score − 30) × 100% [31].
Clinical assessments
The PANSS was applied to assess psychiatric symptoms. Negative symptoms were evaluated using the SANS. The RBANS was applied to assess cognitive performance. The Clinical Global Impression severity (CGI-S), CGI improvement (CGI-I), and CGI efficacy (CGI-E) were applied to assess the symptom severity, treatment responses, and treatment effects. The TESS was applied to evaluate adverse events related to treatment. All psychiatrists were trained in the administration of the assessments, and inter-rater correlation coefficients were all above 0.8.
Laboratory measurement, physical examination, and electrocardiogram (ECG), during the clinical trial
Laboratory measurements, physical examinations, and ECG were performed at baseline, the 6th week timepoint, and the 12th week timepoint. After an overnight fast, blood samples were collected to detect serum clozapine levels, carry out routine blood analysis, obtain a lipid profile, and measure glucose, liver, and renal function. Serum clozapine level was assayed by high performance liquid chromatography. Routine blood analysis was estimated by white blood cell (WBC), neutrophils, red blood cell (RBC), hemoglobin (Hb) and platelet (PLT), measured using a Sysmex hematology analyzer with its supporting reagents. Lipid profiles were estimated by triglyceride (TG), total cholesterol (TC), high density lipoprotein cholesterol (HDLC) and low density lipoprotein cholesterol (LDLC). TG, TC and HDLC were measured using enzymatic assay Kit (Zybio). HDLC was measured using low density lipoprotein assay Kit (Gcell). Glucose was measured using enzymatic assay Kit (Zybio). Liver function was estimated by alaninetransaminase (ALT) and aspartate transaminase (AST), measured using enzymatic assay Kit (Zybio). Renal function was estimated by serum creatinine, blood urea nitrogen (BUN) and uric acid, measured using enzymatic assay Kit (Zybio). All laboratory measurements were carried out according to the protocol provided by the manufacturer. ECG was performed using EDAN ECG machine (SE-1010).
Statistical analysis
The distribution of the data was detected through the Kolmogorov–Smirnov one-sample test. The balance of baseline demographic and clinical characteristics between groups was compared using the chi-square test and analysis of variance (ANOVA). The qualitative data were presented as percentages and the quantitative data were expressed as mean ± standard deviation (SD). An intent-to-treat (ITT) analysis was used for sensitivity purposes, and the principle of last-observation-carrying-forward (LOCF) was used to deal with missing data.
In the beginning, repeated-measure multivariate analysis of variance (RM MANOVA) was applied to obtain the overall P value of PANSS and RBANS scores, respectively. Then, a repeated-measures analysis of variance (RM ANOVA) was used to examine each score of PANSS and RBANS, respectively, setting between-group factors (amisulpride and placebo) and within-group factors (baseline, week 6, and week 12), while also adjusting for confounding covariates. An RM ANOVA was conducted to measure every item of the PANSS subscale in the amisulpride group. An RM ANOVA was conducted to measure SANS scores, CGI scores, TESS total scores, body mass index (BMI), corrected QT (QTc) interval, and each laboratory measurement index, respectively. After performing an RM ANOVA, a follow-up significant multivariate omnibus test was performed, and each univariate effect was detected using an analysis of covariance (ANCOVA). If the group × time interaction was not significant, no statistical testing was further needed. If the group × time interaction had significance, an ANCOVA was used to analyze the group differences at week 6 and week 12, setting baseline score, BMI, age, sex, disease course, and baseline clozapine serum level as covariates. Bonferroni corrections were applied to correct for multiple tests. P values < 0.05 were considered statistically significant. Based on the power and sample size calculation at the 2-tailed 5% significance level, a sample size of 34 per group (total n = 68) yielded 80% of the power to detect significant differences in the primary outcome. In this study, we assumed that the dropout rate was less than 15%. PASW Statistics, version 23.0 (SPSS, Inc., Chicago, USA) was applied for statistical analyses.
Results
Demographic and baseline information
Among the 113 participants assessed for eligibility, 80 were recruited and randomly assigned to one of the groups (Fig. 1). Among these patients, 78 completed the 6-week trial, and 71 completed the 12-week trial. At week 6, one patient (2.5%) in the amisulpride group and one in the placebo group dropped out. At week 12, 3 patients (7.5%) in the amisulpride group and 4 (10.0%) in the placebo group dropped out. The average dose of amisulpride in the amisulpride group was 771.4 mg/d at the end of 12-week. As shown in Table 1, at baseline, except for BMI (F = 4.85, P = 0.03), there was no significant difference in any demographic or clinical characteristics (PANSS, RBANS, SANS, and CGI scores) between the two groups (P > 0.05). The amisulpride group had a higher BMI than the placebo group. Therefore, BMI was adjusted in the subsequent statistical analysis. There was no difference in the clozapine dose or serum clozapine levels at baseline between the amisulpride and the placebo groups (P > 0.05). Furthermore, after adjusting BMI and baseline clozapine dose, RM ANOVA showed no group × time effect, time effect, or group effect on serum clozapine levels (P > 0.05), indicating that there was no difference in the change of serum clozapine levels after 12 weeks of treatment. In addition, there was no difference in demographic or clinical characteristics between dropouts and completers (P > 0.05).
Effect of amisulpride augmentation therapy on PANSS scores
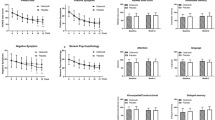
RM MANOVA was first conducted using PANSS subscales and total score as the outcome measurement and BMI as the covariate, and showed a significant group × time effect (Wilks’ lambda F = 10.50, P < 0.0001). Then, RM ANOVA showed group × time effects on PANSS total score (Wilks’ lambda F = 11.75, P < 0.001), positive symptom subscore (Wilks’ lambda F = 3.66, P = 0.03) and general psychopathology subscore (Wilks’ lambda F = 9.03, P < 0.001) (Table 2). Next, after adjusting for BMI, age, sex, disease course, baseline PANSS scores, and baseline clozapine serum levels, an ANCOVA was used to examine the group difference in PANSS total and subscale scores at week 6 and week 12, respectively. As shown in Fig. 2 at week 12, amisulpride group displayed lower PANSS total score, positive symptom subscore, and general psychopathology subscore compared with placebo group (PBonferroni = 0.004, Cohen’s d = 0.45; PBonferroni < 0.0001, Cohen’s d = 0.97; PBonferroni < 0.001, Cohen’s d = 0.92; respectively). At week 6, amisulpride group displayed lower PANSS total score, positive symptom subscore and general psychopathology subscore compared with placebo group (PBonferroni = 0.004, Cohen’s d = 0.88; PBonferroni < 0.0001, Cohen’s d = 0.99; PBonferroni = 0.004, Cohen’s d = 0.89; respectively).
Effect of amisulpride augmentation therapy on Positive and Negative Syndrome Scale (PANSS) scores. At week 6 and 12, the amisulpride group displayed lower PANSS total score, positive symptom subscore, and general psychopathology subscore compared with the placebo group (week 6:F = 11.18, P = 0.001, PBonferroni = 0.004, Cohen’s d = 0.88; F = 16.63, P < 0.0001, PBonferroni < 0.0001, Cohen’s d = 0.99; F = 12.56, P = 0.001, PBonferroni = 0.004, Cohen’s d = 0.89. week 12:F = 11.34, P = 0.001, PBonferroni = 0.004, Cohen’s d = 0.45; F = 17.10, P < 0.0001, PBonferroni < 0.0001, Cohen’s d = 0.97; F = 14.00, P < 0.0001, PBonferroni < 0.001, Cohen’s d = 0.92). **P < 0.01, ***P < 0.001; ns non-significant
Effect of amisulpride augmentation therapy on treatment response rate
ITT analysis showed that, at week 12, the response rate of the amisulpride group (10 patients, 25%) was higher than that of the placebo group (2 patients, 5%) (χ2 = 6.28, P = 0.01, OR = 6.33, 95% CI 1.29–31.12). After adjusting for BMI, age, sex, disease course, and baseline clozapine serum levels, it also showed a significant between-group difference (B = 1.82, Wald’s statistics = 4.06, P = 0.04, OR = 6.15, 95% CI 1.06–36.03), the amisulpride group had a higher response rate than the placebo group.
Effect of amisulpride augmentation therapy on cognitive function
RM MANOVA showed a significant group × time effect (Wilks’ lambda F = 4.64; P = 0.01) on RBANS scores. After adjusting for BMI, an RM ANOVA was applied for RBANS total and subscale scores, respectively. As shown in Table 2, there was a group × time effect of RBANS total score and language score (Wilks’ lambda F = 3.54, P = 0.03; Wilks’ lambda F = 5.49, P = 0.006).
Then, after adjusting the baseline score and other clinical covariates, an ANCOVA was applied to examine the group differences in the RBANS total score and language score at week 6 and week 12, respectively. As shown in Fig. 3, at week 12, the amisulpride group displayed higher RBANS total and language scores compared with placebo group (P = 0.01, Cohen’s d = 0.41; P < 0.0001, Cohen’s d = 0.77). However, only the difference in language score remained significant after Bonferroni correction (PBonferroni < 0.001). At week 6, there were no between-group differences in RBANS total score or language score (P = 0.12; P = 0.08).
Effect of amisulpride augmentation therapy on Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) scores. At week 12, the amisulpride group displayed higher RBANS total and language scores compared with placebo group (F = 6.14, P = 0.01, Cohen’s d = 0.41; F = 14.82, P < 0.0001, Cohen’s d = 0.77). However, only the difference in language score remained significant after Bonferroni correction (PBonferroni < 0.001). At week 6, there were no between-group differences in RBANS total score or language score (F = 2.52, P = 0.12; F = 3.14, P = 0.08). *P < 0.5, ***P < 0.001; ns non-significant
Effect of amisulpride augmentation therapy on SANS and CGI scores
An RM ANOVA was performed on SANS, CGI-S, CGI-I, and CGI-E scores, after controlling for BMI as a covariate. As shown in Table 2, there were group × time effects on CGI-S (Wilks’ lambda F = 10.85, P < 0.001), CGI-I (Wilks’ lambda F = 4.16, P = 0.02) and CGI-E (Wilks’ lambda F = 12.17, P < 0.001) scores. However, there was no significant group × time effect on the SANS score (Fig. 4a).
Effect of amisulpride augmentation therapy on the scores of Scale for the Assessment of Negative Symptoms (SANS) and Clinical Global Impression (CGI). a SANS score. b–d CGI-S, CGI-I, and CGI-E scores. SANS score showed no difference between two groups at week 12 or week 6 (a). At week 12, the amisulpride group had lower CGI-S, CGI-I and CGI-E scores than the placebo group (F = 20.37, P < 0.0001, Cohen’s d = 0.91; F = 5.75, P = 0.02, Cohen’s d = 0.80; F = 19.02, P < 0.0001, Cohen’s d = 1.06; respectively). However, after Bonferroni correction, only CGI-S and CGI-E scores still showed significant between-group differences (both PBonferroni < 0.0001) (b–d). At week 6, the amisulpride group had lower CGI-S and CGI-E scores than the placebo group (F = 10.93, P = 0.001, PBonferroni = 0.003, Cohen’s d = 0.72; F = 8.98, P = 0.004, PBonferroni = 0.01, Cohen’s d = 0.73) (b–d). *P < 0.05, **P < 0.01, ***P < 0.001; ns non-significant
Next, an ANCOVA was conducted to examine the group differences in the CGI-S, CGI-I, and CGI-E scores at week 6 and week 12, respectively, after adjusting for baseline scores and other clinical covariates. As shown in Fig. 4b-d, at week 12, the amisulpride group had lower CGI-S, CGI-I and CGI-E scores than the placebo group (P < 0.0001, Cohen’s d = 0.91; P = 0.02, Cohen’s d = 0.80; P < 0.0001, Cohen’s d = 1.06; respectively). However, after Bonferroni correction, only CGI-S and CGI-E scores still showed significant between-group differences (both PBonferroni < 0.0001). At week 6, the amisulpride group had lower CGI-S and CGI-E scores than the placebo group (PBonferroni = 0.003, Cohen’s d = 0.72; PBonferroni = 0.01, Cohen’s d = 0.73).
Treatment side effects and safety
As shown in Table 3, after adjusting for BMI as a covariate, RM ANOVA showed no significant group × time effect, main time effect, or group effect on the TESS total score (P > 0.05). In addition, after Bonferroni correction, there were no significant group × time effects on BMI, QTc intervals, or laboratory measurements (PBonferroni > 0.05).
At week 12, the most common adverse effects were mild in both groups, including dry mouth, constipation, EPS, gastrointestinal reactions, saliva, hypersomnia, insomnia, and headache. There was no significant difference in the incidence of side effects between the amisulpride group and the placebo group (P > 0.05).
Discussion
This randomized, double-blind, placebo-controlled trial study suggests that amisulpride augmentation therapy can safely improve the clinical symptoms and cognitive function of CTRS patients. Compared with the placebo group, the positive and general psychopathological symptoms of CTRS patients in the amisulpride augmentation group continued to improve at week 6 and week 12. Moreover, the 12-week amisulpride augmentation therapy increased the response rate compared to placebo. Also, compared with the placebo, the CGI-S and CGI-E scores of CTRS patients with amisulpride augmentation therapy were significantly reduced. Our results are partially consistent with a relatively small sample size (n = 16) open-label non-randomized study, which found that amisulpride augmentation therapy improved the positive symptoms of schizophrenia partially responded to clozapine [32].
The theory that amisulpride enhances the efficacy of clozapine is based on the fact that the receptor profiles of these two drugs are complementary. Among those who do not respond to clozapine, clozapine monotherapy may not reach the level of D2 receptor blockade [33, 34], because the level of D2 receptor blockade needs to be about 80% to produce a significant response [13, 35]. In patients who do not respond to clozapine monotherapy, the selective effects of amisulpride in the mesolimbic system may cause D2 receptors to be blocked at the therapeutic level. In addition, amisulpride appears to affect 5HT-7 receptors [36] and presynaptic autoreceptors, which may affect the regulation of endogenous dopamine production [37]. Furthermore, D3 receptors are located in the nucleus accumbens and cerebral cortex, and are associated with neural circuits implicated in schizophrenia [38, 39]. A meta-analysis involving more than 2500 patients showed a slight but significant correlation between D3 receptor coding sequence polymorphisms and susceptibility to schizophrenia [40]. Previous studies have shown that selective D3 antagonists may be effective antipsychotic agents for the treatment of schizophrenia. Because of their anatomical distribution in the ventral striatum [41], their locomotor adverse effects, including extrapyramidal side effects and catalepsy, may be negligible [42,43,44]. For example, a 6-week randomized, double-blind, placebo-controlled trial confirmed the efficacy and safety of D3 antagonists for improving acute exacerbations of schizophrenia [45]. It has also been reported that high D2 antagonism or higher doses of antipsychotic drugs, which are more likely to over-occupy D2 receptors, may increase the risk of secondary negative symptoms [46]. However, our study did not find any effects of amisulpride augmentation therapy on the negative symptoms of CTRS patients based on the PANSS negative subscale and the SANS assessment, which was consistent with the study of Barnes and their colleagues [19]. In clinical practice, negative symptoms of schizophrenia are usually stable and difficult to treat. Amisulpride has been proved to treat schizophrenia patients with predominantly negative symptoms and the approved dose is 50–300 mg/d [47]. In our study, the dose of amisulpride has exceeded 400 mg/d since the beginning of week 3, and we did not perform PANSS scale assessments at the endpoint of week 2. In addition, the high dopamine blockade effect, caused by clozapine and high doses of amisulpride in our study, may induce secondary negative symptoms causing insignificant reductions in negative scores [47].
Previous evidence suggests that amisulpride can improve the cognitive function of schizophrenia patients [48, 49]. However, few studies have examined the effects of amisulpride combined with clozapine on the cognitive function of schizophrenia patients. Park et al. [50] previously reported that amisulpride augmentation therapy improved the working memory of schizophrenia patients treated with aripiprazole. Recently, Molina et al. [51] revealed that the combined use of amisulpride and quetiapine improved both clinical symptoms and cognitive function, especially the executive function of TRS. In this study, we found that amisulpride augmentation therapy also improved the cognitive performance of CTRS patients, particularly language function.
Picture naming and semantic fluency tasks are the RBANS items used in this study to test the language domain. Similar to our research, Salmazo-Silva et al. [52] also used image naming and semantic fluency tasks to assess language abilities, but they employed Parkinson’s disease as their target ailment. Language and perception disorders are the core cognitive impairment symptoms in schizophrenia [53]. The underlying mechanism may be related to the antagonistic effects of amisulpride on D2, D3, and 5-HT7 receptors. For example, previous evidence showed that resting blood flow in the hippocampus of patients with schizophrenia was abnormally increased [54], indicating an increase in resting metabolism in this region [55, 56]. Tregellas et al. [57] found that the resting hyperactivity of the hippocampus strongly relates to cognitive deficits in schizophrenia patients. Interestingly, dopamine D2 antagonists have been shown to reverse the abnormal increase in hippocampal blood flow in patients with schizophrenia [58]. In addition, Shin et al. [59] found that D2 receptor antagonism may improve the working memory function of schizophrenia patients. Previous evidence suggests that 5-HT7 receptor antagonists may affect neuronal morphology [60, 61] and stimulate hippocampal neurogenesis [62, 63], related to schizophrenia and cognitive function. In addition, preclinical studies using a rat model of schizophrenia-like cognitive impairment have demonstrated that 5-HT7 receptor antagonists can improve pro-cognitive function, and that amisulpride can improve stress-related frontal lobe cognitive impairment [64]. Preclinical evidence shows that D3 antagonists can reverse the deficiency of dopamine tension in the prefrontal cortex [65], which may improve cognition [66,67,68].
In this study, there were no differences in side effects or safety between patients receiving amisulpride augmentation therapy and the placebo, which was partially consistent with a previous open-label, non-randomized study [32]. This outcome shows that amisulpride augmentation therapy improves positive symptoms of CTRS patients without exacerbating side effects. It is well established that long-term disease courses and antipsychotics, especially atypical antipsychotics, increase the prevalence of metabolic disorders [69]. In this study, a comparison between patients treated with clozapine alone and patients treated with clozapine plus amisulpride for 12 weeks showed that both groups had similar metabolic outcomes, including BMI, blood lipids, and fasting blood glucose. As for cardiac side effects, an overdose of amisulpride increases the risk of prolonged QTc, but the risk is low at therapeutic doses [70]. Our results indicate that the therapeutic doses of amisulpride augmentation therapy did not increase the risk of QTc interval prolongation in CTRS patients.
The study reported here had many strengths, including acceptable sample size, an appropriate observation period, and multidimensional efficacy and safety assessments. In addition, all plasma samples of participants were obtained, therefore, future study will be conducted to investigate the peripheral protein biomarkers for CTRS and treatment efficacy.
Some limitations of our study should be noted. First, the sample size is relatively small, and our findings should be verified in a larger sample that is drawn from multiple centers. Second, the patients included in this study had chronic conditions, so the results of this study cannot be generalized to other settings. Third, The follow-up time for cognitive function improvement is relatively short.
Conclusions
In summary, our findings demonstrate that amisulpride augmentation therapy can safely improve clinical symptoms and cognitive function in CTRS patients. Amisulpride augmentation therapy has important clinical significance for the treatment of CTRS. Although the results of this study are promising, further multiple-center studies with larger sample sizes should be conducted to confirm the efficacy and safety of this treatment in different clinical settings.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ANCOVA:
-
Analysis of covariance
- ANOVA:
-
Analysis of variance
- BMI:
-
Body mass index
- CGI:
-
Clinical Global Impression scale
- CGI-S:
-
CGI severity
- CGI-I:
-
CGI improvement
- CGI-E:
-
CGI efficacy
- CTRS:
-
Clozapine-resistant treatment-refractory schizophrenia
- DSM-IV:
-
Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition
- ECG:
-
Electrocardiogram
- EPS:
-
Extrapyramidal symptoms
- ITT:
-
Intent-to-treat
- LOCF:
-
Last-observation-carrying-forward
- PANSS:
-
Positive and Negative Syndrome Scale
- QTc:
-
Corrected QT
- RBANS:
-
Repeatable Battery for the Assessment of Neuropsychological Status
- RM ANOVA:
-
Repeated-measures analysis of variance
- RM MANOVA:
-
Repeated-measure multivariate analysis of variance
- SANS:
-
Scale for the Assessment of Negative Symptoms
- TESS:
-
Treatment Emergent Symptom Scale
- TRS:
-
Treatment-resistant schizophrenia
References
He Q, Jantac Mam-Lam-Fook C, Chaignaud J, Danset-Alexandre C, Iftimovici A, Gradels Hauguel J, et al. Influence of polygenic risk scores for schizophrenia and resilience on the cognition of individuals at-risk for psychosis. Transl Psychiatry. 2021;11(1):518.
Pardinas AF, Holmans P, Pocklington AJ, Escott-Price V, Ripke S, Carrera N, et al. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat Genet. 2018;50(3):381–9.
Rodrigues-Silva C, Semedo AT, Neri H, Vianello RP, Galaviz-Hernandez C, Sosa-Macias M, et al. The CYP2C19*2 and CYP2C19*17 polymorphisms influence responses to clozapine for the treatment of schizophrenia. Neuropsychiatr Dis Treat. 2020;16:427–32.
Nucifora FC Jr, Woznica E, Lee BJ, Cascella N, Sawa A. Treatment resistant schizophrenia: clinical, biological, and therapeutic perspectives. Neurobiol Dis. 2019;131:104257.
Zhuo C, Xu Y, Hou W, Chen J, Li Q, Liu Z, et al. Mechanistic/mammalian target of rapamycin and side effects of antipsychotics: insights into mechanisms and implications for therapy. Transl Psychiatry. 2022;12(1):13.
Vita A, Minelli A, Barlati S, Deste G, Giacopuzzi E, Valsecchi P, et al. Treatment-resistant schizophrenia: genetic and neuroimaging correlates. Front Pharmacol. 2019;10:402.
Roerig JL. Clozapine augmentation strategies. Ment Health Clin. 2019;9(6):336–48.
Bioque M, Parellada E, García-Rizo C, Amoretti S, Fortea A, Oriolo G, et al. Clozapine and paliperidone palmitate antipsychotic combination in treatment-resistant schizophrenia and other psychotic disorders: a retrospective 6-month mirror-image study. Eur Psychiatry. 2020;63(1): e71.
Kane JM. Treatment-resistant schizophrenic patients. J Clin Psychiatry. 1996;57(Suppl 9):35–40.
Honer WG, Thornton AE, Chen EY, Chan RC, Wong JO, Bergmann A, et al. Clozapine alone versus clozapine and risperidone with refractory schizophrenia. N Engl J Med. 2006;354(5):472–82.
National Collaborating Centre for Mental H. National institute for health and clinical excellence: guidance. Psychosis and Schizophrenia in Adults: Treatment and management: Updated edition 2014. London; National Institute for Health and Care Excellence (UK); 2014.
Naguy A, Alamiri B. Ultra-treatment resistant schizophrenia- Where do we stand? Asian J Psychiatr. 2019;44:95–6.
Yilmaz Z, Zai CC, Hwang R, Mann S, Arenovich T, Remington G, et al. Antipsychotics, dopamine D(2) receptor occupancy and clinical improvement in schizophrenia: a meta-analysis. Schizophr Res. 2012;140(1–3):214–20.
De Gregorio D, Comai S, Posa L, Gobbi G. d-Lysergic Acid Diethylamide (LSD) as a model of psychosis: mechanism of action and pharmacology. Int J Mol Sci. 2016;17(11):1953.
Lin CH, Chan HY, Hsu CC, Chen FC. Temporal trends in clozapine use at time of discharge among people with schizophrenia at two public psychiatric hospitals in Taiwan, 2006–2017. Sci Rep. 2020;10(1):17984.
Gao L, Hao C, Ma R, Chen J, Zhang G, Chen Y. Synthesis and biological evaluation of a new class of multi-target heterocycle piperazine derivatives as potential antipsychotics. RSC Adv. 2021;11(28):16931–41.
Kang C, Shirley M. Amisulpride: a review in post-operative nausea and vomiting. Drugs. 2021;81(3):367–75.
Genc Y, Taner E, Candansayar S. Comparison of clozapine-amisulpride and clozapine-quetiapine combinations for patients with schizophrenia who are partially responsive to clozapine: a single-blind randomized study. Adv Ther. 2007;24(1):1–13.
Barnes TRE, Leeson V, Paton C, Marston L, Osborn DP, Kumar R, et al. Amisulpride augmentation of clozapine for treatment-refractory schizophrenia: a double-blind, placebo-controlled trial. Ther Adv Psychopharmacol. 2018;8(7):185–97.
Leucht S, Cipriani A, Spineli L, Mavridis D, Orey D, Richter F, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382(9896):951–62.
Assion HJ, Reinbold H, Lemanski S, Basilowski M, Juckel G. Amisulpride augmentation in patients with schizophrenia partially responsive or unresponsive to clozapine. A randomized, double-blind, placebo-controlled trial. Pharmacopsychiatry. 2008;41(1):24–8.
Meltzer HY. Suicide in schizophrenia: risk factors and clozapine treatment. J Clin Psychiatry. 1998;59(Suppl 3):15–20.
Meltzer HY. Treatment of the neuroleptic-nonresponsive schizophrenic patient. Schizophr Bull. 1992;18(3):515–42.
Zhou Y, Li Y, Meng Y, Wang J, Wu F, Ning Y, et al. Neuregulin 3 rs10748842 polymorphism contributes to the effect of body mass index on cognitive impairment in patients with schizophrenia. Transl Psychiatry. 2020;10(1):62.
Ang MJ, Lee S, Kim JC, Kim SH, Moon C. Behavioral tasks evaluating schizophrenia-like symptoms in animal models: a recent update. Curr Neuropharmacol. 2021;19(5):641–64.
Lang X, Zhang W, Song X, Zhang G, Du X, Zhou Y, et al. FOXP2 contributes to the cognitive impairment in chronic patients with schizophrenia. Aging. 2019;11(16):6440–8.
Li Z, Liu L, Lin W, Zhou Y, Zhang G, Du X, et al. NRG3 contributes to cognitive deficits in chronic patients with schizophrenia. Schizophr Res. 2020;215:134–9.
Kay SR, Opler LA, Lindenmayer JP. Reliability and validity of the positive and negative syndrome scale for schizophrenics. Psychiatry Res. 1988;23(1):99–110.
Leucht S, Davis JM, Engel RR, Kane JM, Wagenpfeil S. Defining “response” in antipsychotic drug trials: recommendations for the use of scale-derived cutoffs. Neuropsychopharmacology. 2007;32(9):1903–10.
Mondelli V, Ciufolini S, Belvederi Murri M, Bonaccorso S, Di Forti M, Giordano A, et al. Cortisol and inflammatory biomarkers predict poor treatment response in first episode psychosis. Schizophr Bull. 2015;41(5):1162–70.
Chen H, Fan Y, Zhao L, Hao Y, Zhou X, Guan Y, et al. Successful treatment with risperidone increases 5-HT 3A receptor gene expression in patients with paranoid schizophrenia—data from a prospective study. Brain Behav. 2017;7(9): e00798.
Munro J, Matthiasson P, Osborne S, Travis M, Purcell S, Cobb AM, et al. Amisulpride augmentation of clozapine: an open non-randomized study in patients with schizophrenia partially responsive to clozapine. Acta Psychiatr Scand. 2004;110(4):292–8.
Vauquelin G, Bostoen S, Vanderheyden P, Seeman P. Clozapine, atypical antipsychotics, and the benefits of fast-off D2 dopamine receptor antagonism. Naunyn Schmiedebergs Arch Pharmacol. 2012;385(4):337–72.
Khokhar JY, Henricks AM, Sullivan EDK, Green AI. Unique effects of clozapine: a pharmacological perspective. Adv Pharmacol. 2018;82:137–62.
de Araújo AN, do Nascimento MA, de Sena EP, Baptista AF. Temporomandibular disorders in patients with schizophrenia using antipsychotic agents: a discussion paper. Drug Healthc Patient Saf. 2014;6:21–7.
Hopkins SC, Wilkinson S, Corriveau TJ, Nishikawa H, Nakamichi K, Loebel A, et al. Discovery of nonracemic amisulpride to maximize benefit/risk of 5-HT7 and D2 receptor antagonism for the treatment of mood disorders. Clin Pharmacol Ther. 2021;110(3):808–15.
Alm PA. The dopamine system and automatization of movement sequences: a review with relevance for speech and stuttering. Front Hum Neurosci. 2021;15: 661880.
Gross G, Drescher K. The role of dopamine D(3) receptors in antipsychotic activity and cognitive functions. Handb Exp Pharmacol. 2012;213:167–210.
Laszlovszky I, Barabassy A, Nemeth G. Cariprazine, A Broad-Spectrum Antipsychotic for the treatment of schizophrenia: pharmacology, efficacy, and safety. Adv Ther. 2021;38(7):3652–73.
Dubertret C, Gorwood P, Ades J, Feingold J, Schwartz J, Sokoloff P. Meta-analysis of DRD3 gene and schizophrenia: ethnic heterogeneity and significant association in Caucasians. Am J Med Genet. 1998;81(4):318–22.
Morozova A, Zorkina Y, Pavlov K, Pavlova O, Storozheva Z, Zubkov E, et al. Association of rs4680 COMT, rs6280 DRD3, and rs7322347 5HT2A with clinical features of youth-onset schizophrenia. Front Psychiatry. 2019;10:830.
Moritz AE, Free RB, Sibley DR. Advances and challenges in the search for D2 and D3 dopamine receptor-selective compounds. Cell Signal. 2018;41:75–81.
Stahl S. Drugs for psychosis and mood: unique actions at D3, D2, and D1 dopamine receptor subtypes. CNS Spectr. 2017;22(5):375–84.
Werner F. Neural networks in neurological and psychiatric diseases. Curr Pharm Des. 2019;25(4):374–5.
Bitter I, Lieberman JA, Gaudoux F, Sokoloff P, Groc M, Chavda R, et al. Randomized, double-blind, placebo-controlled study of F17464, a preferential D3 antagonist, in the treatment of acute exacerbation of schizophrenia. Neuropsychopharmacology. 2019;44(11):1917–24.
Zhou X, Wang X, Li R, Yan J, Xiao Y, Li W, et al. Neutrophil-to-Lymphocyte ratio is independently associated with severe psychopathology in schizophrenia and is changed by antipsychotic administration: a large-scale cross-sectional retrospective study. Front Psychiatry. 2020;11: 581061.
Liang Y, Yu X. Effectiveness of amisulpride in Chinese patients with predominantly negative symptoms of schizophrenia: a subanalysis of the ESCAPE study. Neuropsychiatr Dis Treat. 2017;13:1703–12.
Wang YT, Chiu NY, Jou SH, Kuang Yang Y, Hui Lee I, Wang CC, et al. Effects of amisulpride on the cognitive function of patients with schizophrenia who switched from risperidone. Int J Psychiatry Clin Pract. 2008;12(3):180–6.
Kumar S, Chaudhury S. Efficacy of amisulpride and olanzapine for negative symptoms and cognitive impairments: an open-label clinical study. Ind Psychiatry J. 2014;23(1):27–35.
Park JH, Hong JS, Kim SM, Min KJ, Chung US, Han DH. Effects of amisulpride adjunctive therapy on working memory and brain metabolism in the frontal cortex of patients with schizophrenia: a preliminary positron emission tomography/computerized tomography investigation. Clin Psychopharmacol Neurosci. 2019;17(2):250–60.
Molina JD, Quintero J, Garcia-Laredo E, Lopez-Munoz F, Correas-Lauffer J, Barbudo E, et al. Cognitive effects of combined amisulpride and quetiapine treatment in patients with refractory schizophrenia: A naturalistic, prospective study. Am J Ther. 2020;27(4):e346–55.
Salmazo-Silva H, Parente MA, Rocha MS, Baradel RR, Cravo AM, Sato JR, et al. Lexical-retrieval and semantic memory in Parkinson’s disease: the question of noun and verb dissociation. Brain Lang. 2017;165:10–20.
de Boer J, Brederoo S, Voppel A, Sommer I. Anomalies in language as a biomarker for schizophrenia. Curr Opin Psychiatry. 2020;33(3):212–8.
Talati P, Rane S, Skinner J, Gore J, Heckers S. Increased hippocampal blood volume and normal blood flow in schizophrenia. Psychiatry Res. 2015;232(3):219–25.
Schobel SA, Chaudhury NH, Khan UA, Paniagua B, Styner MA, Asllani I, et al. Imaging patients with psychosis and a mouse model establishes a spreading pattern of hippocampal dysfunction and implicates glutamate as a driver. Neuron. 2013;78(1):81–93.
Shin W, Bang M, Kim A, Cho DY, Lee SH. Influence of cytochrome P450 2D6 polymorphism on hippocampal white matter and treatment response in schizophrenia. NPJ Schizophr. 2021;7(1):5.
Tregellas JR, Smucny J, Harris JG, Olincy A, Maharajh K, Kronberg E, et al. Intrinsic hippocampal activity as a biomarker for cognition and symptoms in schizophrenia. Am J Psychiatry. 2014;171(5):549–56.
Heckers S, Konradi C. GABAergic mechanisms of hippocampal hyperactivity in schizophrenia. Schizophr Res. 2015;167(1–3):4–11.
Shin S, Kim S, Seo S, Lee J, Howes O, Kim E, et al. The relationship between dopamine receptor blockade and cognitive performance in schizophrenia: a [11C] -raclopride PET study with aripiprazole. Transl Psychiatry. 2018;8(1):87.
Kvachnina E, Liu G, Dityatev A, Renner U, Dumuis A, Richter D, et al. 5-HT7 receptor is coupled to G alpha subunits of heterotrimeric G12-protein to regulate gene transcription and neuronal morphology. J Neurosci. 2005;25(34):7821–30.
Kobe F, Guseva D, Jensen TP, Wirth A, Renner U, Hess D, et al. 5-HT7R/G12 signaling regulates neuronal morphology and function in an age-dependent manner. J Neurosci. 2012;32(9):2915–30.
Costa L, Sardone LM, Bonaccorso CM, D’Antoni S, Spatuzza M, Gulisano W, et al. Activation of serotonin 5-HT(7) receptors modulates hippocampal synaptic plasticity by stimulation of adenylate cyclases and rescues learning and behavior in a mouse model of fragile X syndrome. Front Mol Neurosci. 2018;11:353.
Juruena MF, de Sena EP, de Oliveira IR. Safety and tolerability of antipsychotics: focus on amisulpride. Drug Healthc Patient Saf. 2010;2:205–11.
Nikiforuk A, Popik P. Amisulpride promotes cognitive flexibility in rats: the role of 5-HT7 receptors. Behav Brain Res. 2013;248:136–40.
Sokoloff P, Le Foll B. The dopamine D3 receptor, a quarter century later. Eur J Neurosci. 2017;45(1):2–19.
Lemercier CE, Schulz SB, Heidmann KE, Kovács R, Gerevich Z. Dopamine D3 receptors inhibit hippocampal gamma oscillations by disturbing CA3 pyramidal cell firing synchrony. Front Pharmacol. 2015;6:297.
Papp M, Gruca P, Lason-Tyburkiewicz M, Litwa E, Niemczyk M, Tota-Glowczyk K, et al. Dopaminergic mechanisms in memory consolidation and antidepressant reversal of a chronic mild stress-induced cognitive impairment`. Psychopharmacology. 2017;234(17):2571–85.
Gou HY, Sun X, Li F, Wang ZY, Wu N, Su RB, et al. The antipsychotic-like effects in rodents of YQA31 involve dopamine D3 and 5-HT1A receptor. Pharmacol Rep. 2017;69(6):1125–30.
Newcomer JW. Second-generation (atypical) antipsychotics and metabolic effects: a comprehensive literature review. CNS Drugs. 2005;19(Suppl 1):1–93.
McKeage K, Plosker G. Amisulpride: a review of its use in the management of schizophrenia. CNS Drugs. 2004;18(13):933–56.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (81401127), the Clinical Research Project of Shanghai Municipal Health Commission (20204Y0173), the Open Project Program of State Key Laboratory of Virtual Reality Technology and Systems, Beihang University (VRLAB2022 B02), the Shanghai Key Laboratory of Psychotic Disorders Open Grant (21-K03), the Scientific Research Project of Traditional Chinese Medicine of Guangdong (20192070), the Guangzhou Municipal Key Discipline in Medicine (2021–2023), and the Science and Technology Plan Project of Guangdong Province (2019B030316001).
Author information
Authors and Affiliations
Contributions
ZZL, MHZ, ZHY and FCW designed the study. MHZ, YJ, NZ, YH, DHS, MJL, HYT and JYY were responsible for recruiting the patients, performing the clinical ratings, and collecting the clinical data. ZHY, QYL, ZJL and QYH collected literatures and cleaned the data. ZZL and MHZ carried out the statistical analyses. LZ coordinated the study. ZZL, MHZ and ZJL wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Regulatory approvals for this study were obtained from the Institutional Review Board of Shanghai Pudong New Area Mental Health Center (No. 2018008). All participants gave their informed consent before their inclusion in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhu, MH., Liu, ZJ., Hu, QY. et al. Amisulpride augmentation therapy improves cognitive performance and psychopathology in clozapine-resistant treatment-refractory schizophrenia: a 12-week randomized, double-blind, placebo-controlled trial. Military Med Res 9, 59 (2022). https://doi.org/10.1186/s40779-022-00420-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40779-022-00420-0