Abstract
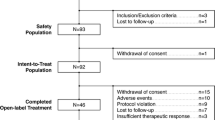
Schizophrenia is a devastating psychiatric disorder. Clozapine has long been the gold standard for treatment of patients with treatment-resistant schizophrenia; however, some patients are only partially responsive to clozapine treatment. Augmentation of clozapine treatment might enhance its effectiveness in partial responders, but only a few studies have investigated possible augmentation strategies. This study compared the effectiveness and tolerability of the combination of amisulpride and clozapine with the combination of quetiapine and clozapine in patients who were only partially responsive to clozapine monotherapy. Fifty-six treatment-resistant patients who were partially responsive to clozapine were randomly assigned to receive amisulpride or quetiapine along with an ongoing stable dose of clozapine. Fifty patients completed the study. Patients were evaluated at baseline and at the first, third, sixth, and eighth weeks. Efficacy measures consisted of the Brief Psychiatric Rating Scale (BPRS), the Scale for the Assessment of Negative Symptoms (SANS), the Scale for the Assessment of Positive Symptoms (SAPS), and the Clinical Global Impression (CGI) scale. Tolerability and adverse effects were assessed with the Udvalg for Kliniske Undersogelser (UKU) Side Effect Rating Scale and the Simpson Angus Scale (SAS). A substantial improvement occurred in both groups by the end of the eighth week; however, the improvement associated with amisulpride was significantly greater than that seen with quetiapine. This difference was noted as early as the third week of follow-up in terms of CGI scores, and by the sixth week with regard to BPRS, SANS, and SAPS scores. Both drugs were well tolerated, as measured by UKU and SAS. Improvement favoring clozapine+amisulpride could be attributed to the selective D2/D3 binding property of amisulpride, which had an additional effect in improving symptoms of schizophrenia. The authors concluded that amisulpride seems to be effective and well tolerated for augmentation purposes in clozapine-resistant patients.
Similar content being viewed by others
References
Essock SM, Hargreaves WA. Clozapine eligibility among state hospital patients.Schizophr Bull. 1996;22:15–25.
Barnes TRE, Buckley P, Schultz SC. Treatment resistant schizophrenia. In: Hirsch SR, Weinberger D, eds.Schizophrenia. Oxford, Mass: Blackwell Science; 2003:489–517.
Taner E, Cosar B, Isik E. The effects of the changes in symptoms of schizophrenia over functioning: two years of clozapine follow-up study [in Turkish].Psych Turkey. 2002;4:84–91.
McEvoy JP, Hogarty GE, Steingard S. Optimal dose of neuroleptic in acute schizophrenia: a controlled study of the neuroleptic threshold and higher haloperidol dose.Arch Gen Psychiatry. 1991;48:739–745.
Kane JM. The current status of neuroleptic therapy.J Clin Psychiatry. 1989;50:322–328.
Kane JM. Clinical efficacy of clozapine in treatment refractory schizophrenia: an overview.Br J Psychiatry Suppl. 1992;17:41–45.
Conley RR, Buchanan RW. Evaluation of treatment-resistant schizophrenia.Schizophr Bull. 1997;23:663–674.
Conley R, Kelly DL. Management of treatment resistance in schizophrenia.Biol Psychiatry. 2001; 50:898–911.
Meltzer HY. The role of serotonin in antipsychotic drug action.Neuropsychopharmacology. 1999; 21:106–115.
Buckley A, Miller J, Olsen D, Garver DD, Csernansky J. When symptoms persist: clozapine augmentation strategies.Schizophr Bull. 2001;27:615–628.
Chong SA, Remington G. Clozapine augmentation: safety and efficacy.Schizophr Bull. 2000; 26:421–440.
Freudenreich O, Goff DC. Antipsychotic combination therapy in schizophrenia: a review of efficacy and risks of current combinations.Acta Psychiatr Scand. 2002;106:323–330.
Buchanan RW, Kirkpatrick B, Bryant N, Ball P, Breier A. Fluoxetine augmentation of clozapine treatment in patients with schizophrenia.Am J Psychiatry. 1996;153:1625–1627.
Silver H, Kushnir M, Kaplan A. Fluvoxamine augmentation in clozapine-resistant schizophrenia: an open pilot study.Biol Psychiatry. 1996;40:671–674.
Small JG, Klapper MH, Malloy FW, Steadman TM. Tolerability and efficacy of clozapine combined with lithium in schizophrenia and schizoaffective disorder.J Clin Psychopharmacol. 2003;23:223–228.
Tiihonen J, Hallikainen T, Ryynanen OP, et al. Lamotrigine in treatment-resistant schizophrenia: a randomized placebo-controlled crossover trial.Biol Psychiatry. 2003;54:1241–1248.
Evins AE, Fitzgerald SM, Wine L, Rosselli R, Goff DC. Placebo-controlled trial of glycine added to clozapine in schizophrenia.Am J Psychiatry. 2000;157:826–828.
Carpenter WT, Breier A, Buchanan RW, Kirkpatrick B, Shepard P, Weiner E. Mazindol treatment of negative symptoms.Neuropsychopharmacology. 2000;23:365–374.
Shiloh R, Zemishlany Z, Aizenberg D, et al. Sulpiride augmentation in people with schizophrenia partially responsive to clozapine: a double-blind, placebo-controlled study.Br J Psychiatry. 1997; 171:569–573.
Anil AEY, Kivircik BBA, Turgut TI, et al. A double-blind controlled study of adjunctive treatment with risperidone in schizophrenic patients partially responsive to clozapine: efficacy and safety.J Clin Psychiatry. 2005;66:63–72.
Josiassen RC, Joseph A, Kohegyi E, et al. Clozapine augmented with risperidone in the treatment of schizophrenia: a randomized, double-blind, placebo-controlled trial.Am J Psychiatry. 2005;162:130–136.
Henderson DC, Goff DC. Risperidone as an adjunct to clozapine therapy in chronic schizophrenics.J Clin Psychiatry. 1996;57:395–397.
Scatton B, Claustre Y, Cudennec A, et al. Amisulpride: from animal pharmacology to therapeutic action.Int Clin Psychopharmacol. 1997;12(suppl 2):29–36.
Leucht S. Amisulpride, a selective dopamine antagonist and atypical antipsychotic: results of a meta-analysis of randomized controlled trials.Int J Neuropsychopharmacol. 2004;7(suppl 1): 15–20.
Munro J, Matthiasson P, Osborne S, et al. Amisulpride augmentation of clozapine: an open non-randomized study in patients with schizophrenia partially responsive to clozapine.Acta Psychiatr Scand. 2004;110:292–298.
Agelink MW, Kavuk I, Ak I. Clozapine with amisulpride for refractory schizophrenia.Am J Psychiatry. 2004;161:924–925.
Zink M, Knopf U, Henn FA, et al. Combination of clozapine and amisulpride in treatmentresistant schizophrenia—case reports and review of the literature.Pharmacopsychiatry. 2004;37: 26–31.
American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition. Washington, DC: American Psychiatric Association; 1994.
Overall JE, Gorham DR. The Brief Psychiatric Rating Scale.Psychol Rep. 1962;10:799–812.
Woerner MG, Mennuzza S, Kane JM. Anchoring the BPRS-A: an aid to improved reliability.Psychopharmacol Bull. 1988;24:112–117.
Andreasen NC. Methods for assessing positive and negative symptoms.Mod Probl Pharmacopsychiatry. 1990;24:73–88.
Guy W, ed.ECDEU Assessment Manual for Psychopharmacology. Publication no. ADM 76-338. Washington, DC: US Department of Health, Education, and Welfare; 1976:218–222.
Lingjaerde O, Ahlfors UG, Bech P, Dencker SJ, Elgen K. The UKU side effect rating scale: a new comprehensive rating scale for psychotropic drugs and cross-sectional study of side effects in neuroleptic-treated patients.Acta Psychiatr Scand Suppl. 1987;334:1–100.
Simpson GM, Angus JWS. A rating scale for extrapyramidal side effects.Acta Psychiatr Scand Suppl. 1970;212:11–19.
Isik E. Equivalent doses of antipsychotic drugs. In: Isik E, ed.Current Schizophrenia. Norwich, England: Format Publishing; 2006;378–380.
Henderson DC, Kunkel L, Nguyen DD, et al. An explanatory open label trial of aripiprazole as an adjuvant to clozapine therapy in chronic schizophrenia.Acta Psychiatr Scand. 2006;113: 142–147.
Stern RG, Schmeidler J, Davidson M. Limitations of controlled augmentation trials in schizophrenia.Biol Psychiatry. 1997;42:138–143.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Genç, Y., Taner, E. & Candansayar, S. Comparison of clozapine-amisulpride and clozapine-quetiapine combinations for patients with schizophrenia who are partially responsive to clozapine: A single-blind randomized study. Adv Therapy 24, 1–13 (2007). https://doi.org/10.1007/BF02849987
Issue Date:
DOI: https://doi.org/10.1007/BF02849987